Scalable Synthesis of Cyclopropyl Chiral Amines for High-Value Pharmaceutical Intermediates
Scalable Synthesis of Cyclopropyl Chiral Amines for High-Value Pharmaceutical Intermediates
The development of efficient synthetic routes for chiral building blocks is a critical bottleneck in modern drug discovery, particularly for complex scaffolds containing cyclopropyl motifs. Patent CN112552184B discloses a robust and economically viable method for synthesizing cyclopropyl-containing chiral amine hydrochlorides, which serve as pivotal intermediates for CRF1 receptor antagonists, quinazoline-based anticancer agents, and Cathepsin S inhibitors. This technology represents a significant paradigm shift from legacy multi-step processes, offering a streamlined four-step sequence that begins with the inexpensive commodity chemical cyclopropanecarboxaldehyde. By leveraging a chiral pool strategy using (R)-1-phenylethylamine, the process achieves exceptional stereocontrol without relying on prohibitively expensive chiral catalysts or hazardous reducing agents. For R&D directors and procurement specialists alike, this innovation addresses the dual challenges of maintaining high optical purity while ensuring the economic feasibility required for commercial-scale API production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of cyclopropyl chiral amines has been plagued by inefficient methodologies that hinder industrial scalability. One prominent prior art route, illustrated in the literature, relies on N-methoxy-N-methyl-2-methoxyacetamide (Weinreb amide) as a starting material. As depicted in 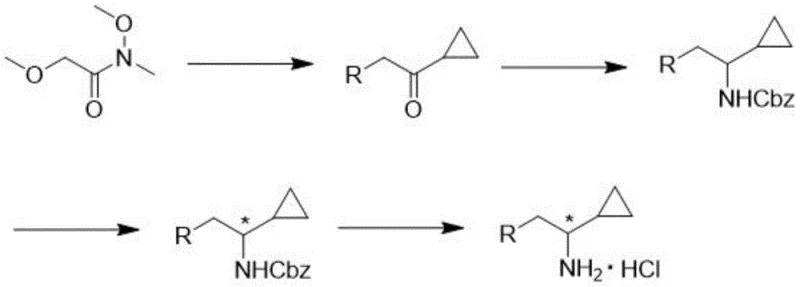 , this approach necessitates a cumbersome five-step sequence involving cyclopropanation, reductive amination, Cbz protection, chiral resolution, and deprotection. The reliance on sodium triacetoxyborohydride for reductive amination introduces significant safety hazards due to its explosive potential upon exposure to moisture or heat, making it unsuitable for multi-ton manufacturing. Furthermore, the chiral resolution in this legacy route often requires preparative HPLC on chiral columns, a technique that is technically feasible only on a gram scale and economically disastrous for kilogram or ton-scale production due to the high cost of stationary phases and solvents.
, this approach necessitates a cumbersome five-step sequence involving cyclopropanation, reductive amination, Cbz protection, chiral resolution, and deprotection. The reliance on sodium triacetoxyborohydride for reductive amination introduces significant safety hazards due to its explosive potential upon exposure to moisture or heat, making it unsuitable for multi-ton manufacturing. Furthermore, the chiral resolution in this legacy route often requires preparative HPLC on chiral columns, a technique that is technically feasible only on a gram scale and economically disastrous for kilogram or ton-scale production due to the high cost of stationary phases and solvents.
The Novel Approach
In stark contrast, the novel methodology disclosed in the patent utilizes a convergent strategy that dramatically simplifies the molecular assembly. Instead of building the carbon skeleton from scratch with expensive precursors, the new route employs cyclopropanecarboxaldehyde as a foundational building block. As shown in 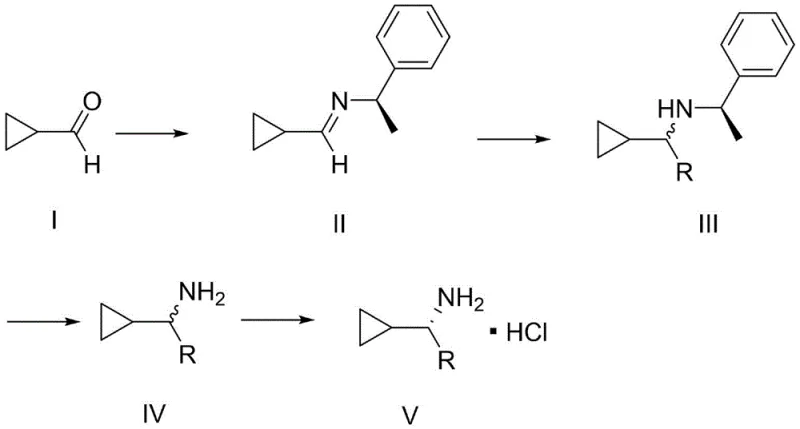 , the process initiates with a condensation reaction to form a chiral imine, followed by a diastereoselective Grignard addition. This approach bypasses the need for dangerous borohydride reductions entirely. Another competing method found in literature utilizes chiral tert-butyl sulfinamide; however, as seen in
, the process initiates with a condensation reaction to form a chiral imine, followed by a diastereoselective Grignard addition. This approach bypasses the need for dangerous borohydride reductions entirely. Another competing method found in literature utilizes chiral tert-butyl sulfinamide; however, as seen in 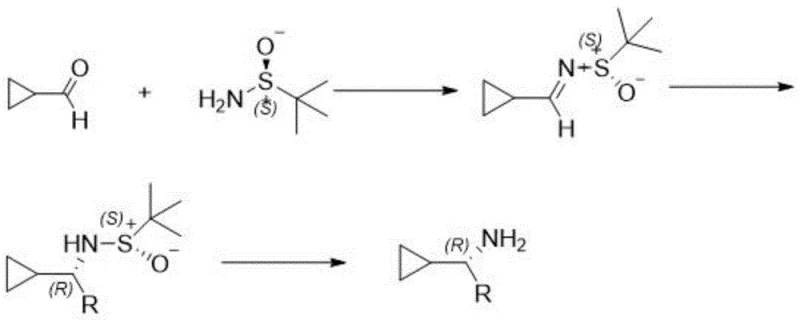 , this reagent is not only costly but also generates malodorous byproducts and typically requires column chromatography for purification. The new invention replaces these problematic steps with a catalytic hydrogenation and a crystallization-based resolution, creating a pathway that is inherently safer, cleaner, and far more amenable to continuous processing and large-batch manufacturing.
