Advanced Electrochemical Synthesis of Fluoroalkyl Hydrazones for Scalable Pharmaceutical Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking greener, more efficient pathways to access fluorinated building blocks, which are critical for enhancing the metabolic stability and lipophilicity of drug candidates. Patent CN114574880A introduces a groundbreaking electrochemical preparation method for fluoroalkyl-substituted hydrazones, representing a significant paradigm shift from traditional chemical oxidation to sustainable electrosynthesis. This technology utilizes commercially available sodium fluoroalkyl sulfinates as the fluoroalkyl source, operating under mild, neutral conditions without the need for toxic metal catalysts or stoichiometric chemical oxidants. By leveraging electrical energy to drive the radical generation, this process not only simplifies the operational workflow but also aligns with the stringent environmental regulations facing modern pharmaceutical intermediate manufacturing. The versatility of this approach allows for the synthesis of diverse fluoroalkyl hydrazones, which serve as pivotal precursors to fluoroalkyl ketones, thereby offering a robust solution for the production of high-value enzyme inhibitors and therapeutic agents.
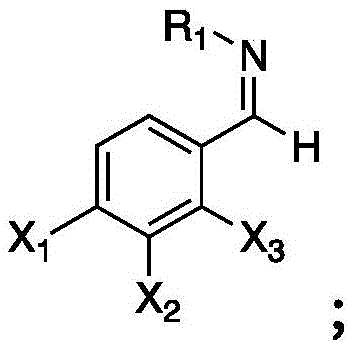
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trifluoromethyl hydrazones has relied heavily on traditional organic transformations that are fraught with significant technical and economic drawbacks. Conventional routes often necessitate the use of expensive and hazardous trifluoromethylation reagents, such as Togni reagents or Umemoto reagents, which drastically inflate the raw material costs for cost reduction in pharmaceutical intermediate manufacturing. Furthermore, these methods frequently require transition metal catalysts like copper or palladium, introducing the risk of heavy metal contamination that demands rigorous and costly purification steps to meet regulatory standards for high-purity pharmaceutical intermediates. The reliance on strong chemical oxidants or reductants also generates substantial amounts of toxic waste, complicating disposal and increasing the environmental footprint of the production facility. Additionally, many traditional protocols suffer from harsh reaction conditions, including extreme temperatures or pressures, which pose safety risks and limit the feasibility of commercial scale-up of complex fluorinated intermediates.
The Novel Approach
In stark contrast, the electrochemical method disclosed in the patent offers a streamlined, catalyst-free alternative that directly addresses these pain points. By employing a simple undivided electrolytic cell equipped with a graphite anode and a platinum cathode, the system generates reactive fluoroalkyl radicals in situ from stable sodium sulfinate salts. This eliminates the need for external oxidants, as the anode potential drives the single-electron oxidation of the sulfinate anion. The reaction proceeds efficiently in a mixed solvent system of acetonitrile and water, maintaining neutral pH conditions that preserve sensitive functional groups on the hydrazone substrate. As illustrated in the reaction scheme below, the process is remarkably operationally simple, requiring only the application of a constant current to achieve high conversion rates.
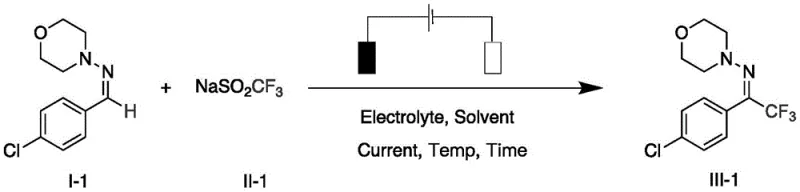
This novel approach not only enhances safety by removing hazardous reagents but also improves atom economy, making it an ideal candidate for reducing lead time for high-purity fluoroalkyl hydrazones. The ability to tune the reaction rate simply by adjusting the current intensity provides process engineers with a powerful lever for optimizing throughput without altering the chemical formulation.
