Advanced FeCl3-Catalyzed Synthesis of Naphtho-imidazo-pyridine-diones for Scalable Pharmaceutical Production
Introduction to Novel NPDO Synthesis Technology
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for complex heterocyclic scaffolds, particularly those exhibiting potent biological activity. Patent CN111253395A introduces a groundbreaking methodology for the synthesis of naphtho[1',2':4,5]imidazo[1,2-a]pyridine-5,6-dione (NPDO) compounds, a class of N-heterocyclic fused ortho-dinaphthoquinones with significant potential in oncology research. These compounds function as effective electron acceptors for NADPH-dependent flavinases, inducing reactive oxygen species (ROS) generation and demonstrating high cytotoxic activity against human lung cancer (A-549) and breast cancer (MCF-7) cell lines. The disclosed technology represents a paradigm shift from traditional condensation methods, utilizing a modular three-step sequence that begins with 2-arylimidazo[1,2-a]pyridines and diazoacetonitrile. By leveraging inexpensive iron catalysis and operating under ambient atmospheric conditions, this process addresses critical pain points in intermediate manufacturing, offering a reliable pharmaceutical intermediate supplier pathway that balances high purity with economic feasibility.
![General reaction scheme showing the three-step synthesis from 2-arylimidazo[1,2-a]pyridines to NPDO compounds](/insights/img/naphtho-imidazo-pyridine-dione-fecl3-catalysis-pharma-supplier-20260306233255-01.webp)
This innovative approach not only streamlines the construction of the tetracyclic core but also ensures excellent functional group tolerance, accommodating diverse substituents such as halogens, alkyls, and trifluoromethyl groups. For R&D directors and process chemists, the ability to access these bioactive scaffolds through a concise, atom-economical route is invaluable for accelerating drug discovery programs. The patent details a comprehensive optimization of reaction parameters, establishing a foundation for commercial scale-up of complex pharmaceutical intermediates that meets the rigorous quality standards required for preclinical and clinical development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this invention, the synthesis of NPDO derivatives predominantly relied on the condensation of 2-aminopyridine compounds with 2,3-dichloro-1,4-naphthoquinone derivatives. While chemically feasible, this legacy approach suffers from significant drawbacks that hinder its utility in modern GMP manufacturing environments. The conventional route is plagued by low product yields and the formation of numerous difficult-to-remove by-products, which complicates downstream purification and drastically increases production costs. Furthermore, the atom economy of the traditional condensation reaction is poor, generating substantial chemical waste that conflicts with green chemistry principles increasingly mandated by regulatory bodies. The reliance on highly reactive quinone starting materials also introduces safety hazards and supply chain vulnerabilities, as these precursors can be unstable and expensive to source in bulk quantities. Consequently, the existing literature methods fail to provide a sustainable solution for the cost reduction in API manufacturing needed to make potential therapies commercially viable.
The Novel Approach
In stark contrast, the methodology described in CN111253395A offers a superior alternative by decoupling the ring construction into manageable, high-yielding transformations. The new strategy employs a tandem sequence initiated by an iron-catalyzed C-H functionalization, followed by esterification and a final polyphosphoric acid (PPA) mediated cyclization. This route utilizes readily available 2-arylimidazo[1,2-a]pyridines and diazoacetonitrile, both of which are stable and commercially accessible feedstocks. The process eliminates the need for hazardous chlorinated quinones and avoids the generation of stoichiometric salt waste associated with neutralization steps in older protocols. By shifting to a catalytic regime driven by earth-abundant metals, the novel approach significantly enhances the overall efficiency of the synthesis. This transition not only improves the environmental profile of the manufacturing process but also provides a more robust platform for producing high-purity OLED material precursors or pharmaceutical candidates with consistent quality batch after batch.
