Advanced Synthesis of Asymmetric Thiophene [7] Helicene Isomers for Next-Gen OLEDs
Introduction to Asymmetric Thiophene [7] Helicene Technology
The rapid evolution of the organic electronics sector demands materials with precisely tuned electronic properties, driving the search for novel molecular architectures beyond traditional symmetrical structures. Patent CN111057087A introduces a groundbreaking class of asymmetric thiophene [7] helicene isomers, representing a significant leap forward in the design of organic semiconductor materials. Unlike their symmetrical counterparts, these asymmetric isomers offer enhanced structural diversity, allowing for finer modulation of HOMO-LUMO energy levels and charge transport characteristics. This technological advancement is particularly critical for the development of high-performance organic light-emitting diodes (OLEDs), organic field-effect transistors (OFETs), and organic solar cells, where charge mobility and stability are paramount. By leveraging a sophisticated multi-step synthetic strategy involving palladium-catalyzed coupling and lithium-mediated cyclization, this patent provides a robust pathway to access these complex polycyclic aromatic hydrocarbons with high purity and yield.
For R&D directors and procurement specialists in the electronic chemicals industry, understanding the nuances of this synthesis is essential for securing a reliable supply chain of next-generation display and energy materials. The ability to produce specific isomers with defined asymmetry opens new avenues for device optimization, potentially leading to longer lifespans and higher efficiency in commercial electronic products. As a leading fine chemical manufacturer, we recognize the strategic importance of mastering these complex synthetic routes to support the global transition toward flexible and printable electronics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of thiophene-based helicenes has been dominated by the production of symmetrical structures, as illustrated in the prior art reaction schemes found in the patent background. These conventional methods often rely on repetitive coupling strategies that inherently limit the structural variety of the final product. The primary drawback of symmetrical thiophene [7] helicenes is their restricted ability to fine-tune electronic properties; the uniform distribution of electron density can lead to suboptimal charge injection barriers or limited solubility in processing solvents. Furthermore, traditional oxidative cyclization methods used to close the helicene backbone frequently suffer from harsh reaction conditions, requiring strong oxidants that can degrade sensitive functional groups or lead to over-oxidation by-products. This lack of regioselectivity often results in complex mixture profiles that are difficult and costly to separate, posing a significant bottleneck for scaling up production to meet the stringent purity requirements of the semiconductor industry.
![Prior art synthesis routes showing symmetrical thiophene [7] helicene compounds](/insights/img/asymmetric-thiophene-helicene-synthesis-oled-supplier-20260305060320-01.webp)
The Novel Approach
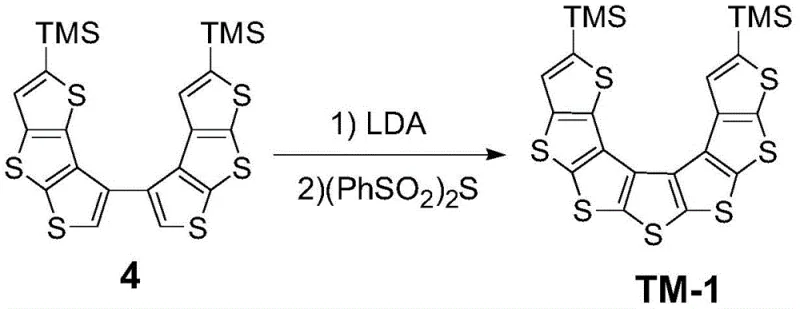
In stark contrast to these legacy techniques, the methodology disclosed in CN111057087A employs a modular assembly strategy that enables the precise construction of asymmetric skeletons. The core innovation lies in the sequential combination of Suzuki-Miyaura cross-coupling and a unique LDA-mediated thio-cyclization. This approach allows chemists to independently modify different segments of the helicene framework before the final ring closure, granting unprecedented control over the molecular geometry. By utilizing trimethylsilyl (TMS) groups as protecting and directing groups, the synthesis achieves high regioselectivity during the lithiation steps, ensuring that the sulfur atoms are incorporated at the exact positions required to form the desired isomer. This level of precision not only enhances the electronic performance of the material, evidenced by mobilities reaching 0.1 cm²/V·s, but also streamlines the purification workflow. The result is a versatile platform capable of generating multiple isomers (TM-1, TM-2, TM-3) from common intermediates, offering a cost-effective solution for screening and optimizing organic semiconductor formulations.
