Optimizing Trilaciclib Intermediate Production: A Technical Analysis of Patent CN115536663A
The pharmaceutical landscape for CDK4/6 inhibitors has been significantly advanced by the introduction of Trilaciclib, a first-in-class agent designed to protect bone marrow during chemotherapy. As global demand for this critical oncology therapeutic escalates, the efficiency of its supply chain becomes paramount. Patent CN115536663A discloses a novel synthetic methodology for a key intermediate, specifically 2'-chloro-7',8'-dihydro-6'H-spiro[cyclohexane-1,9'-pyrazino[1',2':1,5]pyrrolo[2,3-d]pyrimidin]-6'-one, often referred to as Compound 5. This technical disclosure represents a strategic pivot from legacy manufacturing protocols, addressing long-standing bottlenecks in yield and raw material availability. By re-engineering the construction of the spirocyclic core, this innovation offers a robust pathway for high-purity pharmaceutical intermediates that aligns with modern Good Manufacturing Practice (GMP) standards. The structural relationship between the final active pharmaceutical ingredient and this pivotal precursor is illustrated below, highlighting the complexity of the spiro-fused heterocyclic system.
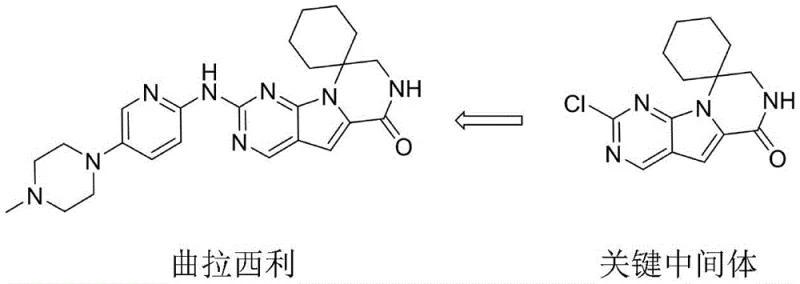
For R&D directors evaluating technology transfer, understanding the mechanistic departure from prior art is essential. The new route leverages a Sonogashira coupling followed by a streamlined cyclization and oxidation sequence, fundamentally altering the economic and operational profile of the synthesis. This report provides a deep-dive analysis into the technical merits of this patent, offering actionable insights for procurement and supply chain stakeholders seeking a reliable pharmaceutical intermediate supplier capable of delivering consistent quality at scale.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this complex spirocyclic scaffold has been plagued by inefficient multi-step sequences that rely on expensive and hazardous reagents. Early methodologies, such as those described in WO2012061156A, utilized 3,3-diethoxyprop-1-yne as a building block. While chemically feasible, this acetal-protected alkyne is commercially expensive and necessitates a cumbersome two-step conversion involving acidolysis followed by oxidation to generate the requisite carboxylic acid functionality. Furthermore, the reliance on Oxone for the oxidation step introduces significant safety liabilities during process amplification, as peroxide-based oxidants can be unstable and difficult to handle in large reactor volumes. Another competing approach, detailed in CN111867592A, attempted to shorten the route by employing methyl propiolate. However, this strategy introduced new challenges; methyl propiolate is highly reactive and prone to base-catalyzed self-polymerization, which drastically reduces the conversion rate of the Sonogashira coupling and complicates purification. Additionally, the starting material 5-iodo-2,4-dichloropyrimidine used in this alternative route commands a premium price compared to bromo-analogs, further inflating the cost of goods sold (COGS). These cumulative inefficiencies result in low overall yields and a process that is ill-suited for the rigorous demands of commercial-scale API manufacturing.
