Revolutionizing Analgin Production: A Deep Dive into Palladium-Carbon Catalytic Hydrogenation Technology
Introduction to Advanced Analgin Synthesis Technology
The pharmaceutical industry is constantly seeking more efficient, sustainable, and cost-effective pathways for the production of essential analgesic and antipyretic agents. Patent CN102659680B introduces a groundbreaking methodology for the synthesis of Analgin (Sulpyrine), a drug with a century-long history of clinical efficacy. This patent delineates a novel operational path that fundamentally restructures the manufacturing landscape by condensing the traditional four-step synthesis into a highly efficient two-step process. By leveraging palladium-carbon catalytic hydrogenation, this technology addresses critical pain points associated with legacy production methods, including excessive energy consumption, complex purification protocols, and the generation of hazardous industrial waste. For stakeholders in the global supply chain, this innovation represents a pivotal shift towards greener chemistry and enhanced economic viability in the production of high-volume pharmaceutical intermediates.
The significance of this technological advancement cannot be overstated when considering the massive global demand for non-steroidal anti-inflammatory drugs. Traditional synthesis routes have long been burdened by low overall yields and reliance on toxic alkylating agents. The disclosed method not only streamlines the reaction sequence but also dramatically improves the purity profile of the final active pharmaceutical ingredient. By transitioning from stoichiometric chemical transformations to catalytic processes, manufacturers can achieve substantial reductions in raw material costs and waste treatment expenses. This report provides a comprehensive technical and commercial analysis of this patented route, offering strategic insights for R&D directors, procurement managers, and supply chain leaders aiming to optimize their analgesic drug portfolios.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
The historical standard for synthesizing Sulpyrine involves a cumbersome four-stage reaction sequence starting from 4-aminoantipyrine (4-AA). As illustrated in the traditional pathway, the process necessitates formylation, followed by methylation using dimethyl sulfate, hydrolysis, and finally condensation. This multi-step approach is inherently inefficient, with each stage introducing opportunities for yield loss and impurity formation. The methylation step, in particular, relies on dimethyl sulfate, a potent carcinogen and alkylating agent that poses severe safety risks to operators and requires stringent containment measures. Furthermore, the subsequent hydrolysis and neutralization steps consume large quantities of sulfuric acid and ammonia, generating massive amounts of inorganic salt byproducts that complicate wastewater treatment and increase the environmental footprint of the facility.
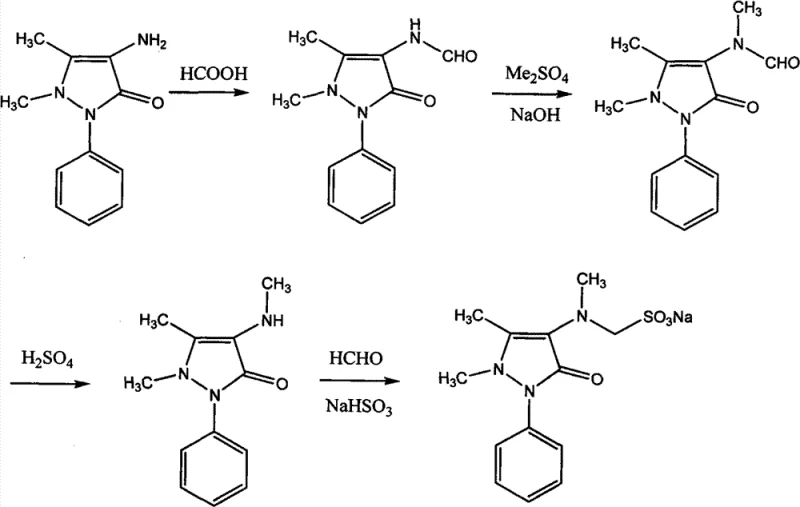
Beyond the safety and environmental concerns, the conventional method suffers from poor atom economy and extended production cycles. The cumulative yield of the traditional four-step process hovers around 76.3%, meaning nearly a quarter of the valuable starting material is lost to side reactions or mechanical losses during isolation. Each reaction step requires distinct work-up procedures, including cooling, crystallization, filtration, and drying, which significantly prolongs the manufacturing lead time. The reliance on harsh acidic and basic conditions also degrades equipment faster, leading to higher maintenance costs and potential downtime. For a commodity chemical produced in massive volumes, these inefficiencies translate into substantial hidden costs that erode profit margins and limit the scalability of production capacity.
