Advanced DTPB-Initiated Synthesis of 3-Ethylcyanobenzothiophene for Commercial Scale-Up
Advanced DTPB-Initiated Synthesis of 3-Ethylcyanobenzothiophene for Commercial Scale-Up
Introduction to Novel Benzothiophene Synthesis Technology
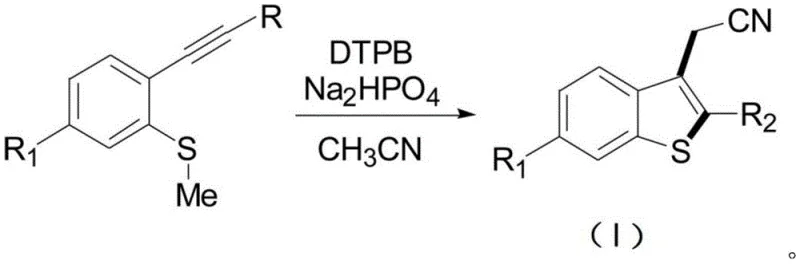
The pharmaceutical and agrochemical industries continuously demand efficient pathways for constructing complex heterocyclic scaffolds, particularly benzothiophene derivatives which serve as critical pharmacophores in numerous therapeutic agents. Patent CN111675691A introduces a groundbreaking one-step synthesis method for 3-ethylcyanobenzothiophene compounds, utilizing a DTPB-initiated tandem cyclization reaction that fundamentally shifts the paradigm from traditional transition metal catalysis. This innovative approach addresses the longstanding challenges of high operational costs and environmental hazards associated with conventional methods, offering a streamlined route that utilizes acetonitrile as both solvent and reactant. By leveraging a radical mechanism under nitrogen protection at controlled temperatures between 110°C and 150°C, this technology enables the direct formation of the benzothiophene core with remarkable simplicity. For R&D directors and procurement managers seeking a reliable fine chemical intermediates supplier, this patent represents a significant opportunity to optimize supply chains and reduce manufacturing overheads through simplified process chemistry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of benzothiophene derivatives has relied heavily on transition metal catalysts such as palladium, copper, or gold, which introduce substantial complexities into the manufacturing workflow. These traditional methods often require harsh reaction conditions, expensive ligands, and generate significant amounts of halogen-containing waste liquids that pose severe environmental compliance challenges. Furthermore, the removal of trace heavy metals from the final active pharmaceutical ingredients necessitates additional purification steps, drastically increasing production time and cost. The reliance on precious metals also creates supply chain vulnerabilities, as price fluctuations and availability issues can disrupt production schedules for high-purity benzothiophene derivatives. Consequently, the industry has long sought a metal-free alternative that maintains high yield and selectivity without the burden of toxic waste management and complex downstream processing.
The Novel Approach
The novel approach detailed in the patent utilizes dodecyl triphenyl phosphine bromide (DTPB) as a radical initiator to drive the intramolecular cyclization of 2-ethynyl anisole sulfide derivatives in a single operational step. This metal-free strategy eliminates the need for costly transition metals and avoids the generation of hazardous halogenated byproducts, aligning perfectly with green chemistry principles and modern environmental regulations. By employing acetonitrile as the solvent, the process further simplifies the reaction system, reducing the number of reagents required and minimizing the potential for side reactions that complicate purification. The reaction proceeds smoothly under nitrogen protection with disodium hydrogen phosphate as an additive, ensuring robust control over the radical species generated during the transformation. This methodology not only enhances the safety profile of the synthesis but also significantly lowers the barrier for commercial scale-up of complex heterocyclic compounds by reducing equipment corrosion and waste treatment costs.
