Optimizing Anti-Osteoporosis Drug Production: A Technical Analysis of Novel CD Ring Fragment Synthesis
Optimizing Anti-Osteoporosis Drug Production: A Technical Analysis of Novel CD Ring Fragment Synthesis
The pharmaceutical industry's relentless pursuit of more efficient pathways for active pharmaceutical ingredients (APIs) often hinges on the optimization of complex intermediate synthesis. Patent CN1272494A presents a significant technological breakthrough in the preparation of novel triethylsilyl ether analogues of the CD ring fragment, a critical structural motif in the synthesis of potent anti-osteoporosis agents such as Vitamin D3 analogues. This patent discloses a robust methodology that not only enhances stereochemical control but also streamlines the production of these high-value intermediates. For R&D directors and process chemists, the introduction of the triethylsilyl (TES) protecting group represents a strategic evolution from traditional methods, offering improved stability and selectivity during key transformation steps. The detailed reaction schemes provided in the patent illustrate a sophisticated approach to constructing the sterically demanding side chain while maintaining the integrity of the sensitive epoxide and enone functionalities inherent to the steroid backbone.
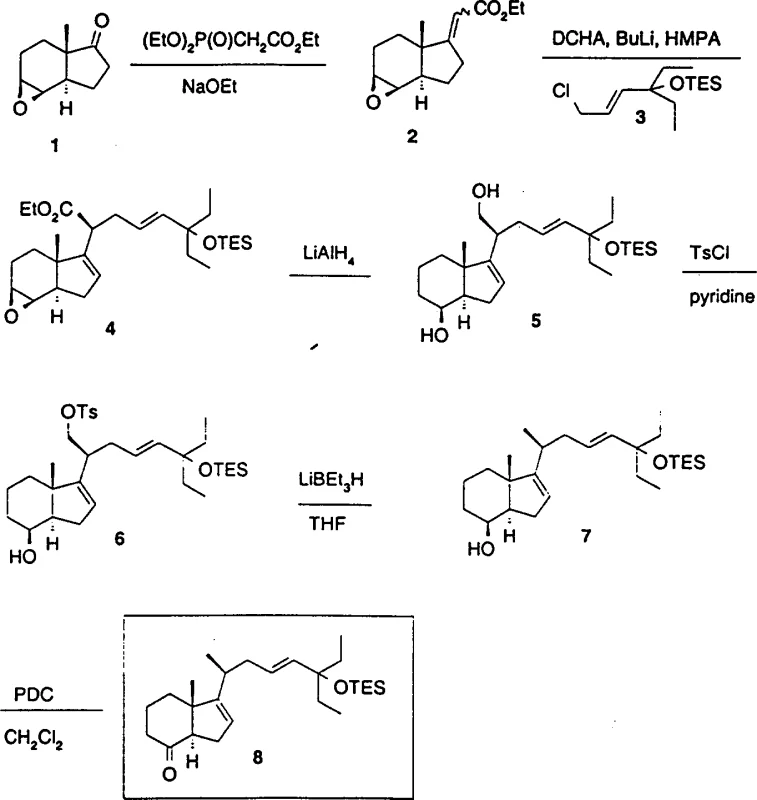
As we delve deeper into the technical specifics, it becomes evident that this process addresses several long-standing challenges in steroid chemistry. The ability to introduce the C-20 chiral center with high fidelity is paramount for the biological activity of the final drug substance. By leveraging specific reagents like lithium dicyclohexylamide (LCA) and hexamethylphosphoramide (HMPA), the inventors have established a protocol that minimizes the formation of unwanted diastereomers. This level of precision is crucial for reducing downstream purification burdens, a factor that directly impacts the cost of goods sold (COGS) and the environmental footprint of the manufacturing process. Furthermore, the scalability of this route is supported by the use of reagents that are accessible on a commercial scale, making it an attractive option for industrial adoption.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Vitamin D3 analogues has relied heavily on the Lythgoe phosphine oxide method, a technique that, while effective, presents notable drawbacks in a modern manufacturing context. The conventional approach typically involves the coupling of a CD-ring fragment with an A-ring fragment, followed by a deprotection sequence that can be fraught with complications. One of the primary limitations is the sensitivity of the intermediates to the reaction conditions required for coupling, often leading to decomposition or the formation of difficult-to-separate impurities. Additionally, the management of phosphine oxide byproducts adds a layer of complexity to waste treatment and purification protocols. In many legacy processes, the control of stereochemistry at the C-20 position is less than optimal, resulting in mixtures of epimers that require extensive chromatographic separation, thereby reducing overall yield and increasing solvent consumption. These inefficiencies translate directly into higher production costs and longer lead times, which are critical pain points for procurement managers seeking reliable supply chains.
