Revolutionizing Antiviral Intermediate Production via Catalytic Asymmetric Dihydroxylation Technology
Introduction to Next-Generation Antiviral Intermediate Synthesis
The pharmaceutical industry continuously seeks more efficient pathways for producing critical antiviral agents, particularly nucleoside analogs like Cidofovir and Buciclovir. A pivotal advancement in this domain is detailed in patent CN108912055B, which discloses a novel method for synthesizing key chiral intermediates through asymmetric dihydroxylation. This technology represents a significant departure from conventional chiral pool strategies, offering a streamlined catalytic approach that enhances both stereochemical control and process economics. By leveraging osmium-catalyzed asymmetric dihydroxylation (AD) on allyl-substituted pyrimidine and purine scaffolds, manufacturers can access high-purity intermediates with exceptional enantiomeric excess. This report analyzes the technical merits of this innovation, providing strategic insights for R&D directors, procurement managers, and supply chain leaders aiming to optimize their antiviral drug portfolios.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of acyclic nucleoside phosphonates like Cidofovir has relied heavily on chiral pool starting materials, such as R-glycidyl alcohol or S-2,3-isopropylidene glyceraldehyde. As illustrated in the traditional synthetic routes, these methods involve a cumbersome sequence of protection, coupling, and deprotection steps to install the necessary chirality. 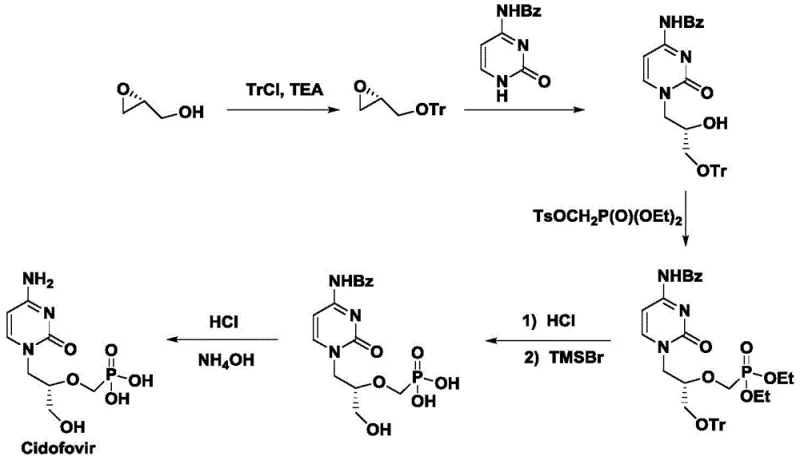 . The reliance on stoichiometric chiral precursors inherently limits cost efficiency, as these materials are often expensive and subject to supply volatility. Furthermore, the multi-step nature of these traditional routes accumulates yield losses at each stage and generates significant chemical waste, complicating purification and increasing the environmental footprint of the manufacturing process. The structural complexity introduced early in the synthesis also restricts flexibility, making process optimization difficult and costly.
. The reliance on stoichiometric chiral precursors inherently limits cost efficiency, as these materials are often expensive and subject to supply volatility. Furthermore, the multi-step nature of these traditional routes accumulates yield losses at each stage and generates significant chemical waste, complicating purification and increasing the environmental footprint of the manufacturing process. The structural complexity introduced early in the synthesis also restricts flexibility, making process optimization difficult and costly.
The Novel Approach
In contrast, the methodology described in CN108912055B introduces a convergent strategy where chirality is induced catalytically rather than purchased stoichiometrically. The core innovation involves the direct asymmetric dihydroxylation of readily available allyl-substituted heterocycles.  . This approach utilizes a catalytic amount of osmium tetroxide precursor paired with chiral cinchona alkaloid ligands to transform simple olefins into chiral diols with high fidelity. By shifting the source of chirality from the starting material to the catalyst system, the process drastically reduces raw material costs and simplifies the supply chain. The reaction conditions are mild, typically operating between 0°C and room temperature in a benign tert-butanol/water solvent system, which facilitates easier handling and safer scale-up compared to harsh acidic or basic conditions found in older methodologies.
