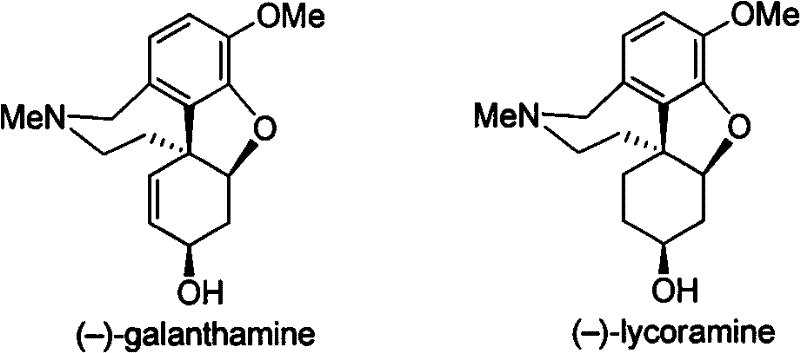
Scalable Asymmetric Synthesis of Galanthamine and Lycoramine for Commercial API Production
Scalable Asymmetric Synthesis of Galanthamine and Lycoramine for Commercial API Production
The pharmaceutical industry's demand for effective treatments for neurodegenerative disorders continues to drive innovation in the synthesis of complex alkaloids. Patent CN102532149A introduces a groundbreaking asymmetric synthesis method for producing Galanthamine and Lycoramine, two potent acetylcholinesterase inhibitors widely used in the treatment of Alzheimer's disease and myasthenia gravis. Unlike traditional methods that rely on the extraction of these compounds from natural sources like Snowdrop or Daffodil, where content is exceedingly low at approximately 1/10,000, this new chemical approach offers a robust, scalable, and economically viable alternative. By utilizing an alpha-aryloxy substituted cyclohexanedione monoethylene glycol ketal as a starting material, the process leverages advanced asymmetric catalytic hydrogenation to establish chirality with high precision. This technological leap not only addresses the supply chain vulnerabilities associated with agricultural extraction but also provides a reliable galanthamine supplier pathway that ensures consistent quality and purity for global pharmaceutical manufacturers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the procurement of Galanthamine and Lycoramine has been fraught with significant logistical and economic challenges. The primary conventional method involves extraction from Amaryllidaceae plants, a process that is inherently inefficient due to the trace amounts of alkaloids present in the biomass. This scarcity drives up costs and creates a volatile supply chain susceptible to seasonal variations, crop failures, and geopolitical instability in sourcing regions. Furthermore, chemical synthesis attempts prior to this invention often resulted in racemic mixtures, necessitating costly and wasteful resolution steps to isolate the biologically active enantiomer. Previous asymmetric syntheses, such as those reported by Trost or Fan, while scientifically elegant, often suffered from excessive step counts (up to 16 steps) and low overall yields (as low as 2.8%), rendering them impractical for cost reduction in API manufacturing on a commercial scale. These inefficiencies create a bottleneck for pharmaceutical companies seeking to secure long-term supplies of these critical neurological therapeutics.
The Novel Approach
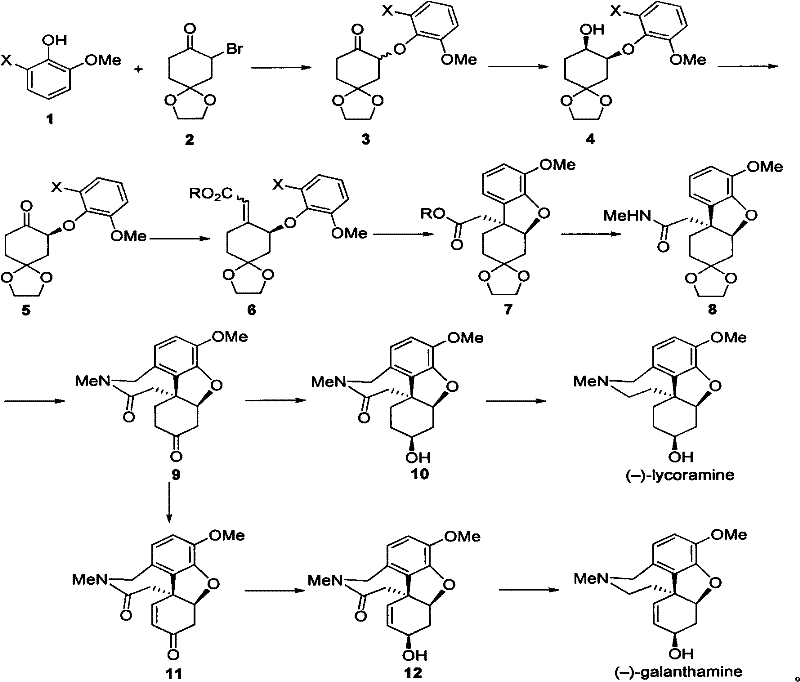
The methodology outlined in patent CN102532149A represents a paradigm shift towards efficiency and scalability in alkaloid synthesis. By employing a chiral spiro bisphosphine ligand Ruthenium-bisphosphine/bisamine catalyst, the process achieves high enantioselectivity early in the synthetic sequence through asymmetric catalytic hydrogenation. This strategic placement of the chirality-defining step minimizes the loss of optical purity in subsequent transformations. The route proceeds through a streamlined series of reactions including oxidation, Horner-Wadsworth-Emmons olefination, and a pivotal intramolecular reductive Heck reaction to construct the tetracyclic core. This novel approach drastically reduces the number of synthetic operations compared to prior art, achieving overall yields of 20.1% over 12 steps for Galanthamine and an impressive 40.2% over 10 steps for Lycoramine. Such improvements in step economy and yield directly translate to enhanced process viability, making this a superior choice for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Ru-Catalyzed Asymmetric Hydrogenation
The cornerstone of this synthetic strategy is the enantioselective hydrogenation of the alpha-aryloxy substituted cyclohexanedione derivative. This transformation utilizes a sophisticated Ruthenium catalyst system, specifically complexes like Ru-(S)-SDP/(R,R)-DPEN, which facilitate the transfer of hydrogen to the prochiral ketone substrate with exceptional stereocontrol. The mechanism involves the coordination of the substrate to the metal center, followed by hydride transfer that is directed by the chiral environment of the spiro bisphosphine ligand. This step is critical as it sets the absolute configuration for the entire molecule, ensuring that downstream intermediates maintain the desired optical activity without the need for chiral chromatography or recrystallization. The high turnover frequency and selectivity of this catalyst system allow for the use of lower catalyst loadings, which is a significant factor in reducing the residual metal burden in the final API, thereby simplifying purification protocols and ensuring compliance with stringent regulatory limits for heavy metals in pharmaceutical products.