The Novel Approach
In contrast, the novel approach detailed in CN1272494A introduces a streamlined pathway centered around the use of triethylsilyl (TES) protected intermediates. This strategy fundamentally alters the reactivity profile of the side chain precursor, allowing for milder and more selective transformations. The new method initiates with a Wittig-Horner reaction that preserves the epoxide ring, a delicate functionality that is often compromised in harsher conditions. Subsequently, the stereoselective alkylation step utilizes a specifically tuned base system to install the side chain with remarkable precision. The use of the TES group provides a balance of steric bulk and electronic properties that protects the alcohol functionality during the alkylation and reduction steps, yet allows for facile removal when necessary. This results in a cleaner reaction profile with fewer side products, simplifying the isolation of the desired (20S)-epimer. The overall sequence demonstrates a marked improvement in atom economy and operational simplicity, positioning it as a superior alternative for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Stereoselective Alkylation and Reduction
The core of this synthetic innovation lies in the meticulous control of stereochemistry during the alkylation of the alpha,beta-unsaturated ester. The mechanism begins with the deprotonation of the ester at the gamma-position using lithium dicyclohexylamide (LCA) in the presence of HMPA. HMPA acts as a powerful Lewis base coordinator, breaking up lithium aggregates and generating a highly reactive, monomeric enolate species. This activation is critical for achieving the necessary kinetic control at low temperatures, specifically between -60°C and -65°C. Under these cryogenic conditions, the enolate attacks the allylic chloride side chain fragment in an SN2'-like fashion, or potentially via a concerted mechanism that favors the formation of the (20S) configuration. The patent data indicates that this specific combination of base and solvent additives suppresses the formation of the thermodynamic (20R) product, yielding the desired isomer in a ratio that allows for efficient chromatographic separation. This mechanistic nuance is vital for R&D teams aiming to replicate the process, as slight deviations in temperature or base stoichiometry could drastically alter the diastereomeric ratio.
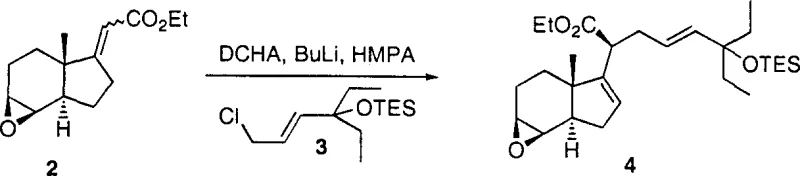
Following the establishment of the carbon skeleton, the process employs a dual-reduction strategy to set the remaining functional groups. The simultaneous reduction of the ester and the epoxide functionalities using lithium aluminum hydride (LiAlH4) is a bold move that showcases the robustness of the TES protecting group. Typically, epoxides are susceptible to nucleophilic attack, but in this specific molecular environment, the reduction proceeds selectively to open the epoxide and reduce the ester to a primary alcohol without compromising the silyl ether. Subsequent steps involve the conversion of the primary alcohol to a tosylate, followed by a selective reduction using Super-Hydride (LiBEt3H). This sequence effectively replaces the hydroxymethyl group with a methyl group, completing the construction of the side chain's terminal architecture. The final oxidation step using pyridinium dichromate (PDC) cleanly converts the secondary alcohol to the corresponding ketone, delivering the target CD ring fragment with the correct oxidation state for subsequent coupling reactions. Each step is designed to minimize impurity generation, ensuring a high-purity output suitable for GMP manufacturing.
How to Synthesize Triethylsilyl Ether Analogues Efficiently
The execution of this synthesis requires strict adherence to the specified reaction conditions to ensure reproducibility and high yield. The process begins with the preparation of the alpha,beta-unsaturated ester via a Wittig-Horner reaction, where temperature control is essential to prevent epoxide degradation. Following this, the critical alkylation step demands precise maintenance of cryogenic temperatures to secure the desired stereochemistry. The subsequent reduction and functional group interconversions utilize standard reagents but require careful workup procedures to remove metal salts and byproducts. For a detailed breakdown of the specific operational parameters, reagent quantities, and purification techniques, please refer to the standardized synthesis guide below.
- Perform Wittig-Horner reaction on the starting epoxide ketone using triethyl phosphonoacetate and sodium ethoxide to form the alpha,beta-unsaturated ester.
- Execute stereoselective alkylation using lithium dicyclohexylamide (LCA) and HMPA at low temperatures (-60 to -65°C) to introduce the side chain with high 20S selectivity.