. This approach utilizes a catalytic amount of osmium tetroxide precursor paired with chiral cinchona alkaloid ligands to transform simple olefins into chiral diols with high fidelity. By shifting the source of chirality from the starting material to the catalyst system, the process drastically reduces raw material costs and simplifies the supply chain. The reaction conditions are mild, typically operating between 0°C and room temperature in a benign tert-butanol/water solvent system, which facilitates easier handling and safer scale-up compared to harsh acidic or basic conditions found in older methodologies.
Mechanistic Insights into Osmium-Catalyzed Asymmetric Dihydroxylation
The success of this synthetic route hinges on the precise orchestration of the Sharpless Asymmetric Dihydroxylation (SAD) mechanism. The catalytic cycle begins with the coordination of the osmium(VI) species to the electron-deficient double bond of the allyl-pyrimidine or allyl-purine substrate. The chiral environment provided by the ligand, such as (DHQD)2PHAL, dictates the facial selectivity of the oxygen transfer, ensuring the formation of the desired (S)- or (R)-diol configuration essential for biological activity. The use of potassium ferricyanide (K3Fe(CN)6) as a co-oxidant is critical; it efficiently re-oxidizes the reduced osmium(VI) glycolate back to the active osmium(VIII) species, allowing the catalyst to turnover multiple times without being consumed. This regeneration loop is vital for maintaining low catalyst loading (typically 0.2-2 mol%), which is economically advantageous given the high cost of osmium metals. Additionally, the inclusion of methanesulfonamide acts as an accelerator, enhancing the rate of hydrolysis of the osmate ester and preventing product inhibition, thereby driving the reaction to completion with high yields often exceeding 90%.
Controlling impurity profiles is another critical aspect where this mechanism excels. The high stereoselectivity of the ligand-catalyst complex minimizes the formation of the undesired enantiomer, achieving enantiomeric excess (ee) values up to 95% as demonstrated in experimental data. This high optical purity at the intermediate stage significantly reduces the burden on downstream purification processes, such as chiral chromatography or recrystallization, which are often bottlenecks in API manufacturing. Furthermore, the chemoselectivity of the osmium catalyst ensures that other functional groups on the heterocyclic base, such as the exocyclic amine protected by benzoyl or acetyl groups, remain intact during the oxidation. This orthogonal reactivity prevents side reactions that could lead to difficult-to-remove impurities, ensuring a cleaner crude product profile that meets stringent pharmaceutical quality standards.
How to Synthesize Cidofovir Intermediate Efficiently
Implementing this technology requires careful attention to reaction parameters to maximize both yield and enantioselectivity. The process begins with the preparation of the allyl-substituted substrate, followed by the asymmetric dihydroxylation step which establishes the critical chiral centers. Subsequent transformations involve deprotection and phosphonation to yield the final active pharmaceutical ingredient. The patent provides detailed experimental protocols that serve as a robust foundation for process development teams looking to adapt this chemistry for commercial production. For a comprehensive breakdown of the standardized operating procedures and specific reagent quantities, please refer to the technical guide below.
- Prepare the reaction mixture by combining the allyl-substituted pyrimidine or purine substrate with potassium ferricyanide, potassium carbonate, and methanesulfonamide in a tert-butanol/water solvent system.
- Add the osmium catalyst (K2Os2(OH)4) and the chiral ligand (such as (DHQD)2PHAL) to the mixture under inert atmosphere and stir at 0°C to room temperature.
- Quench the reaction with sodium thiosulfate, extract the product, and perform desilylation using a fluoride reagent like TBAF to obtain the final chiral dihydroxy intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement and supply chain stakeholders, the adoption of this catalytic asymmetric synthesis offers transformative benefits regarding cost structure and supply reliability. The shift away from expensive chiral building blocks to commodity chemicals represents a fundamental improvement in the cost of goods sold (COGS). By utilizing inexpensive allyl halides and heterocyclic bases as starting materials, manufacturers can decouple their production costs from the volatile pricing of specialized chiral reagents. This strategic sourcing advantage allows for more stable long-term pricing contracts with API customers, enhancing competitiveness in the generic antiviral market. Moreover, the reduction in synthetic steps directly correlates to lower operational expenditures, as fewer unit operations mean reduced labor, energy, and solvent consumption per kilogram of finished product.