Mechanistic Insights into LDA-Mediated Thio-Cyclization
The cornerstone of this synthetic breakthrough is the final ring-closing step, which utilizes Lithium Diisopropylamide (LDA) and diphenyl disulfone ((PhSO2)2S) to forge the central thiophene rings. Mechanistically, this process begins with the regioselective deprotonation of the precursor dithienothiophene dimer at the alpha-positions adjacent to the sulfur atoms. LDA, being a strong, non-nucleophilic base, effectively generates the dilithiated species at cryogenic temperatures (typically -70°C to -90°C), preventing unwanted side reactions such as nucleophilic attack on the thiophene ring. Subsequent addition of diphenyl disulfone acts as an electrophilic sulfur source, facilitating the formation of new carbon-sulfur bonds through a nucleophilic substitution mechanism. This intramolecular cyclization effectively locks the molecule into its helical conformation, creating the rigid, conjugated backbone necessary for efficient charge transport. The use of sulfone reagents instead of elemental sulfur or thionyl chloride offers a milder and more controllable reaction environment, significantly reducing the formation of polymeric by-products.
From an impurity control perspective, this mechanism is highly advantageous for commercial manufacturing. Traditional oxidative coupling often generates quinoid-like impurities or over-oxidized sulfones that are structurally similar to the product and difficult to remove. In this novel pathway, the by-products are primarily phenyl sulfinate salts and unreacted starting materials, which possess vastly different polarities and can be easily separated via standard silica gel chromatography or recrystallization. Furthermore, the absence of transition metal catalysts in this specific cyclization step eliminates the risk of metal contamination, a critical quality attribute for electronic grade materials where trace metals can act as charge traps and degrade device performance. The robustness of this mechanism is demonstrated by the consistent yields of 73% to 78% across different isomers, indicating a high degree of process reliability suitable for industrial scale-up.
How to Synthesize Asymmetric Thiophene [7] Helicene Efficiently
The synthesis of these high-value organic semiconductors follows a logical three-stage progression designed to maximize yield and minimize waste. The process begins with the preparation of key boronic ester intermediates via lithiation-borylation, followed by a palladium-catalyzed dimerization, and concludes with the signature thio-cyclization. Each step requires strict control of atmospheric conditions, particularly the exclusion of moisture and oxygen, to ensure the stability of the organolithium and organopalladium species involved. Detailed operational parameters, including specific solvent choices like anhydrous THF or toluene and precise temperature ramps, are critical for reproducing the high purity levels reported in the patent data. For process chemists looking to implement this route, adherence to the standardized protocols outlined below is essential for achieving the target specifications.
- Perform lithiation of the bromo-thiophene precursor using t-BuLi or n-BuLi at cryogenic temperatures (-70 to -90°C), followed by quenching with pinacol borate to generate the boronic ester intermediate.
- Execute a Palladium-catalyzed Suzuki-Miyaura cross-coupling reaction between the boronic ester and a brominated dithienothiophene derivative using bases like K2CO3 or Cs2CO3 in THF or Toluene.
- Conduct the final ring-closing reaction by treating the dimer with LDA for deprotonation, followed by the addition of diphenyl disulfone ((PhSO2)2S) to form the asymmetric thiophene [7] helicene core.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route offers tangible benefits in terms of cost structure and supply security. The shift towards a modular synthesis using widely available commodity chemicals reduces dependency on exotic or single-source reagents. By breaking down the complex helicene structure into manageable building blocks, manufacturers can optimize inventory levels and mitigate the risks associated with raw material shortages. Additionally, the high efficiency of the coupling and cyclization steps translates directly into improved material throughput, allowing suppliers to meet increasing demand without proportional increases in production capacity or capital expenditure.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts in the final cyclization step represents a significant opportunity for cost optimization. Traditional methods often require expensive palladium or nickel catalysts for ring closure, followed by costly purification steps to remove trace metal residues to ppb levels. By utilizing an organolithium/sulfone strategy for the final ring formation, the process drastically simplifies the downstream processing requirements. This reduction in purification complexity lowers the consumption of silica gel, solvents, and metal scavengers, resulting in substantial cost savings per kilogram of finished product. Furthermore, the high yields observed in the intermediate steps (often exceeding 80%) minimize the loss of valuable precursors, enhancing the overall atom economy of the process.