, this reagent is not only costly but also generates malodorous byproducts and typically requires column chromatography for purification. The new invention replaces these problematic steps with a catalytic hydrogenation and a crystallization-based resolution, creating a pathway that is inherently safer, cleaner, and far more amenable to continuous processing and large-batch manufacturing.
Mechanistic Insights into Diastereoselective Grignard Addition and Resolution
The core chemical innovation lies in the precise control of stereochemistry during the carbon-carbon bond-forming event. The reaction begins with the condensation of cyclopropanecarboxaldehyde and (R)-1-phenylethylamine in a solvent such as toluene at elevated temperatures (80-110°C). This forms a chiral imine intermediate (Compound II) which acts as an electrophile with a defined chiral environment. When a Grignard reagent (RMgX) is introduced at controlled low temperatures (-50 to 100°C), the nucleophilic attack occurs preferentially from one face of the imine double bond, dictated by the steric bulk of the phenylethyl group. This diastereoselective addition establishes the new chiral center at the alpha-position relative to the cyclopropyl ring with high fidelity. The resulting amine (Compound III) contains two chiral centers: the original one from the auxiliary and the newly formed one. This diastereomeric relationship is crucial because diastereomers possess different physical properties, allowing for their separation later in the process.
Following the alkylation, the chiral auxiliary is removed via catalytic hydrogenolysis using palladium on carbon (Pd/C) under mild hydrogen pressure (0.5-1.0 MPa). This step cleaves the benzyl C-N bond, releasing the free chiral amine (Compound IV) and regenerating acetophenone, which can potentially be recycled. The final and perhaps most critical step for ensuring pharmaceutical-grade quality is the chiral resolution. The racemic or enriched amine is treated with a resolving agent such as (S)-mandelic acid or L-dibenzoyl tartaric acid in a protic solvent like isopropanol. Through the formation of diastereomeric salts, the desired enantiomer crystallizes out of the solution while the undesired enantiomer remains in the mother liquor. This crystallization-driven purification is vastly superior to chromatographic methods, as it allows for the removal of trace impurities and the enrichment of optical purity to >99% ee simply through filtration and washing, ensuring the final hydrochloride salt meets the stringent specifications required for clinical applications.
How to Synthesize Cyclopropyl Chiral Amine Efficiently
The operational simplicity of this synthesis makes it an ideal candidate for technology transfer from the laboratory to the pilot plant. The process is designed to minimize unit operations; for instance, the crude imine solution from the first step can often be telescoped directly into the Grignard reaction without isolation, reducing solvent waste and handling time. Similarly, the hydrogenation filtrate can be concentrated and subjected directly to resolution. This "telescoping" capability is a hallmark of modern green chemistry, reducing the overall environmental footprint and equipment occupancy time. For detailed standard operating procedures regarding temperature ramps, quenching protocols, and specific work-up sequences, please refer to the technical guide below.
- Condense cyclopropanecarboxaldehyde with (R)-1-phenylethylamine in toluene at 80-110°C to form the chiral imine intermediate.
- Perform stereoselective alkylation by reacting the imine with a specific Grignard reagent at low temperatures (-50 to 100°C).
