Advanced Synthesis of Carboxylic Acid Intermediates for Commercial Scale Pharmaceutical Manufacturing
Advanced Synthesis of Carboxylic Acid Intermediates for Commercial Scale Pharmaceutical Manufacturing
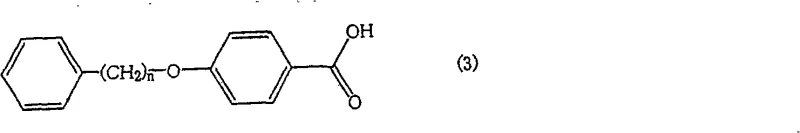
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for higher purity and more cost-effective synthetic routes. A significant breakthrough in this domain is documented in Chinese Patent CN100554236C, which details a robust process for preparing specific carboxylic acid compounds, notably 4-(4-phenylbutoxy)benzoic acid derivatives. These compounds serve as critical building blocks in the synthesis of various active pharmaceutical ingredients (APIs). The patent introduces a novel purification strategy that leverages precise pH control during the hydrolysis stage to eliminate stubborn structural impurities that have historically plagued conventional synthesis methods. This technological advancement offers a compelling value proposition for R&D directors and procurement managers seeking reliable pharmaceutical intermediate suppliers who can deliver high-purity materials without the burden of excessive processing costs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of 4-(4-phenylbutoxy)benzoic acid involves the alkylation of a hydroxybenzoate ester followed by hydrolysis. However, this conventional pathway is fraught with significant chemical challenges that impact overall yield and purity. During the initial alkylation reaction between the hydroxy ester and the halogenated alkyl chain, a competitive side reaction frequently occurs. The starting material itself can undergo self-alkylation or react with solvent components, leading to the formation of structurally similar byproducts, specifically 4-alkoxybenzoic acid esters. When the crude reaction mixture is subsequently hydrolyzed, these ester byproducts convert into carboxylic acid impurities that are chemically analogous to the target molecule.
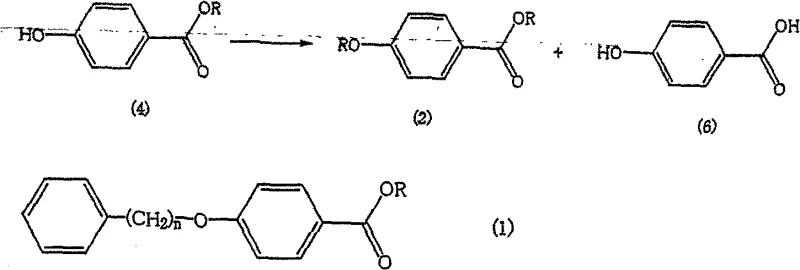
Because these impurities share nearly identical physical and chemical properties with the desired product, separating them becomes an arduous task. Standard purification techniques such as recrystallization often fail to achieve the necessary purity levels, and chromatographic separation is prohibitively expensive and difficult to scale for industrial production. Consequently, manufacturers face a dilemma: either accept lower purity specifications that may compromise downstream API synthesis or incur substantial costs to remove these persistent contaminants. This bottleneck has long been a pain point in the supply chain for complex pharmaceutical intermediates, necessitating a more elegant chemical solution.
The Novel Approach
The methodology outlined in patent CN100554236C presents a paradigm shift by addressing the impurity problem at the hydrolysis and workup stage rather than trying to prevent it entirely during alkylation. The core innovation lies in the realization that while the target carboxylic acid and the impurity carboxylic acid are structurally similar, their solubility profiles in aqueous versus organic phases can be differentially manipulated by controlling the acidity of the medium. By hydrolyzing the mixture of esters first and then carefully adjusting the pH of the resulting solution to a specific window between 4 and 8, the process enables a highly efficient liquid-liquid separation. In this optimized pH environment, the unwanted byproduct acids preferentially migrate into the aqueous layer, while the target carboxylic acid remains dissolved in the organic layer.
This approach effectively turns a difficult solid-state separation problem into a straightforward liquid-phase extraction. It allows manufacturers to utilize crude reaction mixtures containing significant amounts of byproduct esters without compromising the final quality of the carboxylic acid. The result is a streamlined workflow that reduces the number of purification steps, minimizes solvent consumption, and significantly enhances the overall mass balance of the process. For supply chain heads, this translates to a more resilient production capability that is less sensitive to variations in raw material quality or reaction conversion rates.
Mechanistic Insights into pH-Controlled Partitioning
To fully appreciate the technical sophistication of this method, one must understand the underlying physicochemical mechanisms governing the separation. The target molecule, 4-(4-phenylbutoxy)benzoic acid, possesses a longer hydrophobic alkyl chain compared to the primary impurity, typically 4-methoxybenzoic acid or similar short-chain alkoxy derivatives. While both molecules contain a carboxylic acid group that can ionize, the difference in their hydrophobicity dictates their partition coefficients. At a neutral or slightly acidic pH (specifically 4 to 8), the ionization state of the carboxylic acids is modulated. The patent data suggests that within this specific pH range, the more polar, shorter-chain impurity acids exhibit a higher affinity for the aqueous phase, especially when aided by the presence of inorganic salts or specific solvent systems.