- Conduct simultaneous reduction of ester and epoxide functionalities followed by selective tosylation and hydride reduction to establish the final C-21 methyl group and ketone functionality.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this novel synthesis route offers substantial benefits for procurement and supply chain management within the pharmaceutical sector. The primary advantage stems from the enhanced reliability of the raw material supply. The side chain fragment is derived from 3-ethyl-1-pentyn-3-ol, a commodity chemical that is readily available from multiple global suppliers. This diversification of source material mitigates the risk of supply disruptions that often plague specialized, custom-synthesized intermediates. Furthermore, the robustness of the TES protecting group reduces the sensitivity of the intermediates to storage and handling conditions, lowering the logistical costs associated with cold chain transport or inert atmosphere storage. The simplified purification profile, resulting from the high stereoselectivity of the alkylation step, translates into reduced solvent usage and shorter cycle times. This efficiency gain allows for a more responsive manufacturing schedule, enabling suppliers to meet fluctuating market demands with greater agility. Ultimately, the process design prioritizes operational stability, which is a key determinant in securing long-term supply contracts for critical API intermediates.
- Cost Reduction in Manufacturing: The elimination of complex phosphine oxide coupling steps and the reduction in chromatographic purification requirements lead to a significant decrease in processing costs. By minimizing the formation of difficult-to-separate diastereomers, the process reduces the volume of solvents and silica gel needed for purification, directly impacting the variable cost of production. Additionally, the use of commercially available starting materials avoids the premium pricing associated with bespoke reagents. The overall yield improvement, driven by better stereocontrol, means that less raw material is wasted, further enhancing the economic viability of the route. These factors combine to create a leaner manufacturing process that offers a competitive edge in price-sensitive markets.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals for the side chain synthesis ensures a stable and continuous supply of key inputs. Unlike proprietary reagents that may be sourced from a single vendor, the precursors for this route are produced by major chemical manufacturers worldwide, providing a buffer against regional supply shocks. The robustness of the intermediates also simplifies inventory management, as the materials are less prone to degradation over time. This stability allows for the strategic stocking of intermediates, smoothing out production peaks and valleys. For supply chain heads, this translates to a lower risk of stockouts and a more predictable delivery schedule for the final API, fostering stronger relationships with downstream pharmaceutical partners.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing reaction conditions and reagents that are compatible with large-scale batch reactors. The avoidance of exotic catalysts and the use of standard workup procedures facilitate technology transfer from pilot plant to commercial production. From an environmental standpoint, the reduction in solvent consumption and waste generation aligns with green chemistry principles. The simplified purification steps reduce the load on waste treatment facilities, lowering the environmental compliance burden. This sustainability profile is increasingly important for pharmaceutical companies aiming to meet corporate social responsibility goals and regulatory standards regarding waste disposal and carbon footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. They are derived from the specific challenges and solutions outlined in the patent documentation, providing clarity on the practical aspects of the technology. Understanding these nuances is essential for stakeholders evaluating the feasibility of integrating this process into their existing manufacturing portfolios. The answers reflect the empirical data and expert analysis of the reaction mechanisms described in CN1272494A.
Q: What is the primary advantage of the triethylsilyl (TES) protection strategy in this synthesis?
A: The TES protection group offers superior stability during the harsh alkylation conditions compared to other silyl groups, while remaining easily removable in later stages, ensuring high purity of the final CD ring fragment.
Q: How is stereochemical control achieved at the C-20 position?
A: Stereocontrol is achieved through the use of lithium dicyclohexylamide (LCA) in the presence of hexamethylphosphoramide (HMPA) at cryogenic temperatures (-60 to -65°C), which favors the formation of the desired (20S)-epimer over the (20R)-isomer.
Q: Are the starting materials for the side chain commercially available?
A: Yes, the synthesis utilizes commercially available 3-ethyl-1-pentyn-3-ol as the starting material for the side chain fragment, which significantly simplifies the supply chain and reduces raw material procurement risks.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Vitamin D3 Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving medications. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, backed by our rigorous QC labs and state-of-the-art analytical capabilities. Our facility is equipped to handle the specific requirements of steroid chemistry, including the precise temperature control and inert atmosphere conditions necessary for the successful execution of the triethylsilyl ether synthesis route described in this report.
We invite you to collaborate with us to optimize your supply chain for anti-osteoporosis drugs. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. By partnering with NINGBO INNO PHARMCHEM, you gain access to a reliable supply of high-purity pharmaceutical intermediates, empowering your R&D and production teams to bring innovative therapies to market faster and more cost-effectively.
