Advanced Synthesis of 4-Cyanopiperidine Hydrochloride for Scalable Pharmaceutical Manufacturing
Advanced Synthesis of 4-Cyanopiperidine Hydrochloride for Scalable Pharmaceutical Manufacturing
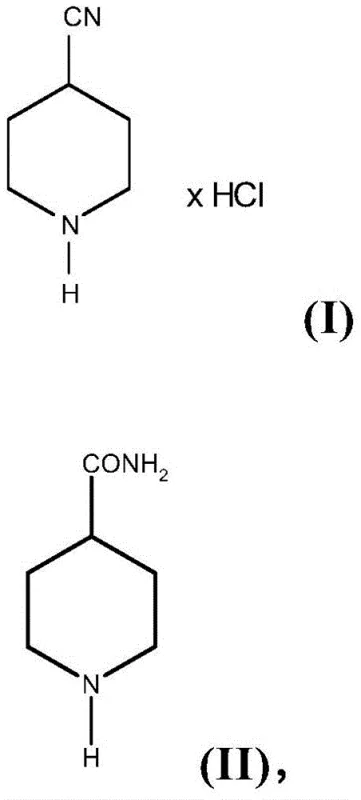
The pharmaceutical and agrochemical industries rely heavily on robust, scalable synthetic routes for key heterocyclic building blocks, and 4-cyanopiperidine represents a critical scaffold in this domain. As detailed in the recent intellectual property disclosure CN107207435B, a novel methodology has emerged that fundamentally alters the production landscape for 4-cyanopiperidine hydrochloride. This patent introduces a streamlined dehydration protocol that utilizes thionyl chloride in the presence of specific formamide derivatives to convert 4-piperidinecarboxamide directly into the desired hydrochloride salt. Unlike traditional methods that struggle with low yields and complex workups involving multiple extractions, this innovative approach leverages the unique reactivity of formamide-promoted chlorination to achieve superior purity and isolation efficiency. For R&D directors and process chemists, this represents a significant opportunity to optimize the supply chain for active pharmaceutical ingredients (APIs) that incorporate this piperidine motif, ensuring both economic viability and technical reliability in large-scale manufacturing environments.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4-cyanopiperidine has been plagued by inefficiencies that render many legacy processes unsuitable for modern industrial scale-up. Prior art, such as the method described in US 5,780,466, relies on phosphorus oxychloride (POCl3) for dehydration, a route that notoriously suffers from abysmal theoretical yields of approximately 29.7% and necessitates a laborious workup involving pH adjustments to 13 and multiple extractions with dichloromethane and diethyl ether. Similarly, alternative pathways utilizing trifluoroacetic anhydride, as seen in DE 3031892, introduce additional complexity by requiring a two-step sequence where an intermediate trifluoroacetyl protected species must be hydrolyzed, ultimately delivering yields as low as 27.1%. Even methods employing excess thionyl chloride without promoters, referenced in WO 2010/104899, often result in poor isolation yields around 36% due to the high water solubility of the free base product, forcing manufacturers to engage in energy-intensive distillation and solvent-heavy extraction protocols that inflate operational costs and environmental waste.
The Novel Approach
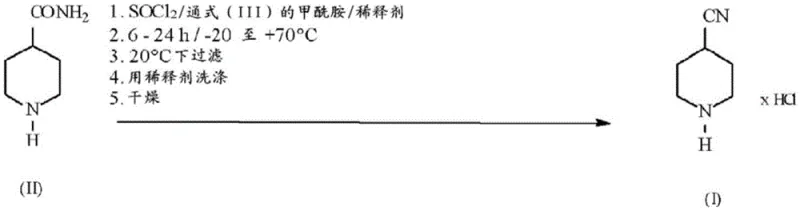
In stark contrast to these cumbersome historical precedents, the process disclosed in CN107207435B offers a paradigm shift by enabling the direct isolation of 4-cyanopiperidine as its hydrochloride salt through a single-pot dehydration reaction. By introducing a formamide derivative, such as dibutylformamide, into the reaction matrix alongside thionyl chloride, the system generates a highly reactive intermediate that facilitates rapid dehydration under mild conditions, typically between 0°C and 50°C. This modification eliminates the need for neutralizing large volumes of acid with strong bases or performing exhaustive liquid-liquid extractions to recover the product from aqueous phases. Instead, the product precipitates directly from the organic diluent as a solid, allowing for simple filtration and washing. This transition from a multi-step extraction-based workflow to a precipitation-based isolation strategy drastically reduces solvent consumption and processing time, providing a clear pathway for cost reduction in pharmaceutical manufacturing while simultaneously enhancing the overall mass balance of the synthesis.