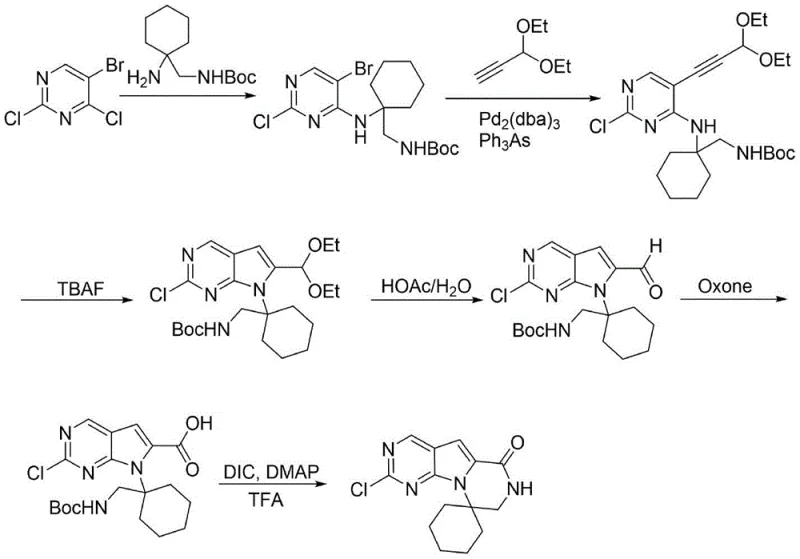
The Novel Approach
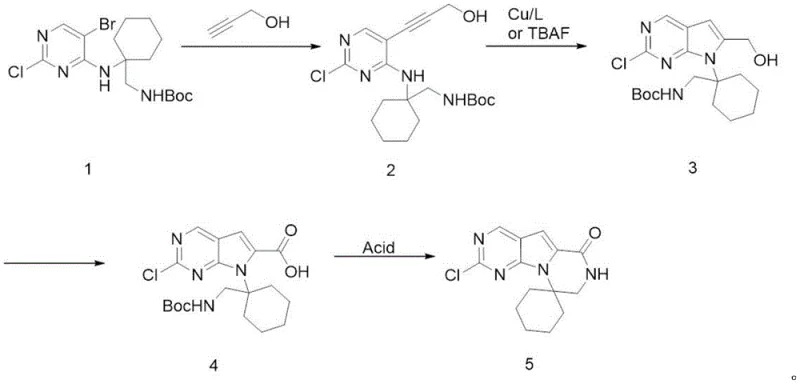
In stark contrast to these legacy methods, the methodology outlined in CN115536663A introduces a paradigm shift by utilizing propargyl alcohol as the alkyne source. This commodity chemical is not only significantly more affordable than protected alkynes or activated esters but also eliminates the need for harsh deprotection steps later in the sequence. The new route achieves the construction of the pyrrolo[2,3-d]pyrimidine core through a direct cyclization of the propargyl alcohol adduct, followed by a mild TEMPO-catalyzed oxidation to install the carboxylic acid. This strategic substitution removes the safety risks associated with Oxone and avoids the polymerization issues inherent to methyl propiolate. The final transformation involves a sophisticated one-pot protocol where the Boc protecting group is removed and the seven-membered lactam ring is closed simultaneously under acidic conditions. This convergence of steps not only reduces the total number of unit operations but also minimizes material loss between stages. For a reliable pharmaceutical intermediate supplier, this translates to a process that is inherently safer, more cost-effective, and easier to scale, ensuring a stable supply of critical materials for downstream drug production.
Mechanistic Insights into Cu-Catalyzed Cyclization and TEMPO Oxidation
The success of this synthetic strategy hinges on the precise control of two critical transformations: the formation of the pyrrole ring and the selective oxidation of the primary alcohol. The cyclization step, converting Compound 2 to Compound 3, can be achieved via two distinct mechanistic pathways depending on the reagents employed. In the copper-mediated variant, a copper(I) salt such as cuprous chloride acts in concert with a strong organic base like DBU (1,8-Diazabicyclo[5.4.0]undec-7-ene). The base deprotonates the terminal alkyne or facilitates the nucleophilic attack of the pyrimidine nitrogen onto the activated alkyne moiety, driving the 5-endo-dig or 5-exo-dig cyclization to close the pyrrole ring. Alternatively, the patent discloses the use of Tetrabutylammonium fluoride (TBAF), which likely activates the alkyne through hypervalent silicon interactions or simply acts as a mild base to promote the intramolecular nucleophilic attack. This flexibility allows process chemists to tune the reaction conditions to minimize side products, ensuring that the resulting intermediate possesses the high purity required for subsequent steps. The ability to switch between copper catalysis and fluoride promotion provides a valuable contingency for troubleshooting during technology transfer.
