Revolutionizing Vitamin B6 Intermediate Synthesis: A High-Yield Triphosgene Catalytic Route
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways for synthesizing critical intermediates, particularly those serving as building blocks for essential vitamins. Patent CN110483319B introduces a groundbreaking preparation method for N-alkoxy oxalyl alanine ester, a pivotal intermediate in the synthesis of Vitamin B6. This innovation addresses long-standing inefficiencies in traditional manufacturing by utilizing triphosgene and organic bases to facilitate a rapid, high-yield esterification and amidation sequence. Unlike legacy processes that rely on toxic solvents and extended reaction times, this novel approach streamlines production through a controlled two-step mechanism that operates under significantly milder thermal conditions. By replacing hazardous benzene with safer chlorinated solvents and eliminating the need for azeotropic water removal, the technology offers a robust solution for manufacturers aiming to enhance both safety profiles and operational throughput in their supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N-alkoxy oxalyl alanine esters has been plagued by significant operational bottlenecks and environmental hazards. Traditional methods, such as those disclosed in earlier patents like CN201110191443.X, frequently employ benzene as a water-carrying agent to drive esterification equilibria, posing severe health risks to operators and creating complex waste disposal challenges. Furthermore, these conventional one-pot syntheses are notoriously sluggish, often requiring reaction periods extending up to 150 hours to reach completion, which severely limits reactor turnover rates and increases energy consumption. The reliance on high-temperature reflux for water separation not only degrades heat-sensitive intermediates but also results in lower overall yields, typically hovering around 80%, due to the formation of stubborn byproducts and incomplete conversions. These factors collectively inflate production costs and complicate regulatory compliance for facilities operating under strict environmental standards.
The Novel Approach
In stark contrast, the methodology outlined in CN110483319B leverages the activating power of triphosgene to bypass the thermodynamic limitations of direct esterification. By converting alanine into an activated alanine ester hydrochloride intermediate in situ, the process eliminates the need for continuous water removal, thereby collapsing a multi-day operation into a matter of hours. This strategic shift allows the reaction to proceed at temperatures ranging from 50°C to 80°C, significantly lower than the harsh conditions required by older techniques. The result is a dramatic improvement in process efficiency, with reported yields consistently exceeding 90% and product purity levels reaching upwards of 96% as determined by gas chromatography. This approach not only mitigates the safety risks associated with benzene but also simplifies the downstream purification workflow, making it an ideal candidate for modern, green chemistry manufacturing initiatives.
Mechanistic Insights into Triphosgene-Mediated Esterification and Amidation
The core of this technological advancement lies in the precise activation of the carboxylic acid group of alanine using triphosgene, a solid and safer alternative to gaseous phosgene. In the initial step, alanine reacts with triphosgene in the presence of an organic base, such as triethylamine, within a chlorinated solvent system like chloroform or dichloromethane. This interaction generates a highly reactive mixed anhydride or acyl chloride species which is immediately intercepted by the alcohol nucleophile to form the alanine ester hydrochloride salt. This activation strategy is crucial because it drives the esterification to completion without the need to physically remove water, a common requirement in Fischer esterification that often necessitates toxic entrainers. The reaction is carefully controlled, starting at low temperatures between -10°C and 0°C during the addition of triphosgene to manage exothermicity, before warming to 50-60°C to ensure full conversion.

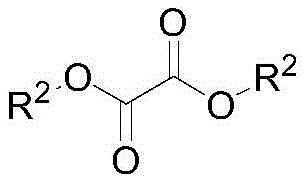
Following the formation of the alanine ester intermediate, the process transitions seamlessly into the amidation phase without isolating the salt, which minimizes material loss and handling time. The crude alanine ester hydrochloride solution is reacted with a dialkyl oxalate, such as dimethyl or diethyl oxalate, under the catalytic influence of additional organic base. This step involves the nucleophilic attack of the amino group on the carbonyl carbon of the oxalate, forming the stable N-alkoxy oxalyl linkage characteristic of the final product. The use of excess oxalate serves a dual purpose: it drives the equilibrium towards the product and acts as a scavenger for residual moisture or impurities. The structural integrity of the final molecule, as depicted in the general formula, is maintained through mild thermal conditions that prevent racemization or decomposition, ensuring the optical purity required for downstream Vitamin B6 synthesis.
Impurity control in this system is inherently superior due to the specificity of the triphosgene activation. Unlike acid-catalyzed methods that can promote oligomerization or dehydration side reactions, the base-mediated pathway selectively targets the carboxyl and amino functionalities. The post-treatment protocol further refines the product quality; after distilling off the solvent and excess oxalate, a mild neutralization with dilute sodium hydroxide removes acidic residues without hydrolyzing the sensitive ester bonds. Subsequent extraction with ethyl acetate and drying ensures that the final isolated material meets stringent purity specifications, often exceeding 96% content by gas phase analysis. This high level of chemical fidelity is essential for R&D directors who require consistent feedstock quality to maintain the efficacy and safety of the final pharmaceutical active ingredient.
