Advanced Synthesis of Calix[n]dihydroacridine Macrocycles for Next-Generation OLED Materials
The landscape of organic optoelectronic materials is undergoing a significant transformation with the emergence of novel macrocyclic structures that offer superior photophysical properties. Patent CN113831340B introduces a groundbreaking class of compounds known as calix[n]dihydroacridine macrocycles, which represent a substantial leap forward in the design of functional materials for the electronics sector. These macrocycles are characterized by a unique structural framework where dihydroacridine units are linked via methylene or alkyl bridges to form a cyclic architecture with a well-defined internal cavity. The general structure, designated as Formula TM, allows for extensive tunability through various substituents (R1, R2, R3, R4), enabling precise control over the electronic and steric environment of the molecule. This structural versatility is critical for tailoring material properties for specific high-end applications.
![General structural formula TM of calix[n]dihydroacridine macrocycle showing variable substituents R1-R4 and repeating unit n](/insights/img/calix-dihydroacridine-macrocycle-synthesis-oled-supplier-20260305061427-01.png)
Beyond their structural novelty, these compounds exhibit exceptional performance metrics that address key challenges in modern display technologies. The patent highlights their capability to function as delayed fluorescence materials, a property that is increasingly sought after for the development of high-efficiency organic light-emitting diodes (OLEDs). Furthermore, the rigid macrocyclic structure imparts good crystallinity in the solid state, which is advantageous for adsorption and separation processes in supramolecular chemistry. For R&D directors and procurement specialists in the electronic chemical industry, understanding the synthesis and potential of these materials is paramount, as they offer a pathway to next-generation devices with improved efficiency and stability. The ability to synthesize these complex architectures from simple starting materials marks a pivotal moment for reliable OLED material suppliers looking to expand their portfolio.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of large macrocyclic molecules has been plagued by significant thermodynamic and kinetic challenges that hinder their widespread adoption in commercial manufacturing. Traditional methods often rely on high-dilution conditions to favor intramolecular cyclization over intermolecular polymerization, a strategy that is inherently inefficient and difficult to scale. These processes typically require vast amounts of solvent, leading to increased operational costs and environmental burdens associated with solvent recovery and waste disposal. Moreover, conventional routes frequently suffer from low yields and poor selectivity, resulting in complex product mixtures that necessitate rigorous and costly purification steps. The use of harsh reaction conditions, such as extreme temperatures or strong bases, can also limit the compatibility of functional groups, restricting the structural diversity of the final macrocyclic products. For supply chain heads, these inefficiencies translate into longer lead times and higher risks of supply discontinuity, making the reliable sourcing of high-purity macrocycles a persistent bottleneck in the value chain.
The Novel Approach
In stark contrast to these legacy techniques, the methodology disclosed in CN113831340B offers a streamlined and robust alternative that effectively circumvents the traditional pitfalls of macrocyclization. This novel approach utilizes a direct condensation reaction between dihydroacridine derivatives and simple aldehyde compounds, catalyzed by accessible Lewis acids. By operating under mild conditions, specifically at temperatures ranging from 0°C to 30°C, the process minimizes energy consumption and reduces the risk of thermal degradation of sensitive intermediates. The reaction demonstrates remarkable efficiency, with specific embodiments achieving yields as high as 90%, which is exceptionally rare for macrocyclic synthesis. This high efficiency not only maximizes the utilization of raw materials but also significantly simplifies the downstream purification process, often requiring only standard column chromatography or recrystallization. For manufacturers focused on cost reduction in electronic chemical manufacturing, this shift from complex, low-yield protocols to a direct, high-yield condensation represents a transformative opportunity to optimize production economics.
Mechanistic Insights into Lewis Acid-Catalyzed Cyclization
The core of this synthetic breakthrough lies in the precise mechanistic pathway facilitated by the Lewis acid catalyst, which orchestrates the formation of the macrocyclic ring with high fidelity. The reaction initiates with the activation of the aldehyde component by the Lewis acid, such as ferric chloride (FeCl3) or aluminum trichloride (AlCl3), generating a highly electrophilic species. This activated electrophile then undergoes an electrophilic aromatic substitution with the electron-rich dihydroacridine ring system, forming a carbon-carbon bond at the para-position relative to the nitrogen atom. The propagation of this reaction involves sequential additions of the aldehyde to multiple dihydroacridine units, eventually closing the ring to form the stable calix[n]structure. The choice of catalyst and the stoichiometric ratio of reactants play a crucial role in directing the oligomerization towards the desired cyclic n-mer rather than linear polymers. Understanding this mechanism allows chemists to fine-tune the reaction parameters, such as the molar ratio of dihydroacridine to aldehyde (optimized at 1:3 to 1:10), to maximize the formation of the target macrocycle while suppressing side reactions.
