Scalable Synthesis of 1,4-Diazaspiro[5,5]undecan-3-one via Safe Catalytic Hydrogenation
Introduction to Advanced Spiroheterocyclic Synthesis
The pharmaceutical industry constantly seeks robust pathways for complex heterocyclic scaffolds, particularly those serving as critical fragments in oncology and sleep disorder therapeutics. Patent CN113754600A introduces a groundbreaking preparation method for 1,4-diazaspiro[5,5]undecan-3-one, a pivotal intermediate in the synthesis of Orexin antagonists and Trilaciclib. This novel approach fundamentally shifts the paradigm from hazardous, high-cost legacy chemistries to a streamlined, safety-first protocol utilizing catalytic hydrogenation. By leveraging commercially available cyclohexanone cyanohydrin and glycine esters, the process circumvents the severe safety liabilities associated with traditional cyanide sources and explosive nitro-compounds. The structural integrity and pharmacological potential of this spiroheterocyclic compound make it a high-value target for scalable manufacturing.
![Chemical structure of 1,4-diazaspiro[5,5]undecan-3-one showing the spiro center and ketone functionality](/insights/img/1-4-diazaspiro-synthesis-catalytic-hydrogenation-pharma-supplier-20260305062517-01.png)
For global procurement teams and R&D directors, the implications of this patent extend beyond mere chemical novelty; it represents a tangible opportunity for cost reduction in pharmaceutical intermediates manufacturing. The elimination of exotic reagents like titanium isopropoxide and trimethylsilyl cyanide (TMSCN) drastically lowers the raw material bill of materials (BOM). Furthermore, the transition to catalytic hydrogenation using earth-abundant metals like nickel aligns with modern green chemistry principles, reducing the environmental footprint and waste treatment costs. As a reliable pharmaceutical intermediate supplier, understanding these mechanistic advantages is crucial for securing a stable supply chain for next-generation API production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1,4-diazaspiro[5,5]undecan-3-one has been plagued by significant safety and economic hurdles that hinder large-scale industrial adoption. Prior art routes, such as those disclosed in WO2015161287A1, rely heavily on the Strecker reaction conditions requiring expensive TMSCN and titanium isopropoxide, followed by a dangerous reduction step using lithium aluminum hydride (LiAlH4). The use of LiAlH4 poses severe explosion risks during scale-up due to its pyrophoric nature and vigorous reaction with protic solvents, necessitating specialized equipment and rigorous safety protocols that inflate operational expenditures. Additionally, alternative pathways described in WO2020/041770A1 utilize nitromethane in Henry reactions, introducing flammable and explosive hazards that complicate regulatory compliance and facility insurance.
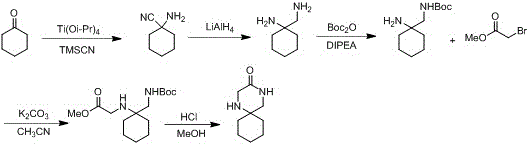
Further compounding these issues, other documented methods employ zinc powder for nitro reduction or bromoacetyl bromide for cyclization, both of which introduce distinct processing bottlenecks. Zinc powder generates substantial solid waste sludge that requires complex filtration and disposal procedures, while bromoacetyl bromide is a potent lachrymator and alkylating agent that demands extreme handling care. These legacy processes often suffer from poor selectivity, leading to the formation of isomeric impurities that are difficult to separate, thereby compromising the overall yield and purity of the final high-purity pharmaceutical intermediate. The cumulative effect of these drawbacks renders conventional methods economically unviable for multi-ton commercial production.
The Novel Approach
In stark contrast, the methodology outlined in CN113754600A offers a streamlined, two-step sequence that elegantly bypasses these historical pain points. The process initiates with a mild nucleophilic substitution between cyclohexanone cyanohydrin and glycine ester hydrochloride in an alcoholic solvent, facilitated by inexpensive organic bases like triethylamine. This step proceeds under ambient to mild thermal conditions, avoiding the cryogenic temperatures often required for sensitive organometallic reactions. The subsequent transformation involves a tandem catalytic hydrogenation and cyclization, where the nitrile group is reduced to a primary amine which immediately undergoes intramolecular amidation with the ester moiety. This telescoped operation not only simplifies the workflow but also utilizes benign catalysts such as Raney Nickel or Palladium on Carbon, which are easily filtered and recycled.
