Advanced Synthesis of 2-Chloronicotinic Acid: Technical Upgrades for Commercial Scale Production
Introduction to Novel Synthetic Pathways for Critical Intermediates
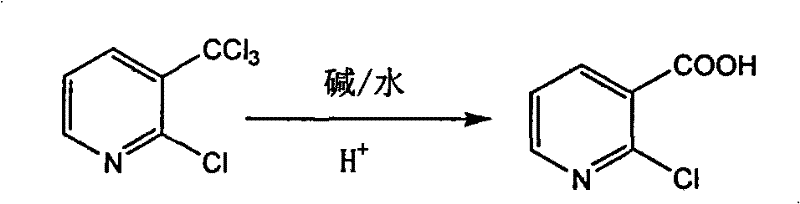
The global demand for high-purity heterocyclic carboxylic acids, specifically 2-chloronicotinic acid, has surged due to its pivotal role as a building block for next-generation herbicides like nicosulfuron and pharmaceutical agents such as niflumic acid and nevirapine. Addressing the critical need for efficient manufacturing, patent CN101602713B discloses a groundbreaking methodology that transforms 2-chloro-3-trichloromethylpyridine into 2-chloronicotinic acid through a streamlined alkaline hydrolysis process. This technical breakthrough represents a paradigm shift from traditional oxidative methods, offering a robust solution that aligns with modern green chemistry principles while delivering exceptional yields of approximately 93 percent. For R&D directors and procurement strategists, this patent provides a validated roadmap for securing a reliable agrochemical intermediate supplier capable of meeting stringent quality specifications without the environmental burden associated with legacy technologies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of 2-chloronicotinic acid has been plagued by significant technical and economic inefficiencies inherent in older oxidative protocols. The nitration mixture oxidation style, for instance, necessitates extreme reaction conditions ranging from 190°C to 210°C, imposing severe demands on reactor metallurgy and energy consumption while posing substantial safety risks. Alternatively, the potassium permanganate oxidation method, while operationally simpler, suffers from a dismal yield of roughly 65 percent and generates massive quantities of manganese-containing wastewater, creating a heavy environmental compliance burden. Furthermore, acid hydrolysis routes utilizing concentrated phosphoric acid, as seen in patents like US4504665, require excessive amounts of corrosive acids and still fail to match the efficiency of newer catalytic systems, resulting in higher operational expenditures and complex waste treatment logistics that erode profit margins.
The Novel Approach
In stark contrast to these cumbersome legacy processes, the novel approach detailed in the patent leverages the direct alkaline hydrolysis of the trichloromethyl group, a transformation that is both atom-economical and operationally elegant. By utilizing 2-chloro-3-trichloromethylpyridine as the starting substrate, the process bypasses the need for hazardous oxidants entirely, relying instead on inexpensive and readily available bases such as sodium hydroxide or potassium carbonate in an aqueous medium. This methodological shift not only simplifies the reaction engineering by allowing operations at moderate temperatures between 80°C and 100°C but also drastically reduces the complexity of downstream processing. The visual representation of this transformation highlights the direct conversion of the CCl3 moiety to the carboxylic acid function, underscoring the chemical elegance that drives the commercial viability of this route for large-scale manufacturing.
Mechanistic Insights into Alkaline Hydrolysis of Trichloromethyl Pyridines
The core chemical mechanism driving this synthesis involves the nucleophilic attack of hydroxide ions on the electron-deficient carbon of the trichloromethyl group attached to the pyridine ring. Under the specified thermal conditions of 80°C to 100°C, the chlorine atoms are sequentially displaced or hydrolyzed, eventually forming a carboxylate salt intermediate which remains soluble in the alkaline aqueous phase. This step is critical because it prevents the formation of insoluble byproducts that often complicate filtration in acidic media, ensuring a homogeneous reaction environment that maximizes contact between the reagents. The stoichiometry is carefully balanced with a molar ratio of substrate to base ranging from 1:1.5 to 1:10, ensuring that sufficient hydroxide is present to drive the equilibrium towards complete conversion without inducing unwanted side reactions on the sensitive chloro-substituent at the 2-position of the pyridine ring.
Impurity control is inherently managed through the selectivity of the alkaline medium, which favors the hydrolysis of the aliphatic trichloromethyl group over the aromatic chlorine atom. In traditional acidic hydrolysis or oxidative conditions, there is a heightened risk of dehalogenation or ring degradation, which can lead to difficult-to-remove impurities that compromise the purity profile required for pharmaceutical applications. By maintaining the reaction in a basic environment followed by a controlled acidification to a pH value of less than or equal to 1, the process ensures that the final precipitation of 2-chloronicotinic acid occurs with high specificity. This precise pH control allows for the crystallization of the target molecule while leaving most inorganic salts and minor organic impurities in the mother liquor, thereby achieving the reported purity levels of over 95 percent directly from the filtration step.
