Advanced Synthesis of Levorgyration Demethyl Phencynonate for Commercial API Manufacturing
The pharmaceutical industry is constantly seeking robust, scalable, and environmentally compliant pathways for complex active pharmaceutical ingredient (API) intermediates. Patent CN102190619B introduces a groundbreaking synthesis method for Levorgyration Demethyl Phencynonate, a potent M4 receptor antagonist currently under preclinical investigation for Parkinson's disease treatment. This novel approach fundamentally restructures the production landscape by replacing hazardous reagents like diazomethane and zinc dust with safer catalytic hydrogenation and transesterification techniques. The strategic shift not only addresses critical safety concerns associated with explosive and toxic reagents but also delivers a substantial improvement in overall process yield, rising from roughly 40.4% to over 51.6%. For R&D directors and procurement managers, this represents a pivotal opportunity to secure a more reliable supply chain for high-value neurological drug candidates while adhering to stricter environmental regulations.
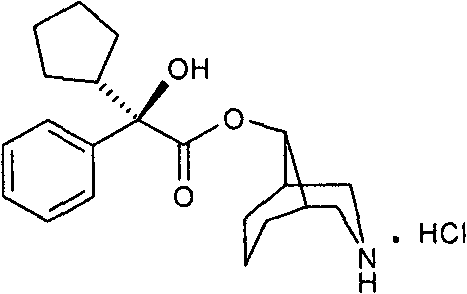
Understanding the limitations of conventional synthesis routes is essential for appreciating the technological leap offered by this patent. Traditional methods for producing Levorgyration Demethyl Phencynonate rely heavily on dangerous chemistry, specifically utilizing diazomethane for the esterification of alpha-hydroxy acids. Diazomethane is notorious for its volatility, explosiveness, and severe toxicity, posing significant risks to plant personnel and requiring specialized, costly containment infrastructure. Furthermore, the historical demethylation steps often employ ethyl chloroformate in benzene, another carcinogenic solvent that complicates waste disposal and regulatory approval. The final reduction stages typically involve zinc powder and acetic acid, which generate large volumes of zinc mud waste and introduce heavy metal residues that are difficult to purge from the final product, necessitating expensive purification protocols.
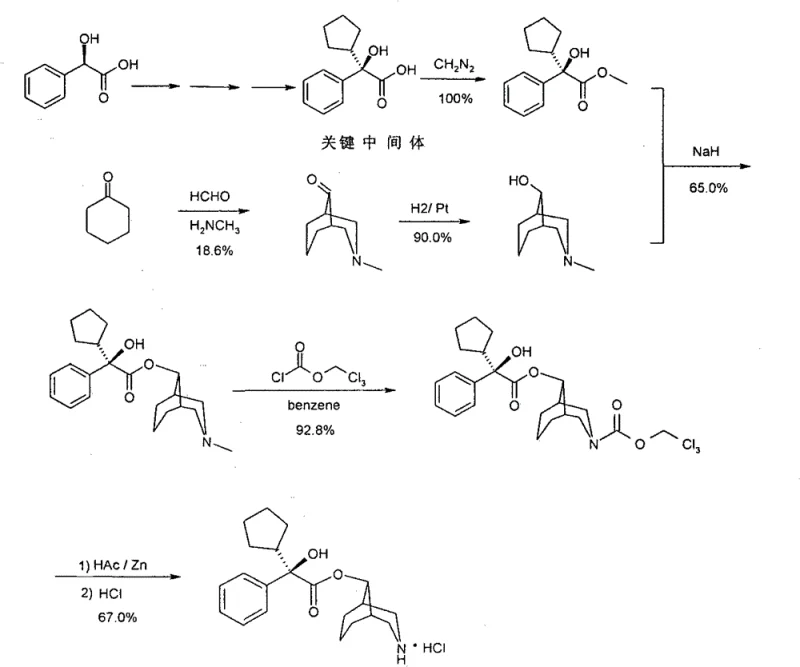
In stark contrast, the novel approach detailed in the patent utilizes a transesterification strategy followed by catalytic debenzylation. Instead of diazomethane, the process employs dimethyl sulfate for esterification, a reagent that, while requiring care, is significantly easier to handle and control on an industrial scale compared to gaseous diazomethane. The core coupling reaction involves reacting (R)-alpha-phenyl-alpha-cyclopentyl-hydroxyacetic acid methyl ester with N-benzyl-3-azabicyclo-[3.3.1]-nonan-9-ol in the presence of sodium hydride. This is followed by a clean catalytic hydrogenation step using palladium or platinum catalysts to remove the benzyl protecting group. This sequence completely bypasses the need for zinc reduction, eliminating zinc waste streams and ensuring a cleaner impurity profile. The result is a streamlined process that is not only safer but also more economically viable due to higher yields and reduced waste treatment costs.