Mechanistic Insights into Formamide-Promoted Dehydration
The core innovation of this technology lies in the synergistic interaction between thionyl chloride and the formamide promoter, which effectively creates a Vilsmeier-Haack-type reagent in situ. When thionyl chloride reacts with the formamide of general formula (III), it generates a chloroiminium salt species that serves as a potent electrophile. This activated complex attacks the carbonyl oxygen of the 4-piperidinecarboxamide substrate, forming an imidoyl chloride intermediate that is far more susceptible to elimination than the parent amide. The presence of the formamide catalyst lowers the activation energy required for the dehydration step, allowing the reaction to proceed efficiently at ambient or slightly elevated temperatures without the need for the harsh thermal conditions often associated with non-catalyzed dehydrations. This mechanistic pathway ensures that the conversion is not only faster but also cleaner, minimizing the formation of polymeric byproducts or degradation species that typically contaminate the crude product in uncatalyzed variations.
Furthermore, the choice of isolating the product as the hydrochloride salt is a strategic masterstroke in impurity control. In conventional free-base syntheses, the high polarity and water solubility of 4-cyanopiperidine make it difficult to separate from inorganic salts and residual solvents, often leading to significant product loss during aqueous workups. By maintaining the acidic environment generated by the thionyl chloride and avoiding neutralization, the product remains protonated and insoluble in the non-polar or moderately polar organic diluents used, such as toluene or alkyl acetates. This differential solubility allows the pure hydrochloride salt to crystallize or precipitate out of the solution, leaving soluble organic impurities and excess reagents in the mother liquor. Consequently, the filtration step acts as a powerful purification event, yielding a solid with purity levels exceeding 95% w/w as confirmed by quantitative NMR and GC analysis in the patent examples, thereby reducing the burden on subsequent recrystallization or chromatographic purification steps.
How to Synthesize 4-Cyanopiperidine Hydrochloride Efficiently
Implementing this advanced dehydration protocol requires careful attention to reagent stoichiometry and temperature control to maximize the benefits of the formamide promotion effect. The process is designed to be operationally simple, utilizing standard reactor equipment and commonly available solvents, making it highly accessible for contract development and manufacturing organizations (CDMOs) looking to upgrade their production capabilities. The following guide outlines the critical operational parameters derived from the patent examples, focusing on the optimal molar ratios and reaction conditions that ensure high conversion and easy isolation. For detailed standard operating procedures and safety guidelines regarding the handling of thionyl chloride, please refer to the specific technical documentation provided by your chemical supplier.
- Suspend 4-piperidinecarboxamide in a suitable organic diluent such as n-propyl acetate or toluene under inert atmosphere.
- Add a catalytic amount of a formamide derivative, preferably dibutylformamide, to the suspension at ambient temperature.
- Slowly add thionyl chloride while maintaining temperature control, then stir the mixture for 6 to 24 hours to complete dehydration.