How to Synthesize N-Alkoxy Oxalyl Alanine Ester Efficiently
The synthesis protocol described in the patent offers a reproducible framework for producing high-quality intermediates suitable for immediate scale-up. The procedure begins with the dissolution of alanine and an organic base in a dry organic solvent, followed by the controlled addition of a triphosgene solution at sub-zero temperatures to manage the reaction exotherm safely. Once the system clarifies, indicating the formation of the activated intermediate, the corresponding alcohol is introduced to complete the esterification. The resulting mixture is then cooled and transferred into a reactor containing the dialkyl oxalate and additional base, where the temperature is raised to facilitate the amidation coupling. For detailed standardized operating procedures, stoichiometry ratios, and specific workup parameters, please refer to the comprehensive guide below.
- Perform esterification of alanine and alcohol using triphosgene and an organic base (e.g., triethylamine) at -10°C to 60°C to form alanine ester hydrochloride.
- Conduct amidation by reacting the crude alanine ester hydrochloride with dialkyl oxalate under organic base catalysis at 60-80°C.
- Execute post-treatment involving solvent distillation, neutralization with dilute alkali, extraction, and vacuum distillation to isolate the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this triphosgene-based methodology represents a significant opportunity to optimize cost structures and mitigate supply risks. The elimination of benzene, a regulated carcinogen, removes the substantial costs associated with specialized containment, monitoring, and hazardous waste disposal, leading to a leaner operational expenditure profile. Furthermore, the drastic reduction in reaction time—from days to mere hours—enhances asset utilization rates, allowing manufacturing facilities to increase batch frequency without capital investment in new reactors. This efficiency gain translates directly into improved responsiveness to market demand fluctuations, ensuring that critical Vitamin B6 precursors can be supplied with greater reliability and shorter lead times.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the simplification of the reaction workflow and the high atom economy of the triphosgene activation. By avoiding the need for azeotropic distillation to remove water, the process saves significant energy costs associated with prolonged heating and solvent recovery. Additionally, the high yields observed, often surpassing 90%, mean that less raw material is wasted per kilogram of finished product, effectively lowering the cost of goods sold. The ability to recover and reuse excess dialkyl oxalate through simple distillation further contributes to material cost savings, creating a more sustainable and economically viable production model compared to legacy methods.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the use of readily available and stable reagents such as triphosgene and common alcohols, which are less subject to the volatility of specialized catalyst markets. The robustness of the reaction conditions, which tolerate a range of temperatures and solvent choices, reduces the risk of batch failures due to minor process deviations. This reliability ensures a steady flow of intermediates to downstream Vitamin B6 manufacturers, preventing bottlenecks that could disrupt the production of essential nutritional supplements. Moreover, the simplified purification steps reduce the dependency on complex chromatographic separations, speeding up the release of finished goods for shipment.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, this method aligns perfectly with modern green chemistry principles and regulatory expectations. The absence of benzene simplifies environmental permitting and reduces the liability associated with toxic solvent emissions. The lower operating temperatures reduce the thermal load on plant utilities, contributing to a smaller carbon footprint for the manufacturing site. As production scales from pilot batches to multi-ton campaigns, the linear scalability of the batch process ensures that quality remains consistent, allowing suppliers to confidently commit to long-term contracts with major pharmaceutical clients who demand strict adherence to EHS (Environment, Health, and Safety) standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and claims presented in patent CN110483319B, providing a clear understanding of the process capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this method into existing production lines or for procurement specialists assessing the quality of potential suppliers.
Q: How does the triphosgene method improve upon traditional benzene-based synthesis?
A: The triphosgene method eliminates the need for toxic benzene as a water-carrying agent and drastically reduces reaction time from over 150 hours to approximately 3-12 hours, while improving yields from ~80% to over 90%.
Q: What are the critical reaction conditions for the esterification step?
A: The esterification requires precise temperature control, initiating at -10°C to 0°C during triphosgene addition and heating to 50-60°C for completion, typically using chloroform or dichloromethane as the solvent.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is designed for industrial scalability due to its mild reaction temperatures (max 80°C), avoidance of complex azeotropic water removal, and high product content exceeding 96% without extensive purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Alkoxy Oxalyl Alanine Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity intermediates play in the global supply chain for vitamins and pharmaceuticals. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to delivering N-alkoxy oxalyl alanine ester with stringent purity specifications, utilizing rigorous QC labs to verify that every batch meets the exacting standards required for Vitamin B6 synthesis. Our facility is equipped to handle the specific solvent systems and temperature controls dictated by the triphosgene method, guaranteeing a product that is free from toxic residues and ready for immediate downstream processing.
We invite you to collaborate with us to leverage this advanced technology for your specific application needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our optimized manufacturing processes can enhance your supply chain resilience and reduce your overall production costs.
