Optimized Synthetic Route for High-Purity S-Ivabradine Intermediate via Chiral Resolution
Optimized Synthetic Route for High-Purity S-Ivabradine Intermediate via Chiral Resolution
The pharmaceutical landscape for cardiovascular treatments has been significantly advanced by the development of Ivabradine (S 16257-2), a selective inhibitor of the hyperpolarization-activated cyclic nucleotide-gated (If) ionic channel. As detailed in the comprehensive analysis of patent CN102249937A, the efficient production of its key chiral building block, 1-(S)-4,5-dimethoxy-1-methylaminomethyl-benzocyclobutane, remains a pivotal challenge for generic API manufacturers seeking market entry. This patent discloses a groundbreaking preparation method that circumvents the severe safety and efficiency bottlenecks associated with legacy synthetic pathways. By leveraging a streamlined sequence of hydrolysis, chiral resolution, amidation, and reduction, the disclosed technology offers a robust framework for producing high-purity intermediates essential for the synthesis of this vital anti-anginal medication.
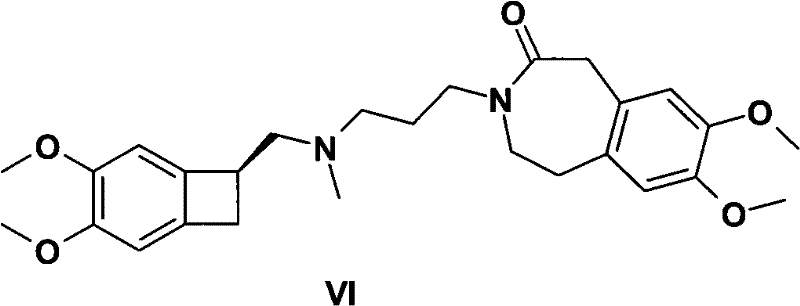
For R&D Directors and Process Chemists, understanding the structural complexity of the target molecule is paramount. The final drug substance, Ivabradine, relies heavily on the stereochemical integrity of the benzocyclobutane core. Any deviation in the chiral center during the intermediate stage can compromise the efficacy and safety profile of the final therapeutic agent. The patent provides a clear roadmap for navigating this complexity, ensuring that the (S)-configuration is established early and maintained throughout the synthesis. This level of control is not merely a regulatory requirement but a fundamental aspect of producing a reliable pharmaceutical intermediate supplier product that meets global quality standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methods, such as those described in patent EPO534859, have long plagued the industry with significant operational hazards and inefficiencies. The traditional route typically involves the reduction of a nitrile to a primary amine followed by a complex resolution step, and crucially, requires the use of vinyl chloroformate to synthesize carbamates. Vinyl chloroformate is a reagent known for its high toxicity and instability, posing severe risks to plant personnel and requiring expensive containment infrastructure. Furthermore, the conventional pathway necessitates two distinct reduction steps, one of which often employs borane, a reagent that is notoriously difficult to control on a large scale due to its pyrophoric nature and the generation of hazardous gaseous byproducts.
The cumulative effect of these drawbacks results in a process that is not only dangerous but also economically unviable for modern cost-sensitive markets. The aftertreatment procedures associated with borane reductions are complex, often involving quenching steps that generate large volumes of waste sludge. This complexity makes the traditional method difficult to realize industrialization, particularly in facilities aiming for green chemistry compliance. For procurement managers, the reliance on such specialized and hazardous reagents translates into volatile supply chains and inflated raw material costs, creating a barrier to sustainable manufacturing.
The Novel Approach
In stark contrast, the methodology presented in CN102249937A introduces a paradigm shift by eliminating the need for vinyl chloroformate and reducing the number of reduction steps to just one. The new route begins with the hydrolysis of 4,5-dimethoxy benzocyclobutyronitrile to a carboxylic acid, followed immediately by a chiral resolution step. This strategic inversion of the synthetic logic allows for the purification of the chiral center at the acid stage, which is chemically more stable and easier to handle than the amine intermediates used in older methods. The subsequent conversion to the target amine is achieved through a direct amidation-reduction sequence that is both operationally simple and highly efficient.