How to Synthesize 2-Chloronicotinic Acid Efficiently
To implement this high-yielding synthesis in a pilot or production setting, operators must adhere to strict parameter controls regarding temperature, stoichiometry, and acidification rates. The process begins with the preparation of an alkaline aqueous solution where the mass ratio of the trichloromethyl precursor to water is maintained between 1:5 and 1:20 to ensure adequate solubility and heat transfer. Following the heating and reaction period of 4 to 6 hours, the cooling phase is crucial for preparing the solution for acidification, which triggers the precipitation of the final product. For a comprehensive understanding of the exact operational parameters and safety protocols required for execution, please refer to the standardized synthesis steps outlined below.
- Prepare the reaction mixture by combining 2-chloro-3-trichloromethylpyridine with an alkaline solution (such as sodium hydroxide or potassium carbonate) and water in a mass ratio of 1: 5 to 1:20.
- Heat the mixture to a temperature between 80°C and 100°C and maintain stirring for 4 to 6 hours to ensure complete hydrolysis of the trichloromethyl group.
- Cool the reaction solution to room temperature, adjust the pH to ≤1 using hydrochloric acid to precipitate the product, then filter, wash, and dry to obtain the white solid powder.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this alkaline hydrolysis technology offers profound advantages in terms of cost structure and supply chain resilience. By eliminating the dependency on expensive and volatile oxidizing agents like potassium permanganate, manufacturers can significantly stabilize their raw material costs and reduce exposure to market fluctuations associated with specialty chemicals. Furthermore, the ability to utilize 2-chloro-3-trichloromethylpyridine, which is often a byproduct of other major agrochemical syntheses, creates a synergistic supply loop that enhances overall resource efficiency. This integration not only lowers the effective cost of goods sold but also mitigates supply risks by diversifying the sources of key starting materials, ensuring a more reliable agrochemical intermediate supplier network for downstream customers.
- Cost Reduction in Manufacturing: The elimination of costly oxidants and the reduction in energy requirements due to lower operating temperatures directly translate to substantial cost savings in 2-chloronicotinic acid manufacturing. Unlike the nitric acid oxidation method which requires specialized high-temperature equipment and significant energy input, this process operates near the boiling point of water, allowing for the use of standard glass-lined or stainless steel reactors. Additionally, the high yield of approximately 93 percent means that less raw material is wasted per unit of output, effectively lowering the material cost basis and improving the overall gross margin for producers who adopt this technology.
- Enhanced Supply Chain Reliability: The simplicity of the reagent profile, relying on commodity chemicals like caustic soda and hydrochloric acid, ensures that production is not bottlenecked by the availability of niche catalysts or reagents. This universality of inputs means that production can be sustained even during periods of supply chain disruption for specialty chemicals, providing a robust continuity of supply for clients. Moreover, the process's compatibility with existing infrastructure for handling aqueous alkaline streams facilitates easier technology transfer and scale-up, reducing the lead time for establishing new production lines.
- Scalability and Environmental Compliance: The absence of heavy metal waste streams, such as manganese sludge from permanganate oxidation, drastically simplifies wastewater treatment and reduces the environmental footprint of the facility. This alignment with green chemistry principles not only lowers the cost of waste disposal but also future-proofs the manufacturing site against increasingly stringent environmental regulations. The scalability of the process is further enhanced by the fact that the reaction is heterogeneous only during the precipitation phase, allowing for efficient mixing and heat management during the critical hydrolysis stage, making it ideal for commercial scale-up of complex agrochemical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route, derived directly from the patent specifications and experimental data. These insights are intended to clarify the operational feasibility and quality outcomes associated with this novel method for stakeholders evaluating potential partnerships. Understanding these details is essential for making informed decisions about integrating this technology into your existing supply chain.
Q: What are the primary advantages of this alkaline hydrolysis method over traditional oxidation routes?
A: Unlike traditional methods using potassium permanganate which generate heavy metal waste and have lower yields (around 65%), or nitric acid oxidation which requires harsh temperatures (190-210°C), this alkaline hydrolysis method operates under mild conditions (80-100°C) with significantly higher yields (up to 93%) and eliminates the need for expensive oxidants.
Q: How does this process impact the purity profile of the final 2-chloronicotinic acid?
A: The process avoids the use of strong oxidizing agents that often lead to complex impurity profiles. By utilizing a controlled alkaline hydrolysis followed by precise acidification to pH ≤1, the method consistently achieves product purity levels exceeding 95%, with specific embodiments reaching up to 97.9% without requiring extensive downstream purification.
Q: Is the raw material 2-chloro-3-trichloromethylpyridine readily available for large-scale production?
A: Yes, the starting material is often available as a byproduct from the synthesis of other agrochemicals like Provado (imidacloprid intermediates). Utilizing this feedstock not only ensures a stable supply chain but also adds value to existing chemical processes by converting a potential waste stream into a high-value pharmaceutical and agrochemical intermediate.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Chloronicotinic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of consistent quality and scalable production capabilities in the fine chemical sector. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volumetric demands of global agrochemical and pharmaceutical companies. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of 2-chloronicotinic acid meets the highest international standards, providing our partners with the confidence needed for their own regulatory filings and product launches.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can be tailored to your specific volume requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits of switching to this greener, more efficient process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our capability as a premier partner in the global supply of high-value heterocyclic intermediates.