- Filter the resulting suspension to isolate the solid 4-cyanopiperidine hydrochloride, wash with the reaction diluent, and dry under vacuum.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route offers tangible benefits that extend beyond mere chemical yield, addressing critical pain points related to cost structure and logistical reliability. The elimination of multiple extraction and distillation steps translates directly into a significant reduction in utility consumption and solvent procurement costs, as the process relies on a simple filtration and wash sequence rather than energy-intensive separation technologies. Furthermore, the ability to use a broader range of inexpensive diluents, such as toluene and various acetates, provides flexibility in sourcing raw materials, mitigating the risk of supply disruptions associated with specialized or regulated solvents. This operational simplicity also enhances the robustness of the supply chain, as the reduced number of unit operations decreases the likelihood of batch failures or deviations, ensuring a more consistent and reliable flow of high-purity intermediates to downstream API manufacturing sites.
- Cost Reduction in Manufacturing: The primary economic driver of this technology is the drastic simplification of the downstream processing workflow. By avoiding the need for large quantities of inorganic bases like sodium hydroxide or potassium carbonate to neutralize reaction mixtures, manufacturers can eliminate the cost of these reagents as well as the disposal costs associated with the resulting saline wastewater. Additionally, the direct precipitation of the product removes the necessity for vacuum distillation of the free base, which is not only energy-intensive but also poses thermal stability risks; this qualitative shift in processing logic leads to substantial cost savings in both energy and waste management without compromising product quality.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes significantly to supply security. Since the process operates effectively at mild temperatures ranging from 0°C to 50°C and tolerates a variety of commercially available solvents, it is less susceptible to the bottlenecks that often plague processes requiring cryogenic conditions or exotic reagents. The use of thionyl chloride, a commodity chemical with a stable global supply, further ensures that raw material availability remains consistent. Moreover, the high purity of the isolated solid reduces the dependency on external purification services or complex chromatography columns, streamlining the internal production schedule and reducing lead times for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this method aligns well with green chemistry principles by minimizing solvent usage and waste generation. The avoidance of chlorinated solvents like dichloromethane in the workup phase, favoring greener alternatives like esters or aromatics that can be easily recovered and recycled, supports corporate sustainability goals. The process is inherently scalable because the exotherm is manageable through controlled addition rates, and the solid-liquid separation is straightforward even in large-scale reactors, removing the technical barriers often encountered when translating extraction-heavy lab processes to multi-ton commercial production scales.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this dehydration technology. These insights are derived directly from the experimental data and scope of protection defined in the patent literature, providing a factual basis for evaluating the feasibility of this route for your specific project needs. Understanding these nuances is essential for process engineers and technical buyers who are assessing the compatibility of this method with existing infrastructure and quality standards.
Q: What is the role of formamide in this dehydration reaction?
A: Formamide acts as a crucial promoter or catalyst that activates the thionyl chloride, facilitating the formation of an reactive iminium intermediate which significantly accelerates the dehydration of the carboxamide group to the nitrile, allowing the reaction to proceed efficiently at mild temperatures.
Q: Why is isolating the hydrochloride salt advantageous over the free base?
A: Isolating the hydrochloride salt avoids the difficult extraction and distillation steps required for the free base, which is highly water-soluble and prone to losses; the salt precipitates directly from the reaction mixture as a high-purity solid, simplifying downstream processing.
Q: What solvents are compatible with this synthesis protocol?
A: The process demonstrates excellent compatibility with a wide range of organic diluents, particularly aromatic hydrocarbons like toluene and xylene, as well as esters such as n-propyl acetate and n-butyl acetate, offering flexibility for solvent recovery and cost optimization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Cyanopiperidine Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and a commitment to quality excellence. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising yields and purity profiles demonstrated in patent literature can be reliably reproduced in our state-of-the-art facilities. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to verify that every batch of 4-cyanopiperidine hydrochloride meets the exacting standards required for pharmaceutical and agrochemical applications, giving you confidence in the consistency of your supply.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can be integrated into your supply chain to drive efficiency and value. By requesting a Customized Cost-Saving Analysis, you can gain a clearer understanding of the economic impact of switching to this precipitation-based method compared to your current sourcing strategy. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments tailored to your volume requirements, ensuring a seamless partnership that supports your long-term growth and innovation goals.