The Novel Approach
In stark contrast to the legacy methodology, the patented innovation simplifies the synthesis of Sulpyrine into a concise two-step sequence that bypasses the need for toxic methylating agents and harsh hydrolysis conditions. The new route begins with a direct condensation reaction where 4-aminoantipyrine reacts with paraformaldehyde and sodium bisulfite to selectively produce 4-N-demethylated Sulpyrine. This intermediate is then subjected to a palladium-carbon catalytic hydrogenation reaction with formaldehyde to yield the final Analgin product. This streamlined approach eliminates the formylation and hydrolysis steps entirely, reducing the number of unit operations and the associated energy consumption. The shift to catalytic hydrogenation not only accelerates the reaction kinetics but also ensures a cleaner reaction profile with fewer byproducts.
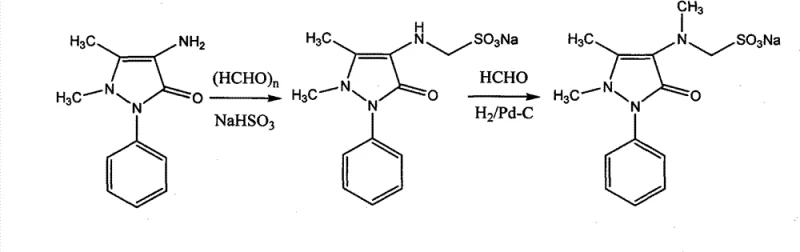
The operational advantages of this novel approach are manifold, offering a robust solution for modern pharmaceutical manufacturing. By removing the requirement for dimethyl sulfate, the process inherently becomes safer and more environmentally compliant, aligning with increasingly strict global regulations on hazardous chemical usage. The elimination of inorganic salt generation during the methylation phase simplifies the downstream purification process, allowing for straightforward filtration and crystallization of the final product. Moreover, the overall yield of the new process has been reported to increase significantly, with the final hydrogenation step alone achieving yields as high as 98%. This dramatic improvement in efficiency directly correlates to reduced raw material costs and a lower cost of goods sold, making it an attractive option for large-scale commercial production.
Mechanistic Insights into Pd-C Catalytic Hydrogenation and Selectivity
The core of this technological breakthrough lies in the precise control of reaction selectivity and the utilization of heterogeneous catalysis. In the first step, the choice of paraformaldehyde over aqueous formalin is critical. Experimental data within the patent reveals that using standard formalin leads to non-selective reactions, resulting in the formation of di-substituted byproducts (Structure III) which constitute about 20% of the product mixture and are notoriously difficult to separate. By employing paraformaldehyde, the reaction selectively targets the formation of the mono-substituted 4-N-demethyl Sulpyrine (Structure II) with a purity exceeding 96%. This selectivity is paramount for ensuring the quality of the intermediate fed into the subsequent hydrogenation step, preventing the carryover of impurities that could poison the catalyst or contaminate the final API.
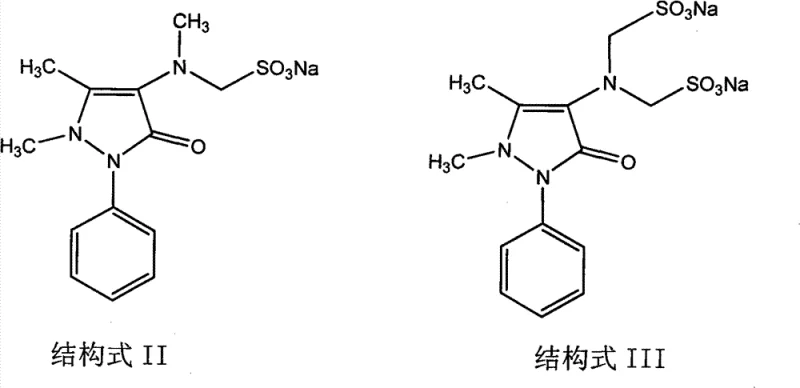
The second step involves a sophisticated palladium-carbon catalytic hydrogenation mechanism that facilitates the N-methylation of the intermediate under mild conditions. A key mechanistic challenge addressed in this patent is the stability of the sulfonic acid methyl group, which is prone to hydrolysis under alkaline or acidic conditions. To mitigate this, the process employs a specific solvent system comprising 92% to 95% ethanol and utilizes a phosphate buffer solution maintained at pH 6.86. This neutral environment is essential; if the ethanol content drops below 92%, hydrolysis increases, while content above 95% hinders reaction completion. The buffer stabilizes the pH, preventing the degradation of the intermediate into 4-AA or 4-novalgin. This delicate balance of solvent composition and pH control ensures that the catalytic hydrogenation proceeds efficiently to form the target Structure I (Sulpyrine) without compromising the integrity of the sulfonate moiety.