Mechanistic Insights into Electrochemical Fluoroalkylation
The core of this innovation lies in the anodic oxidation mechanism, which facilitates the generation of fluoroalkyl radicals under mild conditions. Upon applying a constant current, the sodium fluoroalkyl sulfinate (RfSO2Na) undergoes single-electron oxidation at the graphite anode surface to form a sulfonyl radical intermediate. This unstable species rapidly extrudes sulfur dioxide (SO2) to yield the highly reactive fluoroalkyl radical (Rf•). This radical then adds selectively to the electron-deficient carbon-nitrogen double bond of the hydrazone substrate, forming a new carbon-carbon bond and a nitrogen-centered radical. Subsequent oxidation and deprotonation steps, mediated by the electrode potential and the electrolyte environment, restore the C=N double bond to yield the final fluoroalkyl-substituted hydrazone product. The cathode simultaneously reduces protons from the solvent or substrate to generate hydrogen gas, completing the electrical circuit without producing harmful byproducts. This elegant redox-neutral cycle ensures that the only reagents consumed are the starting materials and electricity, minimizing waste generation.
Impurity control is inherently superior in this electrochemical system due to the absence of metal catalysts that often promote side reactions such as homocoupling or over-oxidation. The use of specific electrolytes, particularly lithium perchlorate (LiClO4), plays a crucial role in stabilizing the radical intermediates and facilitating efficient charge transfer across the electrode interface. Experimental optimization revealed that halide-based electrolytes could lead to competitive oxidation of the halide ions, reducing the Faradaic efficiency for the desired transformation. By selecting non-nucleophilic anions like perchlorate or hexafluorophosphate, the system ensures that the anodic potential is focused exclusively on the sulfinate substrate. Furthermore, the addition of a small amount of water to the acetonitrile solvent was found to prevent the accumulation of black solid deposits on the platinum cathode, which otherwise would hinder electron transfer and reduce reactor longevity. This careful balance of solvent composition and electrolyte selection ensures consistent product quality and high purity profiles essential for downstream pharmaceutical applications.
How to Synthesize Fluoroalkyl Hydrazones Efficiently
To implement this electrochemical protocol effectively, precise control over reaction parameters is essential to maximize yield and reproducibility. The process begins with the preparation of a homogeneous reaction mixture containing the hydrazone substrate, sodium fluoroalkyl sulfinate, and the optimized electrolyte in an acetonitrile-water solvent system. The choice of electrode materials is critical; a graphite rod serves as the sacrificial or inert anode depending on the specific variant, while a platinum sheet acts as the robust cathode. Once the cell is assembled, a constant direct current is applied while maintaining the temperature at approximately 80°C to ensure optimal solubility and reaction kinetics. Detailed standard operating procedures regarding molar ratios, current density, and workup protocols are critical for successful replication.
- Prepare the electrolytic cell by adding solvent (acetonitrile/water), electrolyte (LiClO4), sodium fluoroalkyl sulfinate, and the hydrazone substrate. Stir until homogeneous.
- Insert a graphite rod anode and platinum sheet cathode. Apply constant current (e.g., 12mA) at controlled temperature (e.g., 80°C) for the specified reaction time.
- Upon completion, extract the mixture with ethyl acetate, wash with brine, dry over sodium sulfate, and purify the crude product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this electrochemical technology translates into tangible strategic benefits beyond mere technical novelty. The elimination of precious metal catalysts removes a major cost driver and supply chain bottleneck, as the volatility of metal prices and the complexity of sourcing high-purity catalysts are completely bypassed. Moreover, the use of commodity chemicals like sodium trifluoromethanesulfinate ensures a stable and reliable supply base, mitigating the risk of raw material shortages that often plague specialized synthetic routes. The simplified workup procedure, which avoids complex metal scavenging steps, significantly reduces processing time and solvent consumption, leading to substantial operational cost savings. This streamlined process flow enhances overall equipment effectiveness (OEE) by reducing batch cycle times and minimizing downtime for cleaning and maintenance.