- Execute catalytic hydrogenation using Pd/C to remove the benzyl protecting group, followed by chiral resolution using mandelic acid derivatives.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the transition to this novel synthetic route offers profound strategic benefits that extend beyond mere technical elegance. The primary driver for adoption is the substantial reduction in the cost of goods sold (COGS). By replacing exotic chiral reagents like sulfinamides with commodity chemicals like (R)-1-phenylethylamine and cyclopropanecarboxaldehyde, the raw material cost baseline is lowered significantly. Furthermore, the elimination of column chromatography—a unit operation that is notoriously solvent-intensive and slow—translates directly into faster batch cycle times and higher throughput per reactor volume. This efficiency gain allows suppliers to respond more agilely to fluctuating market demands without the need for massive capital expenditure on additional purification infrastructure.
- Cost Reduction in Manufacturing: The economic model of this process is strengthened by the avoidance of stoichiometric amounts of expensive chiral auxiliaries that are difficult to recover. In traditional sulfinamide routes, the auxiliary is often lost or degraded, representing a sunk cost in every batch. In this new method, the chiral amine auxiliary is cleaved and can theoretically be recovered and reused, further driving down long-term operational expenses. Additionally, the use of heterogeneous catalysts like Pd/C simplifies the removal of metal residues compared to homogeneous systems, reducing the cost associated with downstream metal scavenging and validation testing.
- Enhanced Supply Chain Reliability: Supply continuity is often jeopardized when a synthesis relies on niche reagents with limited global suppliers. The reagents specified in this patent, including various Grignard reagents and common resolving agents like mandelic acid, are produced by multiple chemical manufacturers worldwide. This diversification of the supply base mitigates the risk of shortages or price spikes caused by geopolitical events or single-source production failures. Moreover, the robustness of the reaction conditions—tolerating a range of temperatures and pressures—ensures that production is less susceptible to minor deviations in utility supplies, guaranteeing consistent delivery schedules for downstream API manufacturers.
- Scalability and Environmental Compliance: As regulatory scrutiny on pharmaceutical manufacturing intensifies, the environmental profile of a synthesis route becomes a key differentiator. This process minimizes the generation of hazardous waste by avoiding toxic borohydrides and reducing solvent consumption through telescoped steps. The ability to scale this chemistry from 100 kgs to 100 MT annual commercial production without fundamental changes to the reaction mechanism provides a clear path for capacity expansion. The simplified waste stream, largely consisting of benign organic salts and recoverable solvents, facilitates easier compliance with increasingly strict environmental regulations regarding effluent discharge and VOC emissions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, aiming to clarify the practical implications for potential licensees and manufacturing partners.
Q: What are the primary advantages of this new synthesis route over traditional sulfinamide methods?
A: The new route eliminates the need for expensive chiral tert-butyl sulfinamide and avoids hazardous reagents like sodium triacetoxyborohydride. It utilizes commercially available (R)-1-phenylethylamine as a chiral auxiliary, which is significantly cheaper and easier to recover, thereby drastically reducing raw material costs and improving process safety for large-scale manufacturing.
Q: How does this process ensure high optical purity without HPLC separation?
A: The process achieves high stereocontrol through a combination of diastereoselective Grignard addition directed by the chiral amine auxiliary and a final crystallization-based resolution step using resolving agents like (S)-mandelic acid. This avoids the need for preparative HPLC, which is notoriously difficult and costly to scale up industrially, ensuring ee values exceeding 99% through standard filtration and washing.
Q: Is this synthesis method suitable for producing diverse side-chain analogs?
A: Yes, the methodology is highly modular. By simply varying the Grignard reagent (e.g., ethyl, vinyl, isopropyl, or alkynyl magnesium halides), manufacturers can access a wide library of cyclopropyl chiral amine derivatives with different alkyl or alkenyl side chains, making it a versatile platform technology for medicinal chemistry optimization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cyclopropyl Chiral Amine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of novel therapeutics depends on the reliability and quality of the underlying supply chain. Our team of process chemists has extensively evaluated the technology disclosed in CN112552184B and confirmed its viability for large-scale production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition smoothly from clinical trials to market launch. Our facilities are equipped with state-of-the-art hydrogenation reactors and crystallization units specifically designed to handle the sensitive steps of this synthesis, guaranteeing stringent purity specifications and rigorous QC labs to verify every batch against your requirements.
We invite you to collaborate with us to leverage this cost-effective synthesis route for your next-generation pharmaceutical programs. Whether you require custom synthesis of specific analogs or bulk supply of the standard hydrochloride salt, our technical procurement team is ready to assist. Please contact us today to request a Customized Cost-Saving Analysis tailored to your specific volume needs. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how partnering with us can accelerate your development timeline and optimize your manufacturing budget.