Following cyclization, the oxidation of the hydroxymethyl group to the carboxylic acid (Compound 3 to Compound 4) is executed using a TEMPO (2,2,6,6-Tetramethylpiperidin-1-yl)oxyl catalytic system. Unlike stoichiometric heavy metal oxidants, TEMPO operates via a radical mechanism where the nitroxyl radical cycles between its oxidized and reduced forms, mediated by a co-oxidant such as iodobenzene diacetate (BAIB) or sodium hypochlorite. This method is exceptionally selective for primary alcohols, leaving the sensitive heterocyclic core and the Boc-protected amine intact. The patent specifies a temperature range of -20°C to 90°C, allowing for fine-tuning to prevent over-oxidation or degradation of the spiro-cycle. By avoiding aggressive oxidants like potassium permanganate or chromic acid, this step significantly reduces the burden on wastewater treatment systems and eliminates the risk of heavy metal contamination in the final product. This mechanistic elegance ensures that the commercial scale-up of complex pharmaceutical intermediates can proceed with minimal environmental impact and maximum safety.
How to Synthesize Trilaciclib Intermediate Efficiently
The practical implementation of this patent requires strict adherence to the optimized reaction parameters to maximize yield and purity. The process begins with the Sonogashira coupling of the bromo-pyrimidine starting material with propargyl alcohol, utilizing a palladium catalyst system that balances activity with cost. Following isolation, the cyclization step must be carefully monitored to ensure complete ring closure without inducing decomposition of the sensitive Boc group. The subsequent oxidation requires precise stoichiometry of the co-oxidant to drive the reaction to completion while maintaining the integrity of the pyrrolo-pyrimidine scaffold. Finally, the one-pot deprotection and lactamization represent the most critical stage, where acid concentration and temperature must be controlled to facilitate ring closure without charring or polymerization. For detailed operational parameters, solvent choices, and workup procedures, please refer to the standardized synthesis guide below.
- Perform Sonogashira coupling of compound 1 with propargyl alcohol using Pd/Cu catalysts to yield compound 2.
- Execute cyclization of compound 2 using copper salts/base or TBAF to form the pyrrolo-pyrimidine core (compound 3).
- Oxidize the primary alcohol of compound 3 to carboxylic acid (compound 4) using TEMPO-catalyzed methods.
- Conduct one-pot Boc deprotection and intramolecular cyclization under acidic conditions to obtain target compound 5.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of the synthetic route described in CN115536663A offers profound advantages for procurement managers and supply chain directors tasked with securing raw materials for oncology drug production. The primary driver of value in this new methodology is the drastic simplification of the raw material portfolio. By replacing specialized, high-cost reagents like 3,3-diethoxyprop-1-yne and methyl propiolate with commodity-grade propargyl alcohol, the direct material costs are significantly reduced. Propargyl alcohol is produced on a massive industrial scale for various applications, ensuring a stable and competitive pricing structure that is less susceptible to the volatility often seen in niche fine chemical markets. Furthermore, the elimination of the acetal deprotection step and the replacement of hazardous Oxone oxidation with a milder TEMPO system reduces the consumption of auxiliary chemicals and solvents. This streamlining of the bill of materials (BOM) directly contributes to cost reduction in pharmaceutical intermediate manufacturing, allowing for more competitive pricing models in the final supply agreements.
- Cost Reduction in Manufacturing: The economic benefits of this route extend beyond simple raw material substitution. The consolidation of the final deprotection and cyclization into a single one-pot operation eliminates an entire isolation and purification stage. In industrial chemistry, each unit operation adds labor, energy, equipment time, and solvent waste disposal costs. By removing a discrete step, the overall processing time is shortened, and the throughput of the manufacturing facility is increased. Additionally, the avoidance of expensive palladium-heavy catalyst systems in favor of more economical copper salts for cyclization further drives down the variable costs. The qualitative improvement in process efficiency means that the same production capacity can yield a greater volume of finished intermediate, effectively lowering the fixed cost allocation per kilogram. This structural cost advantage provides a buffer against market fluctuations and ensures long-term financial sustainability for the supply chain.
