Advanced Manufacturing of Chiral 1,4-Dihydro-4-(3-nitrophenyl)-3,5-pyridinedicarboxylates for Calcium Antagonists
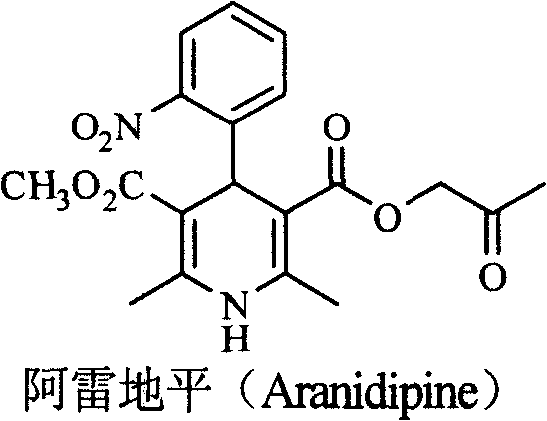
The pharmaceutical industry's relentless pursuit of potent cardiovascular therapeutics has placed chiral 1,4-dihydropyridine calcium antagonists at the forefront of modern medicinal chemistry. Patent CN101357901B discloses a groundbreaking class of chiral 1,4-dihydro-4-(3-nitrophenyl)-3,5-pyridinedicarboxylates, which serve as critical intermediates for next-generation antihypertensive agents. Unlike traditional racemic mixtures, these optically active compounds offer superior pharmacokinetic profiles, specifically designed to delay metabolism through the strategic introduction of bulky groups in the ester side chain. This technological breakthrough addresses the stringent regulatory requirements set by agencies like the FDA, which mandate the quantification of enantiomeric efficacy and toxicity. By providing a robust synthetic route to these high-value intermediates, the patent enables the production of drugs with enhanced bioavailability and prolonged duration of action, representing a significant leap forward in the treatment of hypertension and related vascular disorders.
Historically, the synthesis of optically active 1,4-dihydropyridines has been plagued by significant technical and economic hurdles that hindered widespread commercial adoption. Conventional methods often relied on enzymatic resolution, which suffers from poor stability under the strong acidic or basic conditions frequently required for downstream processing, leading to inconsistent batch quality and difficult separation from substrates. Alternatively, asymmetric synthesis using chiral auxiliary agents was employed, but this approach incurred prohibitive costs due to the high price of the auxiliaries and the large stoichiometric quantities needed, rendering the final API economically unviable for mass markets. Furthermore, direct crystallization of salts was often impossible because the nitrogen at the 1-position of the dihydropyridine ring is weakly basic and fails to form stable salts with resolving agents, leaving manufacturers with few viable options for obtaining high-purity single enantiomers efficiently.
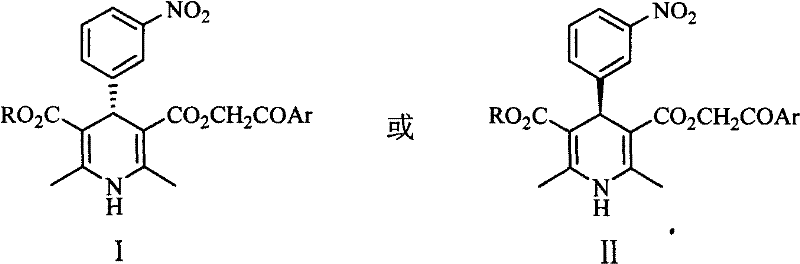
The novel approach detailed in the patent data overcomes these legacy limitations through an ingenious chemical resolution strategy followed by selective esterification. Instead of struggling with the weakly basic nitrogen, the process targets the carboxylic acid functionality on the side chain, utilizing naturally occurring cinchona alkaloids such as cinchonidine or cinchonine as resolving agents. This method leverages the formation of diastereomeric salts that exhibit distinct solubility differences, allowing for the isolation of optically pure 1,4-dihydro-4-(3-nitrophenyl)-3,5-pyridine monocarboxylic acids through simple crystallization. Once the chiral center is established, the intermediate undergoes a mild esterification with alpha-halo aryl ethyl ketones in aprotic polar solvents. This two-step sequence not only bypasses the need for expensive catalysts but also ensures that the stereochemical integrity is maintained throughout the synthesis, delivering the biologically active S-enantiomer with high fidelity.
Mechanistic Insights into Cinchonidine-Mediated Resolution and Esterification
The core of this synthetic innovation lies in the stereoselective recognition between the chiral resolving agent and the racemic substrate. When cinchonidine is introduced to the solution of racemic 1,4-dihydro-4-(3-nitrophenyl)-3,5-pyridine monocarboxylic acid, it selectively complexes with one enantiomer to form a less soluble diastereomeric salt. This precipitation drives the equilibrium, effectively pulling the desired enantiomer out of the solution while leaving the unwanted isomer in the mother liquor. The absolute configuration of the resulting products is strictly defined, with Formula I corresponding to the S-configuration and Formula II to the R-configuration, as confirmed by X-ray crystallography and optical rotation data. The subsequent esterification step involves a nucleophilic substitution where the carboxylate anion attacks the alpha-carbon of the halo-ketone. This reaction is facilitated by bases like potassium carbonate or triethylamine, which deprotonate the acid without racemizing the sensitive chiral center at the 4-position of the dihydropyridine ring.