- Cost Reduction in Manufacturing: The elimination of stoichiometric chiral auxiliaries and the use of catalytic amounts of osmium significantly lower raw material expenses. While osmium is a precious metal, its catalytic turnover number (TON) in this system is high enough to render the metal cost negligible on a per-kg basis. Furthermore, the simplified workup procedures reduce solvent usage and waste disposal costs, contributing to substantial overall cost savings without compromising product quality. The ability to achieve high yields in the key stereodefining step minimizes material loss, ensuring maximum throughput from expensive heterocyclic precursors.
- Enhanced Supply Chain Reliability: Relying on broadly available commodity chemicals like potassium ferricyanide and tert-butanol mitigates the risk of supply disruptions often associated with niche chiral intermediates. This diversification of the supply base ensures business continuity even when specific suppliers face production issues. The robustness of the reaction conditions, which tolerate ambient temperatures and standard atmospheric pressure, means that the process can be executed in a wide range of manufacturing facilities without requiring specialized high-pressure or cryogenic equipment, further expanding the pool of qualified contract manufacturing organizations (CMOs).
- Scalability and Environmental Compliance: The biphasic nature of the reaction system (organic/aqueous) facilitates excellent heat dissipation, a critical factor for safe scale-up from pilot plant to commercial tonnage. The use of potassium ferricyanide as a terminal oxidant avoids the generation of hazardous organic peroxides or chlorinated waste streams common in other oxidation methods. This alignment with green chemistry principles simplifies regulatory compliance and environmental permitting, accelerating the timeline for technology transfer and commercial launch. The high atom economy of the dihydroxylation reaction ensures that the majority of reactant mass is incorporated into the final product, minimizing the environmental footprint of the manufacturing process.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric synthesis technology. These answers are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for decision-making. Understanding these nuances is essential for evaluating the feasibility of integrating this route into existing manufacturing pipelines.
Q: What are the primary advantages of this asymmetric dihydroxylation method over traditional chiral pool synthesis?
A: This method eliminates the need for expensive chiral starting materials like R-glycidyl alcohol, utilizing readily available allyl-substituted heterocycles instead. It achieves high stereoselectivity (up to 95% ee) and simplifies the process flow by reducing the number of synthetic steps required to reach the key chiral intermediate.
Q: Which chiral ligands provide the best enantioselectivity for this transformation?
A: Screening data indicates that cinchona alkaloid-derived ligands, specifically (DHQD)2PHAL (L2), provide superior enantioselectivity compared to other variants like (DHQD)2PYR or (DHQ)2PHAL. The optimal reaction temperature for maximizing ee values is typically around 0°C.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process utilizes robust reagents like potassium ferricyanide as a co-oxidant and operates in a biphasic tert-butanol/water system, which is favorable for heat transfer and safety. The catalytic nature of the osmium species minimizes heavy metal waste, aligning with green chemistry principles for industrial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cidofovir Intermediate Supplier
NINGBO INNO PHARMCHEM stands at the forefront of adopting advanced synthetic methodologies to deliver superior pharmaceutical intermediates. Our technical team has extensively evaluated the catalytic asymmetric dihydroxylation route described in CN108912055B and confirmed its viability for large-scale production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our facility is equipped with rigorous QC labs and stringent purity specifications, guaranteeing that every batch of Cidofovir or Buciclovir intermediate meets the highest international pharmacopoeia standards. We understand that consistency is key in the pharmaceutical supply chain, and our robust process controls ensure batch-to-batch reproducibility.
We invite global partners to collaborate with us to leverage this cutting-edge technology for your antiviral drug projects. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you secure a sustainable, cost-effective, and high-quality supply of critical antiviral intermediates for the global market.
