Mastering Chiral Resolution for Complex Pyridoindole Pharmaceutical Intermediates at Commercial Scale
The escalating regulatory demands for single-enantiomer pharmaceuticals have fundamentally reshaped the landscape of fine chemical manufacturing, compelling producers to adopt sophisticated chiral separation technologies. As highlighted in patent CN108409731B, the efficient resolution of complex heterocyclic compounds containing multiple chiral centers remains a critical bottleneck in the synthesis of advanced therapeutic agents. This specific intellectual property details a robust methodology for the chiral resolution of 1-aryl-1H-pyridine[3,4-b]indole-3-carboxylate derivatives, which are pivotal scaffolds in modern drug discovery. The technology addresses the inherent difficulties in separating diastereomers that possess remarkably similar physical properties, offering a pathway to high optical purity without relying on prohibitively expensive preparative chromatography. For global supply chains, mastering such resolution techniques is not merely a technical exercise but a strategic imperative to ensure compliance with stringent FDA and EMA guidelines regarding chiral impurities.
In the realm of asymmetric synthesis, the presence of two chiral centers within a single molecular framework exponentially increases the complexity of purification, often resulting in four possible stereoisomers. Traditional approaches frequently struggle to distinguish between diastereomers when their polarity and solubility profiles are nearly identical, leading to significant yield losses and excessive waste generation. 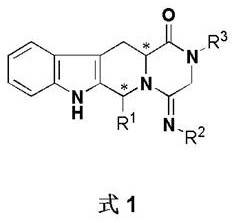 The conventional reliance on column chromatography for such separations is increasingly viewed as unsustainable for large-scale production due to high solvent usage and limited throughput. In contrast, the novel approach described in the reference patent leverages the subtle thermodynamic differences in crystal lattice formation between diastereomeric salts. By introducing a specifically designed chiral amine resolving agent, the process converts the difficult-to-separate enantiomeric mixture into diastereomeric salts that exhibit distinct solubility characteristics, facilitating separation through controlled crystallization rather than complex mechanical separation.
The conventional reliance on column chromatography for such separations is increasingly viewed as unsustainable for large-scale production due to high solvent usage and limited throughput. In contrast, the novel approach described in the reference patent leverages the subtle thermodynamic differences in crystal lattice formation between diastereomeric salts. By introducing a specifically designed chiral amine resolving agent, the process converts the difficult-to-separate enantiomeric mixture into diastereomeric salts that exhibit distinct solubility characteristics, facilitating separation through controlled crystallization rather than complex mechanical separation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the isolation of optically pure intermediates from racemic mixtures or diastereomeric blends has relied heavily on chromatographic techniques or enzymatic kinetic resolution, both of which carry significant operational drawbacks for industrial applications. Chromatographic separation, while effective on a laboratory scale, becomes economically unviable when scaled to metric ton quantities due to the massive volumes of organic solvents required and the slow cycle times associated with column loading and elution. Furthermore, enzymatic methods often suffer from narrow substrate specificity, requiring extensive screening to find a biocatalyst that accepts bulky heterocyclic substrates like pyridoindoles. When dealing with compounds possessing two chiral centers, the physical similarity between the resulting diastereomers often renders standard recrystallization ineffective, forcing manufacturers to accept lower purities or engage in repetitive purification loops that erode profit margins and extend lead times significantly.
The Novel Approach
The innovative strategy outlined in the patent data circumvents these limitations by employing a chemical resolution method centered on the formation of diastereomeric salts using S-1-(2-tert-butylphenyl)ethylamine. This resolving agent is selected for its ability to interact selectively with one specific stereoisomer of the target amino acid intermediate, creating a salt complex that precipitates out of solution under controlled temperature conditions. 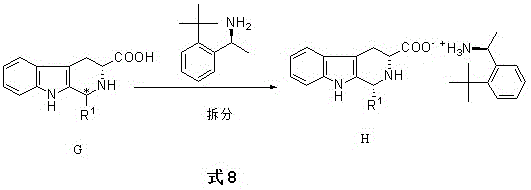 This precipitation allows for a simple filtration step to isolate the desired isomer with high efficiency, bypassing the need for complex chromatography. Crucially, the process is designed with circular economy principles in mind; the mother liquor and the dissociation steps allow for the recovery of the chiral amine resolving agent. This recoverability transforms the resolving agent from a consumable cost center into a recyclable asset, drastically reducing the raw material cost per kilogram of the final active pharmaceutical ingredient and minimizing the environmental footprint associated with chiral waste disposal.