How to Synthesize Sulpyrine Efficiently
Implementing this advanced synthesis route requires adherence to specific operational parameters to maximize yield and purity. The process is designed to be scalable, moving seamlessly from laboratory validation to industrial production. The initial condensation step is conducted under reflux in ethanol, followed by a controlled cooling crystallization to isolate the intermediate. The subsequent hydrogenation step requires a pressurized reactor capable of maintaining hydrogen pressures between 0.1 and 0.5 MPa at temperatures ranging from 40 to 85°C. Strict control over the addition of the phosphate buffer and the concentration of the ethanol solvent is mandatory to prevent side reactions. For detailed standard operating procedures and specific stoichiometric ratios, please refer to the technical guide below.
- Condense 4-Aminoantipyrine with paraformaldehyde and sodium bisulfite in ethanol to selectively form 4-N-demethyl Sulpyrine.
- Perform catalytic hydrogenation on the intermediate using formaldehyde and Pd-C catalyst in a buffered ethanol solution.
- Isolate the final Sulpyrine product through cooling crystallization and filtration, achieving high purity without extensive salt removal.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis method offers compelling economic and logistical benefits. The primary advantage stems from the drastic simplification of the manufacturing process, which directly translates to reduced operational expenditures. By collapsing four reaction stages into two, facilities can significantly lower their utility consumption, labor requirements, and equipment occupancy time. The elimination of hazardous reagents like dimethyl sulfate not only reduces the cost of specialized handling and disposal but also mitigates the risk of regulatory fines and supply disruptions associated with controlled substances. This streamlined workflow enhances the overall agility of the supply chain, allowing for faster response times to market demand fluctuations.
- Cost Reduction in Manufacturing: The transition to a catalytic hydrogenation process eliminates the need for expensive and toxic alkylating agents, replacing them with readily available formaldehyde and hydrogen gas. This substitution results in substantial raw material cost savings. Furthermore, the high yield of the final step minimizes waste of the valuable intermediate, optimizing the overall material balance. The removal of inorganic salt byproducts means that wastewater treatment costs are significantly lowered, as there is no need for complex desalination or neutralization processes. These cumulative efficiencies drive down the total cost of production, providing a competitive pricing advantage in the global marketplace.
- Enhanced Supply Chain Reliability: Relying on a shorter synthesis route reduces the dependency on a long chain of precursors and reagents, thereby minimizing the risk of bottlenecks. The use of robust heterogeneous catalysts like palladium-on-carbon ensures consistent reaction performance and facilitates easy catalyst recovery and reuse. This reliability is crucial for maintaining continuous production schedules and meeting strict delivery deadlines for downstream pharmaceutical customers. Additionally, the improved purity profile of the crude product reduces the burden on quality control laboratories and shortens the release time for finished batches, ensuring a steady flow of high-quality material to the market.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing standard hydrogenation equipment found in most fine chemical plants. The reduction in "three wastes" (wastewater, waste gas, and solid waste) aligns with modern sustainability goals and environmental regulations. By avoiding the generation of large volumes of acidic and saline effluent, manufacturers can operate with a smaller environmental footprint and lower compliance costs. This green chemistry approach not only future-proofs the production facility against tightening environmental laws but also enhances the brand reputation of the supplier as a responsible and sustainable partner in the pharmaceutical value chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this palladium-carbon catalytic hydrogenation technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation. Understanding these nuances is essential for evaluating the feasibility of adopting this route for commercial manufacturing.
Q: Why is paraformaldehyde preferred over formalin in the first step of this synthesis?
A: Using formalin leads to non-selective reactions producing approximately 20% di-substituted impurities which are difficult to remove. Paraformaldehyde ensures high selectivity (>96% purity) for the desired 4-N-demethyl Sulpyrine intermediate.
Q: How does the new catalytic method improve environmental compliance compared to traditional routes?
A: The traditional route utilizes dimethyl sulfate and sulfuric acid, generating significant inorganic salt waste and acidic effluent. The new Pd-C catalytic hydrogenation method operates under neutral conditions, eliminating heavy metal catalysts and reducing three-waste discharge significantly.
Q: What are the critical process parameters to prevent hydrolysis during the hydrogenation step?
A: To prevent the hydrolysis of the sulfonic acid methyl group, the ethanol solvent content must be strictly controlled between 92% and 95%, and a phosphate buffer solution at pH 6.86 should be added to maintain neutrality during the reaction.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Analgin Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the Pd-C catalytic hydrogenation route for Analgin production. As a leading CDMO and supplier in the fine chemical sector, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped with advanced hydrogenation reactors and rigorous QC labs capable of meeting stringent purity specifications required by global pharmacopeias. We are committed to delivering high-purity Sulpyrine and its intermediates, leveraging our technical expertise to ensure consistent quality and supply continuity for our partners.
We invite pharmaceutical companies and chemical distributors to collaborate with us to explore the commercial viability of this innovative synthesis method. Our technical team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific production needs, demonstrating how this technology can optimize your supply chain. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and volume pricing options. Let us partner with you to drive efficiency and excellence in the production of essential analgesic medications.