Mechanistic Insights into DTPB-Catalyzed Cyclization
The core of this technological breakthrough lies in the radical mechanism initiated by the thermal decomposition of DTPB, which generates phosphorus-centered radicals that abstract hydrogen atoms to trigger the cyclization cascade. Unlike electrophilic cyclization methods that rely on stoichiometric oxidants like iodine or NBS, this radical pathway operates under neutral conditions that are compatible with a wide range of functional groups on the aromatic ring. The presence of disodium hydrogen phosphate plays a crucial role in buffering the reaction environment and stabilizing intermediate species, ensuring that the cyclization proceeds with high regioselectivity to form the desired 3-ethylcyanobenzothiophene scaffold. This mechanistic understanding allows chemists to fine-tune reaction parameters such as temperature and initiator loading to maximize yield while minimizing the formation of polymeric byproducts often seen in radical reactions. The ability to control the radical flux precisely is key to achieving the moderate to good yields reported across various substrate derivatives.
Impurity control is another critical aspect where this novel mechanism offers distinct advantages over traditional transition metal-catalyzed routes. The absence of metal catalysts eliminates the risk of metal-ligand complexes forming stable impurities that are difficult to remove during crystallization or chromatography. Furthermore, the one-step nature of the reaction reduces the accumulation of intermediates that could otherwise degrade or react further to form unknown impurities, thereby simplifying the impurity profile of the final product. The use of acetonitrile as a solvent also aids in maintaining a homogeneous reaction mixture, preventing localized hot spots that could lead to thermal decomposition of the product. For quality control teams, this translates to a more consistent product batch-to-batch, facilitating easier validation and regulatory approval for high-purity benzothiophene derivatives intended for sensitive pharmaceutical applications.
How to Synthesize 3-Ethylcyanobenzothiophene Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to the stoichiometry of the radical initiator and the control of reaction temperature to ensure optimal conversion. The patent specifies that DTPB should be used in an amount of 3.4 to 4.5 equivalents, with 4.0 equivalents being the preferred loading to balance reaction rate and cost efficiency. The reaction temperature is maintained between 110°C and 150°C, preferably 130°C to 135°C, for a duration of 20 to 24 hours to allow the radical chain reaction to reach completion without excessive energy consumption. Detailed standardized synthesis steps see the guide below for precise operational parameters and workup procedures that ensure high recovery of the target compound.
- Prepare the reaction mixture by combining 2-ethynyl anisole sulfide derivatives, DTPB initiator, and disodium hydrogen phosphate in acetonitrile solvent.
- Heat the reaction mixture to 110-150°C under nitrogen protection and stir for 20-24 hours to ensure complete radical cyclization.
- Extract the crude product with dichloromethane, dry over anhydrous sodium sulfate, and purify via column chromatography using petroleum ether and ethyl acetate.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthesis method offers transformative benefits for procurement managers and supply chain heads looking to optimize costs and ensure continuity of supply for critical intermediates. The elimination of expensive transition metal catalysts directly translates to substantial cost savings in raw material procurement, while the simplified one-step process reduces labor hours and equipment occupancy time. The use of common solvents like acetonitrile and readily available inorganic salts like disodium hydrogen phosphate ensures that the supply chain is not dependent on specialized or scarce reagents that could cause production delays. Additionally, the reduced environmental footprint of the process lowers waste disposal costs and minimizes regulatory hurdles, making it a sustainable choice for long-term manufacturing partnerships. These factors combined create a robust economic case for adopting this technology in the production of reliable fine chemical intermediates supplier portfolios.
- Cost Reduction in Manufacturing: The removal of precious metal catalysts such as palladium and gold eliminates a major cost driver in the synthesis of heterocyclic compounds, significantly reducing the bill of materials for each production batch. Furthermore, the one-step nature of the reaction reduces energy consumption and labor costs associated with multi-step purification and intermediate isolation, leading to drastic simplification of the overall manufacturing process. The ability to use acetonitrile as both solvent and reactant further streamlines the process, minimizing solvent recovery costs and reducing the volume of waste generated. These cumulative efficiencies result in substantial cost savings that can be passed on to customers or reinvested into process optimization initiatives.