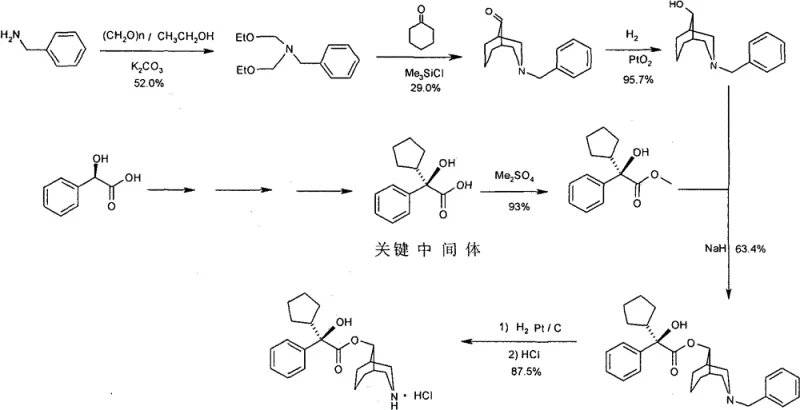
The mechanistic insights into this synthesis reveal a sophisticated understanding of stereoselectivity and functional group tolerance. The transesterification step is driven by the formation of an alkoxide intermediate using sodium hydride in non-aqueous solvents like toluene or heptane at elevated temperatures (130-135°C). This thermal condition facilitates the displacement of the methoxy group by the bulky bicyclic alcohol, driving the equilibrium towards the desired ester product through the distillation of methanol. Crucially, this method preserves the chiral integrity of the (R)-alpha-phenyl-alpha-cyclopentyl moiety, which is vital for the biological activity of the final M4 antagonist. The subsequent hydrogenation step is equally critical; by using a benzyl protecting group on the nitrogen atom of the bicyclic ring, the synthesis allows for a mild and selective deprotection. Catalytic hydrogenation cleaves the benzylic C-N bond without affecting the sensitive ester linkage or the cyclopentyl ring, a level of chemoselectivity that is difficult to achieve with harsher chemical reducing agents.
Impurity control is inherently built into this new mechanism, addressing a major pain point for quality assurance teams. In the old zinc-based reduction method, the formation of zinc acetate and colloidal zinc particles often led to filtration issues and potential heavy metal exceedances in the final API. The new catalytic hydrogenation route produces only toluene or ethanol as byproducts alongside the free amine, which are easily removed during workup. Furthermore, the avoidance of benzene as a solvent eliminates a Class 1 residual solvent concern, simplifying the solvent recovery and recycling process. The use of dimethyl sulfate instead of diazomethane also reduces the risk of forming unpredictable methylation byproducts on other nucleophilic sites within the molecule. This cleaner reaction profile translates directly to reduced downstream processing time and higher confidence in meeting stringent pharmacopeial standards for heavy metals and residual solvents.
How to Synthesize Levorgyration Demethyl Phencynonate Efficiently
The synthesis of this complex bicyclic ester requires precise control over reaction conditions to maximize yield and purity. The patented process outlines a clear, three-stage workflow that begins with the preparation of key intermediates and culminates in the final salt formation. The initial transesterification demands anhydrous conditions and careful temperature management to ensure complete conversion without degrading the sensitive alpha-hydroxy ester. Following this, the catalytic hydrogenation must be monitored to prevent over-reduction or catalyst poisoning. Detailed standard operating procedures for each step, including specific solvent ratios, catalyst loadings, and workup protocols, are essential for successful technology transfer. For a comprehensive guide on executing these steps with industrial precision, please refer to the standardized synthesis instructions provided below.
- Perform transesterification between (R)-alpha-phenyl-alpha-cyclopentyl-hydroxyacetic acid methyl ester and N-benzyl-3-azabicyclo-[3.3.1]-nonan-9-ol using sodium hydride in non-aqueous solvents like toluene.
- Execute catalytic hydrogenation on the resulting benzyl ester intermediate using Pd/C or Pt/C catalysts under controlled pressure to remove the benzyl protecting group efficiently.
