Advanced Catalytic Cyclization for N-Acyl Indole Derivatives: Commercial Scale-Up and Process Optimization
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to construct complex heterocyclic scaffolds, particularly indole derivatives which serve as critical backbones for numerous bioactive molecules. Patent CN107501162B introduces a transformative preparation method for N-acyl-substituted indole derivatives that addresses long-standing challenges in synthetic efficiency and environmental impact. This innovative approach utilizes a cyclization reaction of specific precursors under the influence of a base and a transition metal catalyst within a moderate temperature range of 40-170°C. By shifting away from traditional stoichiometric reagents towards a catalytic system, this technology offers a robust solution for generating high-value intermediates with exceptional purity profiles. For R&D directors and procurement specialists, understanding the nuances of this patent is essential for optimizing supply chains and reducing the overall cost of goods sold in competitive therapeutic areas.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of indole cores has relied heavily on classical methodologies such as the Fischer indole synthesis or the Larock indole synthesis, which, while foundational, present significant drawbacks for modern industrial applications. These traditional routes often struggle with poor regioselectivity, leading to complex mixtures of isomers that require expensive and time-consuming purification steps to resolve. Furthermore, many established protocols necessitate the use of strong acid systems or harsh reaction conditions that pose safety risks and increase the burden on waste treatment facilities. The reliance on stoichiometric amounts of reagents in older methods also inflates raw material costs and generates substantial chemical waste, contradicting the principles of green chemistry that are increasingly mandated by global regulatory bodies. Consequently, manufacturers face difficulties in scaling these processes without compromising on yield or environmental compliance.
The Novel Approach
In stark contrast, the methodology disclosed in CN107501162B leverages a catalytic cyclization strategy that dramatically simplifies the synthetic landscape for N-acyl indoles. By employing catalytic amounts of common copper or palladium salts in conjunction with organic or inorganic bases, the reaction proceeds smoothly in various organic solvents without the need for corrosive strong acids. This shift not only enhances the atom economy of the process but also significantly improves the operational safety profile for plant personnel. The versatility of this system allows for the accommodation of diverse substituents on the benzene ring, providing a flexible platform for synthesizing a wide library of derivatives. This technological leap enables reliable pharmaceutical intermediates supplier networks to offer more consistent quality and faster turnaround times for custom synthesis projects.

Mechanistic Insights into Copper and Palladium Catalyzed Cyclization
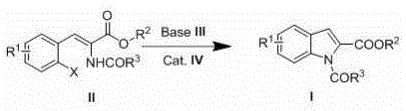
The core of this technological advancement lies in the precise orchestration of the cyclization mechanism, where the interaction between the base, the catalyst, and the substrate dictates the reaction outcome. The process initiates with the activation of the precursor compound (Formula II) by the base, which facilitates the subsequent intramolecular coupling mediated by the transition metal catalyst. Whether utilizing cuprous iodide, cuprous bromide, or palladium complexes like PdCl2(dppf), the catalyst serves to lower the activation energy barrier for the ring-closing step, allowing the reaction to proceed efficiently at temperatures between 90-150°C. This mechanistic pathway ensures that the formation of the indole ring is highly selective, minimizing the generation of side products that typically plague non-catalytic thermal cyclizations. For technical teams, this level of control over the reaction trajectory is paramount for ensuring batch-to-batch consistency in commercial production.
Furthermore, the impurity profile of the final product is inherently managed by the mildness of the reaction conditions and the specificity of the catalytic cycle. Avoiding strong acid systems prevents the degradation of sensitive functional groups that might be present on the substrate, thereby preserving the integrity of the molecular structure throughout the synthesis. The use of thin-layer chromatography (TLC) for monitoring ensures that the reaction is quenched precisely upon completion, preventing over-reaction or decomposition of the product. This rigorous control over the chemical environment translates directly into higher crude purity, which reduces the load on downstream purification units. Such mechanistic robustness is a key factor in achieving cost reduction in pharmaceutical intermediates manufacturing by lowering the overall processing time and solvent consumption.
How to Synthesize N-Acyl Indole Derivatives Efficiently
Implementing this synthesis route requires a clear understanding of the operational parameters defined in the patent to maximize yield and safety. The process begins with the careful selection of the solvent system, with options ranging from polar aprotic solvents like DMF and DMSO to aromatic solvents like toluene, depending on the solubility of the specific substrate. The molar ratios of the base and catalyst relative to the starting material are critical, with the patent specifying a base ratio of 0.2-4.0:1.0 and a catalyst ratio of 0.01-0.2:1.0 to ensure optimal kinetics without excessive reagent waste. Operators must maintain strict temperature control within the 40-170°C window and utilize real-time monitoring to determine the exact endpoint of the reaction. Detailed standardized synthesis steps see the guide below.