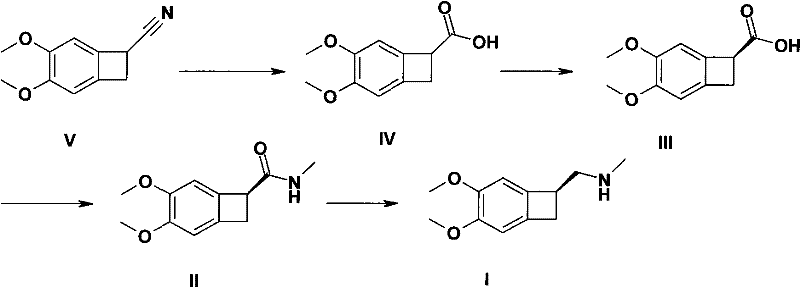
Visualizing the process flow reveals the elegance of this design. As illustrated in the reaction scheme, the pathway moves from the nitrile precursor (Compound V) to the racemic acid (Compound IV), then to the resolved (S)-acid (Compound III). From there, it proceeds to the amide (Compound II) and finally to the target amine (Compound I). This linear progression minimizes the number of unit operations and isolations required. By avoiding the formation of unstable carbamate intermediates, the process stability is markedly improved. For supply chain heads, this simplicity translates directly into reliability; fewer steps mean fewer points of failure, ensuring consistent batch-to-batch quality and shorter overall cycle times.
Mechanistic Insights into Chiral Resolution and Amidation-Reduction
The heart of this synthetic strategy lies in the chiral resolution of the carboxylic acid (Compound IV). The patent specifies the use of alkaline resolving agents such as cinchonidine, quinine, or (S)-(-)-alpha-phenylethylamine to form diastereomeric salts. The mechanism relies on the differential solubility of these salts in solvents like ethanol or methanol. By carefully controlling the temperature, typically refluxing to form the salt and then cooling slowly to 0°C to 25°C for crystallization, the desired (S)-enantiomer can be isolated with exceptional purity. Experimental data from the patent indicates that using cinchonidine or (S)-(-)-alpha-phenylethylamine can achieve enantiomeric excess (ee) values exceeding 99%, which is critical for downstream API synthesis.
Following resolution, the conversion of the chiral acid (Compound III) to the amide (Compound II) is a pivotal transformation. The patent offers flexibility here, allowing for activation via thionyl chloride to form an acyl chloride intermediate, or through the use of peptide coupling agents like EDCI and HOBt. The acyl chloride route is particularly advantageous for large-scale manufacturing due to the low cost of thionyl chloride and the ease of removing excess reagent by evaporation. The subsequent reduction of the amide carbonyl to a methylene group is achieved using powerful reducing agents like lithium aluminum hydride (LiAlH4) or the safer sodium borohydride-trifluoroacetic acid system. This single reduction step replaces the multiple reductions of the prior art, significantly streamlining the process while maintaining high yields, reported between 65% and 95% depending on the specific reductant chosen.
How to Synthesize 1-(S)-4,5-dimethoxy-1-methylaminomethyl-benzocyclobutane Efficiently
The execution of this synthesis requires precise adherence to the reaction conditions outlined in the patent to ensure optimal yield and purity. The process begins with the base-catalyzed hydrolysis of the nitrile, followed by the critical resolution step where temperature control dictates the optical purity. Once the chiral acid is secured, the amidation can be performed under mild conditions, and the final reduction completes the sequence. For technical teams looking to implement this, the detailed standardized synthesis steps are provided in the guide below, which breaks down the specific molar ratios, solvent choices, and workup procedures necessary for success.
- Hydrolyze 4,5-dimethoxy benzocyclobutyronitrile (Compound V) using alcoholic potassium hydroxide to form the racemic carboxylic acid (Compound IV).