This precipitation allows for a simple filtration step to isolate the desired isomer with high efficiency, bypassing the need for complex chromatography. Crucially, the process is designed with circular economy principles in mind; the mother liquor and the dissociation steps allow for the recovery of the chiral amine resolving agent. This recoverability transforms the resolving agent from a consumable cost center into a recyclable asset, drastically reducing the raw material cost per kilogram of the final active pharmaceutical ingredient and minimizing the environmental footprint associated with chiral waste disposal.
Mechanistic Insights into Diastereomeric Salt Crystallization
The success of this resolution protocol hinges on the precise manipulation of intermolecular forces during the crystallization phase, where the steric bulk of the tert-butyl group on the resolving amine plays a pivotal role. When the racemic amino acid intermediate reacts with the optically pure amine, two distinct diastereomeric salts are formed, each possessing a unique three-dimensional arrangement that influences how the molecules pack into a crystal lattice. One diastereomer forms a more stable, less soluble crystal structure that precipitates preferentially when the solution is cooled from reflux temperatures to approximately 30°C, while the other remains in the supernatant. 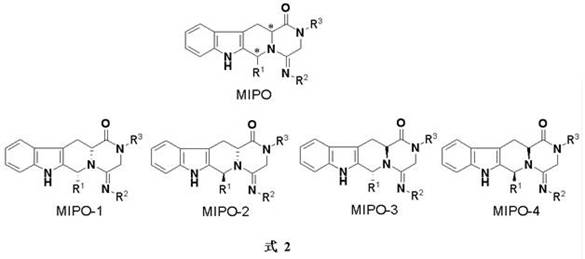 This differential solubility is the driving force behind the separation, allowing the target isomer to be harvested as a solid cake while the unwanted isomer stays dissolved. The mechanism also accounts for the basicity of the nitrogen atoms within the pyridoindole ring system, ensuring that the acid-base reaction proceeds quantitatively to form the salt before crystallization begins, thereby maximizing the theoretical yield of the separation process.
This differential solubility is the driving force behind the separation, allowing the target isomer to be harvested as a solid cake while the unwanted isomer stays dissolved. The mechanism also accounts for the basicity of the nitrogen atoms within the pyridoindole ring system, ensuring that the acid-base reaction proceeds quantitatively to form the salt before crystallization begins, thereby maximizing the theoretical yield of the separation process.
Beyond the primary separation, the protocol incorporates a sophisticated dissociation and regeneration cycle that ensures the integrity of the chiral information is maintained throughout the workflow. After filtration, the diastereomeric salt cake is treated with a base in a biphasic solvent system, which breaks the ionic bond between the amino acid and the resolving amine. The free chiral amine partitions into the organic layer, from which it can be recovered, dried, and reused in subsequent batches with minimal loss of optical purity. The aqueous layer retains the now optically pure amino acid, which is subsequently acidified and extracted. This meticulous control over the dissociation step prevents racemization, a common pitfall in chiral processing where harsh conditions might invert the stereocenters. The final esterification step restores the molecule to its required ester form, ready for downstream coupling reactions, with the chiral centers locked in the desired configuration as verified by HPLC analysis showing purity levels exceeding 99%.
How to Synthesize 1-Aryl-1H-Pyridin[3,4-b]indole-3-carboxylate Efficiently
Implementing this synthesis route requires strict adherence to the specified reaction conditions to ensure the reproducibility of the high optical purity outcomes. The process begins with the hydrolysis of the crude ester mixture to the free acid, followed by the critical resolution step where temperature control is paramount to induce selective crystallization. Detailed standardized operating procedures regarding solvent ratios, cooling rates, and stirring speeds are essential to prevent the co-precipitation of the unwanted isomer. For a comprehensive breakdown of the specific molar equivalents, reaction times, and workup procedures validated in the patent examples, please refer to the structured guide below which outlines the exact operational parameters for scaling this chemistry.