- Cost Reduction in Manufacturing: The most significant economic advantage stems from the complete removal of expensive transition metal catalysts and stoichiometric oxidants. In traditional synthesis, the cost of palladium or copper catalysts, combined with the ligands required to stabilize them, can account for a large portion of the COGS (Cost of Goods Sold). By replacing these with electricity and inexpensive graphite electrodes, the variable cost per kilogram of product is drastically reduced. Additionally, the avoidance of metal residues means that the costly and time-consuming purification steps typically required to meet ppm-level metal specifications are no longer necessary. This reduction in downstream processing directly improves the gross margin of the final API intermediate, making the end product more competitive in the global market.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available, commercial-grade starting materials. Sodium fluoroalkyl sulfinates are produced on a multi-ton scale by multiple chemical suppliers globally, ensuring that there is no single point of failure in the raw material supply. Unlike specialized photoredox catalysts or air-sensitive reagents that require cold chain logistics and strict storage conditions, the reagents for this electrochemical process are stable solids that can be stored at ambient conditions. This stability simplifies inventory management and reduces the risk of material degradation during transport. Furthermore, the modular nature of electrochemical reactors allows for flexible capacity expansion; production volume can be scaled by adding more cells or increasing current density without the need for massive new infrastructure investments.
- Scalability and Environmental Compliance: From an EHS (Environment, Health, and Safety) perspective, this method offers a compelling value proposition. The reaction generates hydrogen gas as the only byproduct at the cathode, which can be safely vented or captured, eliminating the generation of heavy metal waste streams that require specialized hazardous waste disposal. The neutral reaction conditions reduce the risk of thermal runaway or corrosive accidents, enhancing plant safety. As regulatory bodies worldwide tighten restrictions on solvent emissions and heavy metal discharge, adopting a metal-free, electrochemical process future-proofs the manufacturing site against compliance risks. The ability to run the reaction in a continuous flow electrochemical cell further enhances scalability, allowing for the safe production of large quantities of energetic intermediates with a minimal physical footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this electrochemical synthesis route. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the practical aspects of adopting this technology for industrial production.
Q: What are the primary advantages of this electrochemical method over traditional trifluoromethylation?
A: This method eliminates the need for expensive transition metal catalysts and harsh chemical oxidants. It operates under neutral conditions using electricity as the clean reagent, significantly reducing toxic waste and simplifying the purification process for high-purity intermediates.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent highlights strong prospects for industrial scale-up. The use of commercial sodium salts and the ability to accelerate reaction rates by adjusting current intensity allows for flexible throughput management without compromising safety or yield.
Q: How does the choice of electrolyte impact the reaction yield?
A: Experimental data indicates that lithium perchlorate (LiClO4) provides superior yields compared to halide-based electrolytes. Halide anions, particularly iodide, can interfere with the reaction pathway, whereas perchlorate and hexafluorophosphate anions support efficient electron transfer.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fluoroalkyl Hydrazone Supplier
The electrochemical synthesis of fluoroalkyl hydrazones represents a cutting-edge advancement in green chemistry, offering a pathway to high-value intermediates with superior purity and sustainability profiles. At NINGBO INNO PHARMCHEM, we pride ourselves on staying at the forefront of such technological innovations, translating academic and patent breakthroughs into robust, commercial-scale manufacturing processes. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to pilot plant and finally to full-scale manufacturing is seamless and efficient. We understand that consistency is key in the pharmaceutical supply chain; therefore, our stringent purity specifications and rigorous QC labs guarantee that every batch of fluoroalkyl hydrazone meets the exacting standards required for GMP API synthesis.
We invite R&D directors and procurement leaders to explore how this metal-free electrochemical route can optimize your supply chain and reduce manufacturing costs. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific project needs, demonstrating exactly how switching to this methodology can improve your bottom line. We encourage you to contact our technical procurement team today to request specific COA data for our fluoroalkyl intermediates and to discuss route feasibility assessments for your upcoming projects. Let us help you build a more resilient and cost-effective supply chain for your critical fluorinated building blocks.