- Perform chiral resolution on Compound IV using alkaline resolving agents like cinchonidine or (S)-(-)-alpha-phenylethylamine to isolate the (S)-enantiomer (Compound III).
- Condense the resolved acid (Compound III) with methylamine using thionyl chloride or coupling agents like EDCI to form the amide (Compound II).
- Reduce the amide carbonyl of Compound II to a methylene group using lithium aluminum hydride or sodium borohydride/trifluoroacetic acid to yield the final amine (Compound I).
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this novel synthetic route offers profound advantages for organizations focused on cost reduction in pharmaceutical intermediate manufacturing. The elimination of toxic and expensive reagents like vinyl chloroformate and borane removes a significant portion of the raw material cost burden. Furthermore, the simplification of the workflow reduces the demand for specialized equipment capable of handling pyrophoric materials, thereby lowering capital expenditure requirements for production facilities. This economic efficiency is compounded by the reduction in waste treatment costs, as the process generates fewer hazardous byproducts compared to the conventional nitrile reduction pathways.
- Cost Reduction in Manufacturing: The streamlined nature of this process directly impacts the bottom line by minimizing reagent consumption and energy usage. By consolidating the synthesis into fewer steps, manufacturers can reduce labor hours and utility costs associated with heating, cooling, and stirring over extended periods. The ability to use common, commodity chemicals like thionyl chloride and sodium borohydride instead of specialized, high-cost reagents ensures that the variable cost of goods sold remains competitive even in fluctuating market conditions. This structural cost advantage provides a buffer against raw material price volatility, securing margins for the long term.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the reliance on niche reagents that may face shortages. This method utilizes widely available starting materials and resolving agents, mitigating the risk of supply disruptions. The robustness of the chiral resolution step ensures that even if minor variations occur in upstream feedstock quality, the purification capability of the crystallization process can correct these deviations, guaranteeing a consistent output of high-purity intermediate. This reliability is essential for maintaining the production schedules of downstream API manufacturers who depend on just-in-time delivery models.
- Scalability and Environmental Compliance: Scaling chemical processes often exposes hidden inefficiencies, but this route is designed with scalability in mind. The unit operations involved—hydrolysis, crystallization, filtration, and standard reduction—are well-understood and easily transferable from pilot plant to commercial scale. Moreover, the avoidance of heavy metals and highly toxic gases aligns with increasingly stringent environmental regulations globally. Facilities adopting this method can demonstrate a commitment to sustainability, reducing their environmental footprint and simplifying the permitting process for expansion, which is a key consideration for long-term strategic planning.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity on the feasibility and benefits of the process for potential partners and licensees.
Q: Why is the new synthesis route for Ivabradine intermediates considered safer than previous methods?
A: The novel route eliminates the use of highly toxic vinyl chloroformate and difficult-to-handle borane reagents required in conventional methods, significantly improving operational safety and reducing hazardous waste.
Q: What represents the critical step for ensuring optical purity in this process?
A: The chiral resolution of the racemic carboxylic acid (Compound IV) using specific alkaline resolving agents like cinchonidine is the critical step, achieving enantiomeric excess (ee) values greater than 99%.
Q: How does this method impact production costs for cardiovascular API manufacturers?
A: By reducing the number of reduction steps from two to one and utilizing cheaper, more accessible reagents like thionyl chloride instead of specialized coupling agents, the method drastically lowers raw material and processing costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-(S)-4,5-dimethoxy-1-methylaminomethyl-benzocyclobutane Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global supply of cardiovascular medications. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We are committed to delivering products that meet stringent purity specifications, utilizing our rigorous QC labs to verify every batch against the highest international standards. Our infrastructure is designed to handle complex chiral resolutions and sensitive reduction reactions safely and efficiently.
We invite pharmaceutical partners to collaborate with us to leverage this advanced synthetic technology for their Ivabradine supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, ensuring that your project moves forward with the confidence of a secure and optimized supply partner.