- Enhanced Supply Chain Reliability: Supply chain resilience is heavily dependent on the availability and lead times of starting materials. The reliance on 5-iodo-2,4-dichloropyrimidine in previous methods created a bottleneck, as iodinated heterocycles often have longer lead times and fewer qualified suppliers compared to their brominated counterparts. The new route utilizes 5-bromo-2-chloropyrimidine derivatives, which are more readily accessible from a broader network of chemical manufacturers. Moreover, the stability of propargyl alcohol simplifies logistics; unlike reactive esters that may require cold chain shipping or special stabilizers, propargyl alcohol is robust and easy to transport. This shift to more common, stable raw materials reduces the risk of production stoppages due to material shortages. For a reliable pharmaceutical intermediate supplier, this translates to a more predictable delivery schedule and the ability to maintain strategic inventory levels without the fear of rapid degradation or supply disruption.
- Scalability and Environmental Compliance: Scaling a chemical process from the laboratory to multi-ton production often reveals hidden safety and environmental challenges. The legacy use of Oxone posed significant risks due to its potential for exothermic runaway reactions and the generation of sulfate-heavy waste streams. The transition to TEMPO-catalyzed oxidation mitigates these safety hazards, making the process inherently safer for operators and equipment. From an environmental standpoint, the reduction in step count and the use of greener oxidation chemistry align with modern sustainability goals and regulatory requirements. The simplified waste profile facilitates easier treatment and disposal, reducing the environmental compliance burden on the manufacturing site. This operational robustness ensures that the commercial scale-up of complex pharmaceutical intermediates can be achieved smoothly, meeting the stringent quality and safety standards required by global regulatory bodies without costly retrofitting or process re-validation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthetic route. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing clarity on the operational benefits and chemical rationale behind the process changes. Understanding these nuances is critical for technical teams evaluating the feasibility of adopting this new methodology for their specific production needs.
Q: Why is propargyl alcohol preferred over methyl propiolate in this synthesis?
A: Propargyl alcohol is significantly more cost-effective and stable than methyl propiolate, which is prone to self-polymerization under basic conditions, leading to lower yields and higher purification costs in conventional routes.
Q: How does the new oxidation method improve process safety?
A: The patent utilizes TEMPO-catalyzed oxidation with reagents like NBS or iodobenzene diacetate, avoiding the use of Oxone (potassium peroxymonosulfate), which presents significant safety hazards and handling difficulties during large-scale industrial amplification.
Q: What are the purity advantages of the one-pot cyclization step?
A: The one-pot removal of the Boc protecting group followed immediately by lactam ring closure minimizes intermediate isolation steps, thereby reducing the accumulation of impurities and simplifying the overall downstream processing workflow.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trilaciclib Intermediate Supplier
The technical innovations detailed in patent CN115536663A underscore the continuous evolution of chemical manufacturing towards greater efficiency and safety. At NINGBO INNO PHARMCHEM, we recognize the strategic importance of securing a stable supply of high-value oncology intermediates. As a dedicated CDMO partner, we possess the technical expertise to translate these patented laboratory methods into robust, GMP-compliant manufacturing processes. Our facilities are equipped to handle complex heterocyclic chemistry, including sensitive palladium-catalyzed couplings and controlled oxidation reactions. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our commitment to quality is backed by stringent purity specifications and rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch against the highest industry standards.
We invite you to collaborate with us to leverage this optimized synthetic route for your Trilaciclib supply chain. Our team is prepared to conduct a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how the adoption of this propargyl alcohol-based methodology can improve your bottom line. We encourage you to contact our technical procurement team to request specific COA data, route feasibility assessments, and preliminary quotations. By partnering with NINGBO INNO PHARMCHEM, you gain access to a supply chain that is not only cost-effective but also resilient, scalable, and fully aligned with the future of pharmaceutical manufacturing.