Furthermore, the structural integrity and purity of the final product are heavily influenced by the steric and electronic properties of the substituents on the dihydroacridine core. The presence of bulky groups like tert-butyl or methyl at the 9-position of the acridine ring helps to pre-organize the molecule and prevent unwanted conformational flexibility that could lead to polymerization. This steric hindrance, combined with the specific electronic activation provided by the nitrogen substituent, ensures that the cyclization proceeds with high regioselectivity. From a quality control perspective, this mechanistic clarity is vital for ensuring batch-to-batch consistency, a key requirement for pharmaceutical and electronic grade materials. The ability to predict and control the impurity profile through mechanistic understanding means that manufacturers can achieve stringent purity specifications without resorting to excessive reprocessing, thereby enhancing the overall sustainability and economic viability of the production process.
How to Synthesize Calix[n]dihydroacridine Macrocycle Efficiently
The practical implementation of this synthesis route is straightforward and relies on standard laboratory equipment, making it highly amenable to technology transfer and scale-up. The process begins by dissolving the dihydroacridine precursor and the aldehyde source, such as paraformaldehyde, in a common organic solvent like dichloromethane or chloroform. Once the solution is homogenized, the Lewis acid catalyst is introduced, initiating the cyclization cascade. The reaction is allowed to proceed at ambient or slightly cooled temperatures for a duration of 2 to 24 hours, depending on the specific reactivity of the substrates involved. Upon completion, the reaction mixture is quenched with a mild base, such as saturated sodium bicarbonate, to neutralize the acid catalyst and stop the reaction. The crude product is then isolated through standard workup procedures including extraction, drying, and concentration, followed by purification via column chromatography to afford the pure macrocyclic compound. The detailed standardized synthesis steps are outlined in the guide below.
- Mix the dihydroacridine derivative (Formula A) with an aldehyde compound such as paraformaldehyde in a suitable solvent like dichloromethane.
- Add a Lewis acid catalyst such as ferric chloride (FeCl3) at a molar ratio of approximately 1: 0.1 relative to the substrate.
- Stir the reaction mixture at a temperature between 0°C and 30°C for 2 to 24 hours, followed by quenching and purification via column chromatography.
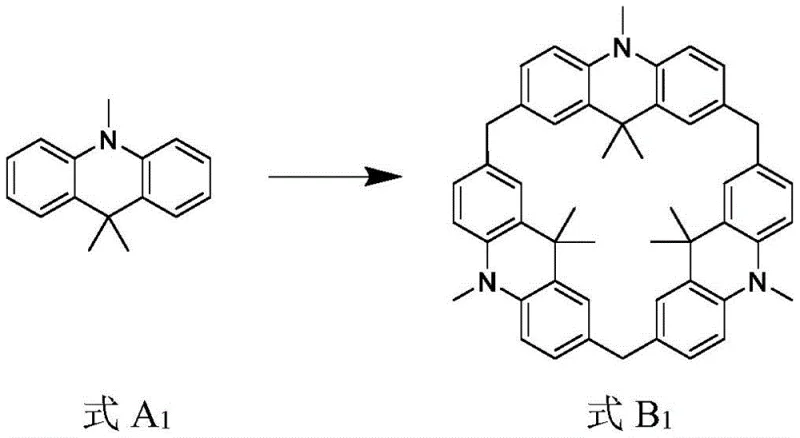
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this new synthetic methodology offers compelling strategic advantages that extend beyond mere technical feasibility. The primary benefit lies in the drastic simplification of the supply chain for raw materials, as the process utilizes commodity chemicals like paraformaldehyde and commercially available dihydroacridine derivatives. This reliance on widely sourced feedstocks mitigates the risk of supply disruptions that often plague specialized fine chemical manufacturing. Additionally, the elimination of exotic or hazardous reagents reduces the regulatory burden and safety costs associated with handling dangerous materials. The high yields reported in the patent examples suggest a significant reduction in material waste, which directly correlates to lower cost of goods sold (COGS). By minimizing the number of synthetic steps and avoiding complex protection-deprotection sequences, the overall production timeline is compressed, allowing for faster response to market demands. These factors collectively contribute to a more resilient and cost-effective supply chain for high-purity electronic chemicals.