The strategic avoidance of hazardous reagents translates directly into enhanced process safety and operational simplicity. By eliminating the need for explosive nitromethane and toxic cyanide salts, the new route significantly lowers the barrier to entry for contract manufacturing organizations (CMOs) and reduces the regulatory burden associated with handling controlled substances. The mild reaction conditions (20-60°C) and atmospheric or low-pressure hydrogen requirements allow for the use of standard glass-lined or stainless steel reactors, facilitating the commercial scale-up of complex pharmaceutical intermediates. This approach ensures a consistent supply of high-quality material while minimizing the risk of batch failures due to thermal runaways or reagent instability.
Mechanistic Insights into Catalytic Reductive Cyclization
The core innovation of this synthesis lies in the efficient reductive cyclization mechanism that constructs the spiro-fused diazepine ring system. In the second step, the (1-cyanocyclohexyl) glycine ester undergoes catalytic hydrogenation where the cyano group (-CN) is sequentially reduced to an imine and then to a primary amine (-CH2NH2) on the catalyst surface. The presence of a base catalyst, such as DABCO or DBU, plays a critical role in deprotonating the newly formed amine, increasing its nucleophilicity. This activated amine then attacks the adjacent ester carbonyl carbon in an intramolecular nucleophilic acyl substitution. The resulting tetrahedral intermediate collapses, expelling the alkoxide leaving group (methoxide or ethoxide) to form the stable seven-membered lactam ring fused to the cyclohexane spiral center. This cascade reaction is thermodynamically driven by the formation of the stable amide bond and the release of volatile alcohol byproducts.
From an impurity control perspective, this mechanism offers superior selectivity compared to alkylation-based routes. Traditional methods using bromoacetyl bromide often suffer from over-alkylation or N-alkylation side reactions, generating regioisomers that share similar physical properties with the target molecule, making purification via crystallization or chromatography challenging and yield-destructive. In the hydrogenation route, the cyclization is intramolecular and kinetically favored due to the proximity of the reactive centers, inherently suppressing intermolecular polymerization or oligomerization side reactions. The use of heterogeneous catalysts like Raney Nickel further aids in purity by allowing for the removal of trace metal impurities through simple filtration, ensuring the final product meets the stringent specifications required for GMP-grade API intermediate production without extensive downstream processing.
How to Synthesize 1,4-Diazaspiro[5,5]undecan-3-one Efficiently
The practical execution of this synthesis requires careful attention to solvent selection and catalyst activation to maximize yield and throughput. The first step is typically conducted in methanol or ethanol, where the solubility of the glycine ester hydrochloride and the cyanohydrin is optimized. Maintaining a slightly alkaline environment is crucial to free the glycine ester from its salt form without triggering premature hydrolysis of the nitrile group. For the reduction step, the choice between Raney Nickel and Palladium Carbon depends on the specific pressure capabilities of the reactor; Raney Nickel is highly active at lower pressures (around 30 psi), making it ideal for cost-sensitive operations, while Pd/C may offer faster kinetics at slightly elevated pressures. Detailed standardized operating procedures regarding catalyst loading, hydrogen uptake monitoring, and workup pH adjustments are essential for reproducible results.
- React cyclohexanone cyanohydrin with glycine ester hydrochloride in an alcohol solvent under alkaline conditions to form (1-cyanocyclohexyl) glycine ester.
- Perform catalytic hydrogenation reduction on the nitrile group using Raney Nickel or Palladium Carbon in the presence of a base like DABCO.