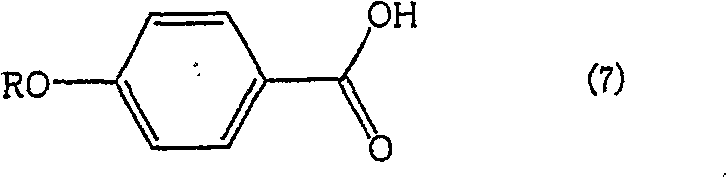
Furthermore, the process meticulously avoids pH extremes that could jeopardize yield. If the pH drops below 4, the ionization of the impurity acids is suppressed, causing them to remain in the organic layer and contaminating the product. Conversely, if the pH rises above 8, the target carboxylic acid begins to ionize excessively, forming water-soluble salts that migrate into the aqueous waste stream, thereby reducing recovery yields. The patent specifies an optimal range of pH 5 to 7, which strikes a delicate balance. This precision ensures that the organic layer, typically composed of solvents like toluene or xylene, retains the maximum amount of the target product while acting as a barrier against the hydrophilic impurities. This mechanistic understanding allows process chemists to fine-tune the workup conditions for maximum efficiency.
How to Synthesize 4-(4-Phenylbutoxy)benzoic Acid Efficiently
Implementing this synthesis route requires careful attention to the hydrolysis and pH adjustment parameters to replicate the high purity demonstrated in the patent examples. The process begins with the hydrolysis of the ester mixture, followed by the critical pH tuning step that defines the invention. Operators must monitor the acidity closely to ensure it remains within the narrow window that favors impurity removal. The following guide outlines the standardized operational framework derived from the patent data, providing a clear roadmap for technical teams aiming to adopt this superior manufacturing protocol.
- Hydrolyze the mixture containing the target ester compound and structural analog impurities using water and a base or acid catalyst under controlled temperature conditions.
- Adjust the pH of the resulting hydrolysis liquid to a specific range between 4 and 8, preferably 5 to 7, to alter the solubility profile of the impurities.
- Perform liquid phase separation to isolate the organic layer containing the target carboxylic acid, leaving the unwanted byproduct acids in the aqueous layer.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this pH-controlled hydrolysis method offers tangible strategic benefits that extend beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the downstream processing workflow. By eliminating the need for complex chromatographic purification or multiple recrystallization cycles to remove structural analogs, the process significantly reduces the consumption of high-purity solvents and stationary phases. This reduction in material usage directly correlates to a lower cost of goods sold (COGS), allowing for more competitive pricing structures in the global market for pharmaceutical intermediates. Additionally, the robustness of the liquid-liquid separation technique means that the process is less susceptible to batch-to-batch variability.
- Cost Reduction in Manufacturing: The elimination of expensive purification steps serves as a major driver for cost optimization. Traditional methods often require costly adsorbents or large volumes of specialty solvents to achieve similar purity levels. By relying on simple aqueous extraction facilitated by pH control, the process minimizes waste generation and utility consumption. This lean manufacturing approach ensures that resources are allocated efficiently, resulting in substantial cost savings that can be passed down the supply chain. Furthermore, the ability to tolerate higher levels of byproducts in the crude feedstock reduces the pressure on the upstream alkylation reaction to achieve perfect conversion, further enhancing overall process economics.
- Enhanced Supply Chain Reliability: A simpler process is inherently a more reliable one. The reliance on standard unit operations such as mixing, heating, and phase separation means that the technology can be implemented on existing multipurpose equipment without the need for specialized hardware. This flexibility reduces the risk of production bottlenecks and equipment downtime. For supply chain planners, this translates to shorter lead times and a more consistent supply of high-purity intermediates. The method's tolerance for variation in raw material quality also mitigates the risk of batch failures due to minor fluctuations in starting material specifications, ensuring a steady flow of product to downstream API manufacturers.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges, but this method scales linearly with ease. Liquid-liquid extraction is a well-understood operation in chemical engineering, readily adaptable from pilot plant to multi-ton commercial reactors. Moreover, the environmental footprint of the process is favorably reduced. By avoiding heavy metal catalysts and minimizing the use of hazardous organic solvents for purification, the process aligns better with modern green chemistry principles. The aqueous waste streams generated are easier to treat, and the overall reduction in solvent waste contributes to a more sustainable manufacturing profile, which is increasingly important for compliance with international environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and claims presented in the patent documentation, offering clarity on the operational nuances and potential advantages for industrial application. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing production portfolios.
Q: Why is pH adjustment critical in this synthesis process?
A: Adjusting the pH to between 4 and 8 is the key innovation that allows for the selective partitioning of impurities. At this specific pH range, the unwanted byproduct carboxylic acids (such as 4-methoxybenzoic acid) become significantly more soluble in the aqueous phase, while the target product remains in the organic phase, enabling high-purity isolation without complex chromatography.
Q: What are the primary impurities addressed by this patent technology?
A: The process specifically targets structural analogs formed during the initial alkylation step, such as 4-alkoxybenzoic acid derivatives. These impurities have chemical structures very similar to the target molecule, making them traditionally difficult to separate. This method effectively removes them via aqueous extraction.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process relies on standard unit operations such as hydrolysis, pH adjustment, and liquid-liquid separation. It avoids the need for expensive transition metal catalysts or complex purification columns, making it highly scalable and cost-effective for commercial manufacturing of pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-(4-Phenylbutoxy)benzoic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team of process chemists has extensively analyzed advanced synthetic routes like the one described in CN100554236C to ensure we offer the most efficient solutions for our clients. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this pH-controlled purification method are fully realized at an industrial scale. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of carboxylic acid intermediate meets the exacting standards required for pharmaceutical synthesis.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage these process improvements for their supply chains. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us demonstrate how our commitment to advanced process chemistry can drive efficiency and reliability in your production of high-value pharmaceutical intermediates.