- Hydrolyze the racemic ester mixture under alkaline conditions to obtain the corresponding alpha-amino acid intermediate.
- Perform chiral resolution by reacting the amino acid with S-1-(2-tert-butylphenyl)ethylamine to form diastereomeric salts and crystallize the target isomer.
- Dissociate the crystals to recover the optically pure amino acid and the chiral resolving agent, then esterify to obtain the final high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the adoption of this resolution technology offers substantial advantages in terms of cost stability and supply security for high-value pharmaceutical intermediates. By eliminating the dependency on preparative HPLC or SFC (Supercritical Fluid Chromatography) for purification, manufacturers can significantly reduce the capital expenditure required for specialized equipment and the ongoing operational costs associated with high-purity solvents and chromatography media. The ability to recover and recycle the chiral resolving agent acts as a hedge against price volatility for specialty amines, ensuring that the bill of materials remains predictable even as production volumes increase. This predictability is crucial for long-term supply agreements where fixed pricing models are often required by downstream pharmaceutical clients.
- Cost Reduction in Manufacturing: The economic model of this process is strengthened by the high recovery rate of the chiral auxiliary, which minimizes the net consumption of expensive reagents per batch. Unlike catalytic asymmetric synthesis which may require precious metal catalysts that are difficult to remove to ppm levels, this resolution method relies on organic crystallization, avoiding the need for costly metal scavenging resins or additional purification steps to meet heavy metal specifications. The simplification of the downstream processing train reduces labor hours and utility consumption, translating into a more competitive cost of goods sold (COGS) for the final intermediate without compromising on quality standards.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness of the crystallization process, which is less susceptible to the variability often seen in biocatalytic or sensitive catalytic reactions. The starting materials, including the resolving agent and the racemic precursor, are derived from commercially available feedstocks, reducing the risk of single-source bottlenecks. Furthermore, the scalability of crystallization is well-understood in the chemical industry, allowing for seamless technology transfer from pilot plant to full commercial production scales, thereby ensuring consistent delivery schedules and reducing the lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The environmental profile of this manufacturing route aligns with modern green chemistry initiatives by reducing solvent intensity and waste generation compared to chromatographic alternatives. The ability to recycle solvents and reagents within the loop decreases the volume of hazardous waste requiring disposal, simplifying regulatory compliance and lowering environmental fees. This sustainability angle is increasingly important for multinational corporations aiming to meet carbon reduction targets, making suppliers who utilize such efficient resolution technologies preferred partners for green supply chain initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral resolution technology in an industrial setting. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on the feasibility and performance metrics of the process. Understanding these details helps stakeholders evaluate the suitability of this method for their specific project requirements and risk assessment protocols.
Q: What is the primary advantage of this chiral resolution method over chromatographic separation?
A: This method utilizes diastereomeric crystallization which significantly reduces solvent consumption and operational complexity compared to column chromatography, while allowing for the recovery and reuse of the expensive chiral resolving agent.
Q: What optical purity can be achieved using this specific resolution protocol?
A: Experimental data indicates that the process can achieve an HPLC purity of over 99% for the target enantiomer, effectively removing the less active or potentially harmful diastereomer.
Q: Is the chiral resolving agent reusable in this manufacturing process?
A: Yes, the protocol includes a dissociation step that allows for the recovery of the S-1-(2-tert-butylphenyl)ethylamine resolving agent with a recovery rate of approximately 95%, enabling multiple recycling cycles.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Aryl-1H-Pyridin[3,4-b]indole-3-carboxylate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires deep technical expertise and robust infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the intricate details of chiral resolution are preserved during scale-up. We maintain stringent purity specifications across all our product lines, supported by rigorous QC labs equipped with advanced chiral HPLC and polarimetry tools to verify optical purity at every stage of manufacturing. Our commitment to quality assurance means that every batch of intermediate delivered meets the exacting standards required for GMP pharmaceutical synthesis.
We invite you to collaborate with us to optimize your supply chain for these complex chiral building blocks. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how our efficient resolution processes can lower your overall project costs. We encourage potential partners to contact us to request specific COA data and route feasibility assessments, allowing you to validate our capabilities against your internal technical criteria before committing to production schedules.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