- Facilitate spontaneous amine-ester exchange cyclization to yield the final spiroheterocyclic ketone product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this patented route offers compelling economic and logistical benefits that directly impact the bottom line. The primary advantage stems from the drastic simplification of the raw material portfolio; replacing exotic, import-dependent reagents like TMSCN and Titanium Isopropoxide with commodity chemicals like glycine ester and cyclohexanone cyanohydrin insulates the supply chain from geopolitical volatility and price spikes. These commodity feedstocks are produced by multiple vendors globally, ensuring a competitive bidding environment and preventing single-source bottlenecks. Furthermore, the elimination of hazardous waste streams associated with lithium aluminum hydride quenching and zinc sludge disposal significantly reduces environmental compliance costs and waste treatment fees, contributing to a leaner manufacturing cost structure.
- Cost Reduction in Manufacturing: The shift to catalytic hydrogenation eliminates the need for stoichiometric amounts of expensive reducing agents like LiAlH4, which require strict anhydrous conditions and generate substantial aluminum waste. Instead, the process utilizes catalytic quantities of reusable metals, driving down the variable cost per kilogram. Additionally, the avoidance of cryogenic cooling and specialized explosion-proof infrastructure for nitromethane handling reduces capital expenditure (CAPEX) and utility costs. The overall process efficiency is improved by the telescoped nature of the reduction-cyclization step, which reduces solvent consumption and labor hours associated with intermediate isolation, leading to substantial operational savings.
- Enhanced Supply Chain Reliability: By relying on widely available starting materials, manufacturers can mitigate the risk of supply disruptions that often plague specialty chemical markets. The robustness of the reaction conditions means that production can be easily transferred between different manufacturing sites without requiring highly specialized equipment or niche expertise. This flexibility ensures business continuity and allows for rapid scaling to meet fluctuating market demand for downstream oncology drugs. The simplified logistics of handling non-hazardous solids and liquids also streamline transportation and storage, reducing lead times and inventory holding costs.
- Scalability and Environmental Compliance: The process is inherently designed for scale, utilizing standard unit operations common in fine chemical plants. The absence of pyrophoric reagents and explosive intermediates simplifies the safety validation process (HAZOP studies), accelerating the timeline from pilot plant to commercial production. From a sustainability standpoint, the atom economy is superior to legacy routes, and the generation of non-toxic byproducts (alcohols and ammonia/amines) facilitates easier wastewater treatment. This alignment with green chemistry metrics enhances the corporate social responsibility (CSR) profile of the supply chain, appealing to end-clients who prioritize sustainable sourcing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. Understanding these details is vital for technical teams evaluating the feasibility of integrating this intermediate into their existing pipelines. The answers are derived directly from the experimental data and comparative analysis provided in the patent literature, ensuring accuracy and relevance for decision-makers.
Q: Why is this new synthesis route safer than previous methods?
A: This route eliminates the use of highly toxic sodium cyanide, expensive TMSCN, and explosive nitromethane, significantly reducing industrial safety risks.
Q: What catalysts are suitable for the reduction step?
A: The patent specifies Raney Nickel, Palladium Carbon, or Palladium Hydroxide, with Raney Nickel being preferred for cost-effectiveness and efficiency.
Q: How does this method improve impurity profiles?
A: By avoiding dangerous reagents like bromoacetyl bromide used in other routes, this method prevents the formation of difficult-to-remove isomers, ensuring higher purity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,4-Diazaspiro[5,5]undecan-3-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality spiroheterocyclic intermediates play in the development of life-saving medications. Our technical team has extensively analyzed the pathway described in CN113754600A and possesses the expertise to optimize this route for maximum efficiency and purity. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. Our facilities are equipped with state-of-the-art hydrogenation reactors and rigorous QC labs capable of meeting stringent purity specifications required by global regulatory bodies, guaranteeing a consistent and reliable supply of this key building block.
We invite you to collaborate with us to leverage these technological advancements for your drug development programs. Our team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this optimized route can reduce your overall COGS. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments. Let us partner with you to secure a sustainable and cost-effective supply chain for your next-generation pharmaceutical products.