- Enhanced Supply Chain Reliability: The synthetic route relies on robust, well-understood chemical transformations such as Suzuki coupling and lithiation, which are staples of the fine chemical industry. This familiarity means that the process can be easily transferred between different manufacturing sites or contract manufacturing organizations (CMOs) without extensive re-validation. The reagents used, including LDA, pinacol borates, and basic palladium catalysts, are globally sourced commodities with stable supply chains, reducing the risk of disruption due to geopolitical factors or raw material scarcity. This reliability ensures consistent delivery schedules for downstream electronics manufacturers who depend on just-in-time inventory models.
- Scalability and Environmental Compliance: From an environmental and safety standpoint, the process is designed with scalability in mind. The use of diphenyl disulfone as a sulfur source avoids the generation of hazardous hydrogen sulfide gas, a common by-product in traditional thiol-based syntheses. The reaction conditions, while requiring low temperatures for lithiation, utilize standard cryogenic equipment available in most pilot and production plants. The waste streams generated are primarily organic salts and solvents, which can be managed through established recovery and treatment protocols. This alignment with green chemistry principles facilitates regulatory compliance and supports the sustainability goals of major electronics brands seeking to reduce the carbon footprint of their supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of asymmetric thiophene [7] helicenes. These insights are derived directly from the experimental data and technical specifications provided in the patent literature, offering a clear understanding of the material's capabilities and the manufacturing process.
Q: What are the key electronic properties of the synthesized asymmetric thiophene [7] helicenes?
A: The synthesized isomers exhibit excellent organic semiconductor properties, including hole mobilities ranging from 0.07 to 0.1 cm²/V·s, threshold voltages between -58V and -62V, and high on-off ratios up to 1.5 x 10^6, making them superior to existing symmetrical heptathiophenes.
Q: How does the novel LDA-mediated cyclization improve the synthesis compared to traditional methods?
A: The novel approach utilizes LDA and diphenyl disulfone for the final ring closure, which avoids the use of expensive transition metal catalysts in the critical cyclization step. This significantly simplifies the purification process by eliminating the need for rigorous heavy metal scavenging, thereby reducing production costs and environmental impact.
Q: Are these asymmetric isomers suitable for large-scale commercial production?
A: Yes, the synthetic route relies on standard organic transformations such as Suzuki coupling and lithiation, which are well-established in industrial settings. The use of commercially available reagents like LDA, pinacol borates, and palladium catalysts ensures that the process is scalable from gram-scale laboratory synthesis to multi-kilogram commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Asymmetric Thiophene [7] Helicene Supplier
At NINGBO INNO PHARMCHEM, we understand that the transition from laboratory discovery to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team specializes in the scale-up of complex organic semiconductors, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are equipped with state-of-the-art cryogenic reactors and stringent purity specifications to ensure that every batch of asymmetric thiophene [7] helicene meets the rigorous demands of the OLED and OFET industries. Our rigorous QC labs employ advanced analytical techniques, including HPLC, GC-MS, and ICP-MS, to guarantee the absence of metal impurities and isomeric contaminants that could compromise device performance.
We invite technology leaders and procurement professionals to collaborate with us to optimize their material sourcing strategies. Whether you require custom synthesis of specific isomers or large-volume supply of standard grades, our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your project needs. Contact us today to request specific COA data and route feasibility assessments, and let us help you accelerate the development of your next-generation electronic devices with high-purity, reliably sourced organic semiconductor materials.