- Prepare the reaction mixture by combining the precursor compound (Formula II) with a catalytic amount of copper or palladium reagent and a stoichiometric base in an organic solvent.
- Heat the reaction system to a temperature range of 40-170°C and maintain stirring while monitoring progress via thin-layer chromatography until starting material consumption is complete.
- Upon completion, cool the mixture, perform standard workup procedures including column chromatography and reduced pressure distillation to isolate the pure N-acyl indole product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this catalytic cyclization technology offers profound benefits for procurement managers and supply chain heads looking to optimize their vendor portfolios. The elimination of strong acid systems and the use of catalytic rather than stoichiometric metal reagents fundamentally alter the cost structure of production, leading to substantial savings in raw material expenditure. Additionally, the simplified workup procedure, which involves standard column chromatography and distillation, reduces the complexity of the manufacturing process, thereby enhancing the reliability of supply timelines. This efficiency is crucial for maintaining commercial scale-up of complex pharmaceutical intermediates without encountering the bottlenecks typical of older synthetic routes. Companies can expect a more resilient supply chain capable of adapting to fluctuating market demands with greater agility.
- Cost Reduction in Manufacturing: The transition to a catalytic system significantly lowers the consumption of expensive metal reagents, as only minute quantities are required to drive the reaction to completion. By avoiding the need for specialized corrosion-resistant equipment associated with strong acid processes, capital expenditure for production facilities is also minimized. Furthermore, the high yields reported, reaching up to 88%, mean that less raw material is wasted per unit of product, directly improving the margin profile for bulk purchases. These factors combine to create a highly competitive pricing structure for end-users seeking high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The simplicity of the raw materials required for this synthesis ensures that sourcing remains stable even during periods of global chemical supply volatility. Since the reaction conditions are mild and the operation is straightforward, the risk of batch failure due to operational errors is drastically reduced, guaranteeing consistent delivery schedules. This reliability is essential for downstream manufacturers who depend on just-in-time inventory models to keep their own production lines running smoothly. Partnering with a supplier proficient in this technology mitigates the risk of delays caused by complex purification struggles or low-yield batches.
- Scalability and Environmental Compliance: The environmentally friendly nature of this process, characterized by the absence of harsh acids and the generation of less hazardous waste, aligns perfectly with modern sustainability mandates. Scaling this reaction from laboratory to multi-ton production is facilitated by the robust nature of the catalytic system, which performs consistently across different batch sizes. This scalability ensures that reducing lead time for high-purity pharmaceutical intermediates is achievable without compromising on regulatory compliance or environmental safety standards. It represents a future-proof investment for long-term supply partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for decision-makers evaluating this technology for their supply chains. Understanding these details helps in assessing the feasibility of integrating this route into existing manufacturing frameworks.
Q: What are the primary advantages of this catalytic method over classical indole synthesis?
A: Unlike classical methods such as Fischer or Larock synthesis which often suffer from regioselectivity issues or require harsh acidic conditions, this patented process utilizes mild basic conditions with catalytic metal salts. This results in significantly higher yields (up to 88%), simpler purification, and better environmental compatibility, making it ideal for industrial scale-up.
Q: Which catalysts are most effective for this cyclization reaction?
A: The patent data indicates that both copper and palladium reagents are highly effective. Specifically, Cuprous Iodide (CuI), Cuprous Bromide (CuBr), and Palladium Acetate have demonstrated robust performance. The choice between copper and palladium often depends on the specific substrate substituents and cost considerations, with copper offering a more economical option for large-scale production.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process is explicitly designed for industrial applicability. It features short reaction steps, uses easily obtainable raw materials, and avoids strong acid systems which simplifies equipment corrosion management. The high yields and straightforward workup procedures significantly reduce production costs and lead times for supply chain operations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Acyl Indole Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient synthetic routes in maintaining a competitive edge in the global pharmaceutical market. Our team of expert chemists has extensively analyzed the potential of the CN107501162B process and is fully equipped to translate this laboratory-scale innovation into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project moves seamlessly from development to full-scale manufacturing. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that verify every batch against the highest industry standards, giving you confidence in the consistency of your supply.
We invite you to collaborate with us to leverage this advanced technology for your specific product needs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis that details how implementing this catalytic route can optimize your budget. We encourage potential partners to reach out for specific COA data and route feasibility assessments to understand how our capabilities align with your strategic goals. Let us help you streamline your supply chain and secure a reliable source of high-quality intermediates for your future projects.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