- Cost Reduction in Manufacturing: The economic impact of this process is profound, primarily driven by the use of inexpensive Lewis acid catalysts and the avoidance of high-dilution conditions. Traditional macrocycle synthesis often requires thousands of liters of solvent to produce kilograms of product, whereas this method operates at much higher concentrations, drastically reducing solvent purchase and disposal costs. Furthermore, the high selectivity of the reaction minimizes the formation of byproducts, which reduces the load on purification units and lowers the consumption of silica gel and eluents during chromatography. The ability to run the reaction at near-room temperature also translates to substantial energy savings compared to processes requiring reflux or cryogenic cooling. These cumulative efficiencies result in a leaner manufacturing process that delivers significant cost savings without compromising on the quality of the final OLED material.
- Enhanced Supply Chain Reliability: From a logistics perspective, the robustness of this synthesis ensures a steady and predictable flow of materials. Since the reaction tolerates a variety of solvents and catalysts, manufacturers have the flexibility to switch suppliers or substitute reagents in case of shortages without needing to revalidate the entire process. The simplicity of the workup procedure, involving basic aqueous washes and standard drying, means that production can be easily scaled across different facilities with varying levels of infrastructure. This flexibility is crucial for maintaining continuity of supply in a volatile global market. Moreover, the stability of the intermediates and the final macrocyclic products facilitates easier storage and transportation, reducing the risk of degradation during transit. For supply chain heads, this reliability translates into reduced safety stock requirements and improved inventory turnover rates.
- Scalability and Environmental Compliance: The environmental footprint of this manufacturing process is significantly lower than conventional alternatives, aligning with the increasing global emphasis on green chemistry and sustainability. The reduction in solvent usage and the use of less toxic catalysts contribute to a cleaner production profile, easing compliance with stringent environmental regulations. The high atom economy of the condensation reaction ensures that a large proportion of the starting materials are incorporated into the final product, minimizing waste generation. Scalability is further supported by the exothermic nature of the reaction being manageable under the specified temperature range, allowing for safe operation in large-scale reactors. This combination of environmental stewardship and scalable engineering makes the process ideal for commercial scale-up of complex polymer additives and electronic materials, positioning companies to meet future demand sustainably.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the integration of these materials into their existing workflows, we have compiled a set of frequently asked questions based on the technical disclosures in the patent. These queries address common concerns regarding the versatility of the synthesis, the functional properties of the resulting macrocycles, and their potential applications in advanced devices. The answers provided are derived directly from the experimental data and theoretical framework presented in CN113831340B, ensuring accuracy and relevance for R&D professionals. Understanding these details is essential for making informed decisions about material selection and process optimization.
Q: What are the primary applications of calix[n]dihydroacridine macrocycles?
A: According to patent CN113831340B, these macrocycles exhibit excellent photophysical properties such as delayed fluorescence, making them highly suitable for organic light-emitting diodes (OLEDs). Additionally, their defined cavity sizes allow for host-guest chemistry applications in adsorption and separation.
Q: What catalysts are used in the synthesis of these macrocycles?
A: The synthesis utilizes common Lewis acids as catalysts, including ferric chloride (FeCl3), aluminum trichloride (AlCl3), boron trifluoride etherate, and trifluoromethanesulfonic acid, which facilitates the condensation reaction under mild conditions.
Q: Is the synthesis process scalable for industrial production?
A: Yes, the process is designed for scalability. It employs readily available raw materials, operates at mild temperatures (0-30°C), and achieves high yields (up to 90% in specific examples), indicating strong potential for commercial scale-up of complex electronic chemicals.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Calix[n]dihydroacridine Macrocycle Supplier
As the demand for high-performance organic electronic materials continues to surge, partnering with an experienced CDMO like NINGBO INNO PHARMCHEM becomes a strategic imperative for success. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to industrial reality is seamless. We understand the critical importance of stringent purity specifications in the OLED and semiconductor industries, and our rigorous QC labs are equipped to verify every batch against the highest international standards. By leveraging our expertise in Lewis acid catalysis and macrocyclization, we can help you optimize the synthesis of calix[n]dihydroacridine macrocycles to achieve maximum yield and minimal impurity levels. Our commitment to quality and scalability makes us the ideal partner for bringing next-generation fluorescent materials to market.
We invite you to engage with our technical procurement team to discuss how we can tailor this synthesis route to your specific volume and purity requirements. Whether you need a Customized Cost-Saving Analysis for your current supply chain or require specific COA data to validate our capabilities, we are ready to provide comprehensive support. Let us collaborate to accelerate your development timeline and secure a reliable supply of these cutting-edge electronic chemicals. Contact us today to request route feasibility assessments and discover how our manufacturing prowess can drive your innovation forward.