- React the deprotected free base with hydrochloric acid in an appropriate solvent system to form the final stable hydrochloride salt of the target compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route offers tangible strategic benefits beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the supply chain for raw materials. By eliminating the need for diazomethane, which often requires on-site generation due to its instability, facilities can rely on stable, commercially available reagents like dimethyl sulfate and benzylamine derivatives. This shift reduces the logistical burden and the capital expenditure associated with specialized hazardous material handling equipment. Moreover, the removal of zinc dust and acetic acid from the final reduction step significantly lowers the volume of hazardous solid waste generated per kilogram of product. This reduction in waste volume directly correlates to lower disposal costs and a smaller environmental footprint, aligning with modern green chemistry initiatives and corporate sustainability goals.
- Cost Reduction in Manufacturing: The transition to this new pathway drives cost efficiency through multiple mechanisms, primarily by improving the overall mass balance of the process. The patent data indicates a yield increase from 40.4% to over 51.6%, which means that for every ton of starting material, significantly more product is obtained, effectively lowering the cost of goods sold (COGS). Additionally, the elimination of expensive and difficult-to-handle reagents like diazomethane reduces raw material costs and the overhead associated with their safe storage and usage. The simplified workup procedures, which no longer require extensive filtration of zinc mud or complex molecular distillation steps for intermediates, further reduce labor and utility consumption, leading to substantial operational savings.
- Enhanced Supply Chain Reliability: Relying on a synthesis route that avoids exotic or highly regulated reagents enhances the resilience of the supply chain. Diazomethane is a controlled substance in many jurisdictions due to its explosive nature, and its supply can be intermittent or restricted. By substituting it with standard industrial chemicals, the manufacturing process becomes less vulnerable to regulatory bottlenecks and supplier disruptions. Furthermore, the robustness of the catalytic hydrogenation step ensures consistent batch-to-batch quality, reducing the risk of production delays caused by failed quality control tests for heavy metals or residual solvents. This reliability is crucial for maintaining continuous supply to downstream API manufacturers and meeting strict delivery timelines.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety and environmental challenges, but this new method is inherently designed for scalability. The replacement of batch-wise hazardous reductions with continuous or semi-continuous catalytic hydrogenation allows for safer scale-up in larger reactors. The absence of benzene and zinc waste simplifies the permitting process for new manufacturing lines, as the effluent profile is much cleaner and easier to treat. This environmental compliance advantage accelerates the time-to-market for new drug candidates utilizing this intermediate, as regulatory filings will benefit from a greener and safer manufacturing description, reducing the likelihood of queries from health authorities regarding process safety and waste management.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on yield expectations, safety improvements, and intermediate availability. Understanding these details is vital for stakeholders evaluating the feasibility of adopting this route for commercial production.
Q: How does the new synthesis route improve safety compared to traditional methods?
A: The novel method eliminates the use of highly toxic and explosive diazomethane for esterification and avoids hazardous zinc dust reduction steps, significantly enhancing operational safety and environmental compliance.
Q: What is the expected yield improvement with this patented process?
A: According to patent data, the total yield calculated from key intermediates increases from approximately 40.4% in prior art to over 51.6% using this optimized transesterification and hydrogenation pathway.
Q: Does this process reduce heavy metal contamination risks?
A: Yes, by replacing zinc powder reduction with catalytic hydrogenation, the process avoids the generation of zinc mud and acetate residues, thereby minimizing heavy metal contamination risks in the final API.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Levorgyration Demethyl Phencynonate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and safe synthesis routes for high-value pharmaceutical intermediates like Levorgyration Demethyl Phencynonate. Our team of expert chemists has thoroughly analyzed the patented methodology and possesses the technical capability to implement this advanced transesterification and hydrogenation protocol on a commercial scale. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the yield improvements and safety benefits described in the patent are fully realized in our manufacturing operations. Our rigorous QC labs and stringent purity specifications guarantee that every batch meets the highest standards required for clinical and commercial API production, providing our partners with a secure and high-quality supply source.
We invite global pharmaceutical companies and contract research organizations to collaborate with us to optimize their supply chains for M4 receptor antagonist programs. By leveraging our expertise in this specific synthesis, we can offer a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team to request specific COA data, route feasibility assessments, and sample availability. Let us help you accelerate your drug development timeline with a reliable, cost-effective, and environmentally responsible supply of this critical intermediate.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