Mechanistic Insights into FeCl3-Catalyzed Cyclization
Understanding the underlying reaction mechanism is crucial for process optimization and troubleshooting during scale-up. The patent elucidates a plausible catalytic cycle driven by ferric chloride (FeCl3), which acts as a Lewis acid to activate the diazoacetonitrile species. As depicted in the mechanistic proposal, the iron center coordinates with the diazo compound, facilitating the extrusion of nitrogen gas and generating a reactive metal-carbenoid or radical intermediate. This activated species then undergoes electrophilic attack or radical addition at the C-3 position of the imidazopyridine ring, forming the critical carbon-carbon bond that installs the cyanomethyl group. Experimental evidence supporting a radical pathway includes inhibition studies using TEMPO, where the addition of this radical scavenger significantly suppressed product formation, confirming the involvement of free radical intermediates in the transformation.
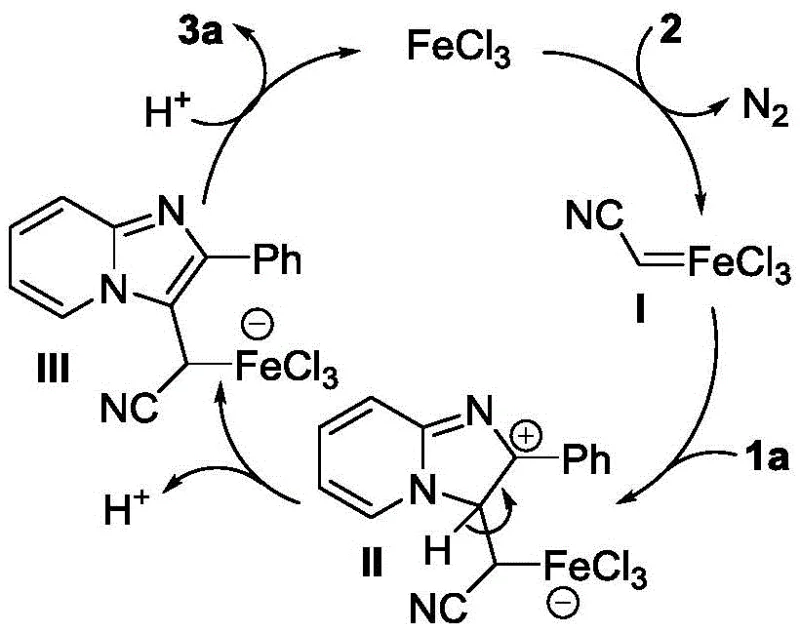
Following the initial functionalization, the subsequent steps involve acid-catalyzed hydrolysis and esterification of the nitrile group, followed by an intramolecular Friedel-Crafts-type acylation promoted by PPA. The use of PPA serves a dual purpose: it acts as both a solvent and a dehydrating agent, driving the equilibrium towards the formation of the fused dione system by removing water generated during cyclization. This mechanistic understanding allows chemists to fine-tune reaction conditions, such as temperature and acid concentration, to maximize yield while minimizing side reactions. For quality control teams, knowing that the reaction proceeds through defined intermediates helps in establishing appropriate in-process controls (IPCs) to monitor the consumption of starting materials and the appearance of key intermediates, ensuring the final product meets stringent purity specifications.
How to Synthesize Naphtho-imidazo-pyridine-dione Efficiently
The synthesis protocol outlined in the patent is designed for operational simplicity, requiring standard laboratory equipment and avoiding the need for specialized high-pressure reactors or glovebox techniques. The process is divided into three distinct stages: the initial cyanomethylation, the esterification of the nitrile side chain, and the final oxidative cyclization. Each step has been optimized to balance reaction rate with selectivity, ensuring that the final tetracyclic product is obtained with minimal impurities. The following guide summarizes the critical operational parameters derived from the patent examples, providing a roadmap for technical teams looking to implement this chemistry.
- React 2-arylimidazo[1,2-a]pyridine with diazoacetonitrile using FeCl3 catalyst in acetonitrile at 60°C under air to form the 3-cyanomethyl intermediate.