Controlling impurities in this process is achieved through the rigorous selection of solvents and reaction temperatures. The patent specifies a wide operational window, from -20°C to 150°C, allowing manufacturers to optimize conditions for maximum yield and purity. For instance, using mixed solvent systems like ethanol-water or acetone-water during the resolution step enhances the selectivity of crystallization, minimizing the co-precipitation of the undesired enantiomer. In the esterification phase, the use of anhydrous aprotic solvents such as N,N-dimethylformamide (DMF) or tetrahydrofuran (THF) prevents hydrolysis of the reactive alpha-halo ketone and ensures complete conversion. The final workup involves standard aqueous extraction and drying, which effectively removes inorganic salts and residual bases, resulting in a product that meets stringent pharmaceutical purity specifications without the need for complex chromatographic purification on a commercial scale.
How to Synthesize Chiral 1,4-Dihydro-4-(3-nitrophenyl)-3,5-pyridinedicarboxylates Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for producing these high-value intermediates with consistent quality. The process begins with the preparation of the racemic monocarboxylic acid, which is then subjected to optical resolution using cinchonidine in a suitable solvent system. Following the isolation of the optically active acid, it is reacted with various alpha-halo aryl ethyl ketones to introduce the diverse side chains necessary for tuning pharmacological properties. The detailed standardized synthesis steps below describe the precise reagents, stoichiometry, and conditions required to replicate the high yields and optical purities reported in the experimental examples, ensuring a smooth transition from bench-scale research to pilot plant operations.
- Resolve racemic 1,4-dihydro-4-(3-nitrophenyl)-3,5-pyridine monocarboxylic acid using cinchonidine or cinchonine in solvents like DMF or ethanol-water mixtures to obtain optically active intermediates.
- React the optically active monocarboxylic acid with alpha-halo aryl ethyl ketones in the presence of a base such as potassium carbonate or triethylamine in aprotic polar solvents.
- Work up the reaction mixture by dilution with water, extraction with ethyl acetate, drying, and purification via crystallization or column chromatography to isolate the final chiral diester.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this resolution-based manufacturing route offers transformative benefits in terms of cost structure and supply reliability. By eliminating the dependency on fragile enzymes or costly chiral ligands, the process drastically simplifies the raw material portfolio, relying instead on commodity chemicals and abundant natural alkaloids. This shift reduces exposure to volatile pricing associated with specialized catalytic reagents and minimizes the risk of supply disruptions caused by the limited availability of biocatalysts. Furthermore, the robustness of the crystallization steps ensures high throughput and consistent batch-to-batch quality, which is critical for maintaining uninterrupted production schedules in a regulated pharmaceutical environment.
- Cost Reduction in Manufacturing: The economic model of this process is fundamentally superior to asymmetric synthesis because it avoids the purchase of expensive chiral auxiliaries that are often consumed in stoichiometric amounts. Additionally, the ability to recover and recycle the cinchonidine resolving agent from the mother liquor further drives down the net cost per kilogram. The elimination of transition metal catalysts also removes the need for expensive and time-consuming heavy metal scavenging steps, streamlining the downstream processing and reducing waste disposal costs significantly.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, including 3-nitrobenzaldehyde derivatives and cinchona alkaloids, are globally sourced commodities with well-established supply chains. This diversity of supply sources mitigates the risk of single-vendor dependency, ensuring that production can continue even if one supplier faces logistical challenges. The chemical stability of the intermediates allows for safer storage and transportation, reducing the complexity of logistics and enabling the strategic stocking of key precursors to buffer against market fluctuations.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, the process utilizes standard unit operations that are easily transferable to large-scale reactors. The solvents used, such as ethanol and ethyl acetate, are greener alternatives to chlorinated solvents and are easier to recover and recycle, aligning with modern green chemistry principles. The high atom economy of the esterification step and the efficient recovery of resolving agents minimize the generation of hazardous waste, simplifying regulatory compliance and reducing the environmental footprint of the manufacturing facility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these chiral intermediates. The answers are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity on the feasibility and advantages of this manufacturing route for potential partners and licensees seeking to integrate this technology into their pipelines.
Q: What is the primary advantage of the chemical resolution method described in CN101357901B?
A: The primary advantage is the avoidance of expensive chiral auxiliaries or unstable enzymes. By utilizing readily available cinchona alkaloids like cinchonidine, the process achieves high optical purity through simple crystallization, significantly lowering production costs and improving scalability compared to asymmetric synthesis.
Q: Why is the S-enantiomer preferred for calcium antagonist activity?
A: Structure-activity relationship studies indicate that the S-type enantiomer of 1,4-dihydropyridine compounds typically exhibits stronger pharmacological activity. Specifically, for derivatives like Aranidipine, the S-enantiomer demonstrates superior vasodilation rates and receptor binding affinity compared to the R-enantiomer or the racemate.
Q: Can this process be scaled for commercial API production?
A: Yes, the process is highly scalable. It relies on standard unit operations such as dissolution, crystallization, filtration, and conventional esterification. The use of common industrial solvents like DMF, ethanol, and ethyl acetate, along with stable inorganic bases, ensures that the technology can be transferred from laboratory to multi-ton commercial production without significant engineering hurdles.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral 1,4-Dihydropyridine Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity chiral intermediates play in the development of life-saving cardiovascular medications. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated resolution and esterification techniques described in CN101357901B can be implemented with precision and efficiency. We operate stringent purity specifications and maintain rigorous QC labs equipped with advanced chiral HPLC and NMR capabilities to guarantee that every batch of 1,4-dihydro-4-(3-nitrophenyl)-3,5-pyridinedicarboxylates meets the exacting standards required for API synthesis.
We invite global pharmaceutical partners to collaborate with us to leverage this advanced technology for your next-generation calcium antagonist projects. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how our optimized process can reduce your overall cost of goods. Please contact us today to request specific COA data for our available intermediates and to discuss route feasibility assessments that will accelerate your path to clinical trials and commercial launch.