- Enhanced Supply Chain Reliability: By relying on commercially available and stable reagents like DTPB and disodium hydrogen phosphate, the manufacturing process is insulated from the volatility of the precious metal market and supply chain disruptions. The robustness of the radical mechanism ensures consistent yields across different batches, reducing the risk of production failures that could impact delivery schedules for high-purity agrochemical intermediates. The simplified workflow also allows for faster turnaround times from order to delivery, enabling manufacturers to respond more agilely to fluctuating market demands. This reliability is crucial for maintaining trust with downstream partners who depend on timely delivery of key building blocks for their own synthesis campaigns.
- Scalability and Environmental Compliance: The metal-free nature of this synthesis aligns perfectly with increasingly stringent environmental regulations regarding heavy metal discharge and waste management, facilitating easier permitting and compliance audits. The process generates minimal hazardous waste compared to traditional halogen-mediated cyclizations, reducing the burden on waste treatment facilities and lowering the overall environmental impact of production. Scalability is enhanced by the use of standard reaction conditions that do not require specialized high-pressure or cryogenic equipment, allowing for seamless transfer from laboratory to commercial scale. This ensures that the production of complex heterocyclic compounds can be expanded to meet growing market demand without compromising on safety or sustainability standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this DTPB-initiated synthesis route for benzothiophene derivatives. These answers are derived directly from the patent data and practical considerations for scaling this chemistry in an industrial setting. Understanding these details is essential for technical teams evaluating the feasibility of integrating this method into their existing manufacturing workflows. The insights provided here aim to clarify the operational advantages and potential applications of this novel technology.
Q: What are the advantages of using DTPB over traditional transition metal catalysts?
A: DTPB acts as a metal-free radical initiator, eliminating the need for expensive palladium or gold catalysts and avoiding heavy metal contamination in the final product, which simplifies purification and reduces environmental waste.
Q: How does this method improve substrate adaptability for benzothiophene derivatives?
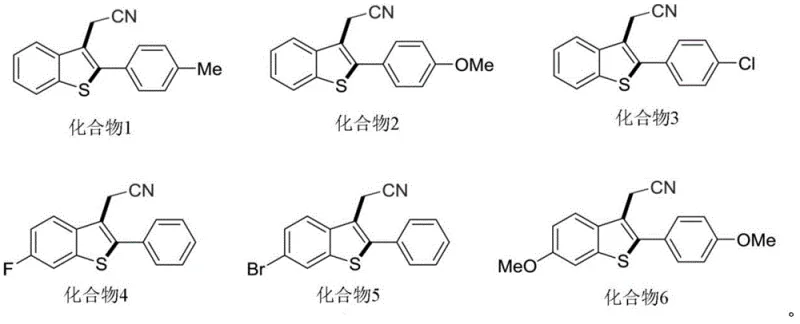
A: The radical mechanism tolerates various substituents on the aromatic ring, including methoxy, fluoro, and bromo groups, allowing for the synthesis of diverse derivatives without harsh reaction conditions that might degrade sensitive functional groups.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the one-step process uses common reagents like acetonitrile and operates at moderate temperatures, making it highly scalable with simplified operational procedures and reduced energy consumption compared to multi-step traditional methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Ethylcyanobenzothiophene Supplier
NINGBO INNO PHARMCHEM stands at the forefront of chemical manufacturing innovation, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production for complex heterocyclic intermediates. Our technical team is fully equipped to adapt the DTPB-initiated radical cyclization process described in CN111675691A to meet your specific volume and purity requirements, ensuring stringent purity specifications are met through our rigorous QC labs. We understand the critical nature of supply chain continuity for pharmaceutical and agrochemical clients, and our infrastructure is designed to deliver consistent quality and reliability. By partnering with us, you gain access to a CDMO expert capable of navigating the complexities of metal-free synthesis to deliver high-value intermediates efficiently.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can optimize your current supply chain and reduce overall manufacturing costs. Request a Customized Cost-Saving Analysis today to evaluate the economic benefits of switching to this metal-free protocol for your specific project needs. Our team is ready to provide specific COA data and route feasibility assessments to support your decision-making process. Let us help you secure a stable and cost-effective supply of high-quality benzothiophene derivatives for your next development phase.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