- Perform acid-catalyzed esterification of the nitrile group using alcohol solvents like ethanol with sulfuric acid to generate the acetate derivative.
- Execute intramolecular cyclization using polyphosphoric acid (PPA) at elevated temperatures (140°C) to form the final dione structure.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this FeCl3-catalyzed route offers transformative benefits that extend beyond mere chemical yield. The shift away from precious metal catalysts like rhodium or palladium to inexpensive iron salts represents a direct opportunity for cost reduction in pharmaceutical intermediate manufacturing. Iron catalysts are not only orders of magnitude cheaper but also eliminate the regulatory burden associated with removing trace heavy metals from final drug substances, thereby simplifying the purification workflow and reducing the consumption of expensive scavenging resins. Additionally, the ability to conduct all reaction steps under ambient air conditions removes the dependency on inert gas infrastructure, such as nitrogen blanketing systems, which lowers both capital expenditure for facility upgrades and ongoing operational costs related to gas consumption.
- Cost Reduction in Manufacturing: The replacement of noble metal catalysts with ferric chloride drastically lowers the raw material cost per kilogram of product. Since iron is abundant and non-toxic, waste disposal costs are also significantly reduced compared to processes generating heavy metal-contaminated effluent. Furthermore, the high atom economy of the reaction minimizes the purchase of excess reagents, contributing to substantial cost savings over the lifecycle of the product. The simplified workup procedures, which often involve basic extraction and crystallization rather than complex chromatography, further enhance the economic viability of the process for large-scale production.
- Enhanced Supply Chain Reliability: Utilizing common, commodity-grade chemicals like FeCl3, diazoacetonitrile, and PPA ensures a stable and resilient supply chain. Unlike specialized ligands or sensitive organometallic complexes that may have long lead times or single-source suppliers, the reagents for this process are widely available from multiple global vendors. This diversification mitigates the risk of production delays caused by raw material shortages. Moreover, the robustness of the reaction to air and moisture means that storage and handling requirements are less stringent, reducing the likelihood of material degradation during transit and warehousing.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated in batch reactors with straightforward thermal control requirements. The absence of pyrophoric reagents or extreme pressure conditions makes it safer to operate at the multi-kilogram or tonne scale. From an environmental standpoint, the reduced waste generation and the use of less hazardous solvents align with green chemistry metrics, facilitating easier regulatory approval and permitting. This compliance is increasingly critical for maintaining a social license to operate and meeting the sustainability goals of major pharmaceutical partners.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating this technology, we have compiled answers to common questions regarding the practical implementation and scope of the patented synthesis method. These insights are derived directly from the experimental data and comparative studies presented in the patent documentation, ensuring accuracy and relevance for process development teams.
Q: What are the advantages of using FeCl3 over noble metal catalysts for this synthesis?
A: FeCl3 is significantly more cost-effective than rhodium or palladium catalysts, reduces heavy metal contamination risks in API intermediates, and operates efficiently under ambient air conditions without inert gas protection.
Q: Does this method require strict anhydrous or anaerobic conditions?
A: No, the patent explicitly states that all three reaction steps can be conducted in air without inert gas protection, simplifying operational requirements and reducing equipment costs for scale-up.
Q: What is the substrate scope for the aryl group in this reaction?
A: The method tolerates a wide range of substituents including halogens (Cl, Br, F), alkyl groups (Me, Et), alkoxy groups (OMe), and trifluoromethyl groups on both the imidazopyridine and phenyl rings.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Naphtho-imidazo-pyridine-dione Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient synthetic routes in bringing novel therapeutics to market. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries can be successfully translated into viable manufacturing processes. We are committed to delivering high-purity pharmaceutical intermediates that adhere to stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to implement the FeCl3-catalyzed synthesis described in CN111253395A allows us to offer competitive pricing and reliable supply continuity for clients developing oncology agents based on the NPDO scaffold.
We invite you to collaborate with us to leverage this advanced technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing expertise can accelerate your development timeline and optimize your supply chain economics.