Following the establishment of chirality, the synthesis employs a sequence of robust carbon-carbon bond-forming reactions to build molecular complexity. The intramolecular reductive Heck reaction is particularly noteworthy for its ability to close the ring system efficiently under mild conditions using formic acid or sodium formate as the hydrogen source. This palladium-catalyzed cyclization avoids the use of high-pressure hydrogen gas typically required for standard Heck reductions, enhancing operational safety and ease of handling in a manufacturing setting. Furthermore, the subsequent amidation and cyclization steps utilize common reagents like paraformaldehyde and trifluoroacetic acid, which are readily available and cost-effective. The final reduction of the amide and carbonyl groups using triethoxysilane and zinc acetate or L-Selectride provides a chemoselective pathway to the target amines, preserving the sensitive ether linkages and double bonds within the structure. This careful orchestration of reactivity ensures high purity and minimizes the formation of difficult-to-remove impurities.
How to Synthesize Galanthamine Efficiently
The synthesis of Galanthamine via this patented route is designed for operational simplicity and high fidelity. It begins with the preparation of the chiral alcohol intermediate via asymmetric hydrogenation, followed by oxidation to the corresponding ketone. The chain is then extended via phosphonate chemistry before undergoing the crucial ring-closing reductive Heck reaction. Subsequent functional group manipulations, including hydrolysis, amidation, and Pictet-Spengler type cyclization, construct the nitrogen-containing ring. The final stages involve stereoselective reduction of the ketone and the amide to yield the target alkaloid. This logical progression allows for the isolation and characterization of stable intermediates, providing multiple control points for quality assurance. For detailed laboratory protocols and specific reaction conditions optimized for scale, please refer to the standardized synthesis guide below.
- Initiate the synthesis with asymmetric catalytic hydrogenation of an alpha-aryloxy substituted cyclohexanedione monoethylene glycol ketal using a chiral Ruthenium-bisphosphine catalyst to establish stereochemistry.
- Perform oxidation followed by a Horner-Wadsworth-Emmons reaction and an intramolecular reductive Heck reaction to construct the core tetracyclic framework.
- Complete the synthesis through intramolecular cyclization and selective reduction of carbonyl and amide functionalities to yield the final natural or unnatural enantiomers.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this asymmetric synthesis method offers substantial strategic benefits beyond mere technical feasibility. The shift from extraction to total synthesis eliminates the dependency on agricultural cycles and the environmental degradation associated with harvesting wild plant populations. This transition ensures a stable, year-round supply of high-purity Galanthamine and Lycoramine, mitigating the risks of shortage-induced price spikes. Moreover, the streamlined nature of the synthetic route, characterized by fewer steps and higher overall yields compared to historical methods, inherently lowers the cost of goods sold. By avoiding the massive material losses associated with resolving racemic mixtures, manufacturers can achieve significant cost reduction in API manufacturing, passing these savings down the supply chain or reinvesting them into further R&D. The use of commercially available starting materials and standard reagents further enhances the robustness of the supply chain, reducing lead time for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The elimination of resolution steps and the high efficiency of the catalytic hydrogenation significantly reduce raw material consumption and waste disposal costs. The process avoids the use of expensive chiral auxiliaries that are stoichiometric rather than catalytic, leading to a leaner cost structure. Additionally, the high yields in key steps such as the reductive Heck cyclization minimize the volume of solvent and reagents required per kilogram of product, driving down utility and processing expenses substantially.
- Enhanced Supply Chain Reliability: Synthetic production decouples the supply of these critical neurological drugs from the vagaries of climate and harvest seasons. This reliability allows pharmaceutical companies to maintain consistent inventory levels and meet market demand without interruption. The scalability of the chemical process means that production capacity can be ramped up quickly in response to increased clinical demand or new indications, ensuring business continuity and protecting market share against competitors reliant on limited natural sources.
- Scalability and Environmental Compliance: The synthetic route is designed with green chemistry principles in mind, utilizing catalytic processes that generate less waste than stoichiometric alternatives. The avoidance of heavy metal contaminants through efficient catalyst removal protocols simplifies the environmental compliance burden. Furthermore, the robustness of the reaction conditions allows for safe operation in large-scale reactors, facilitating the transition from pilot plant to multi-ton commercial production without the need for extensive process re-engineering or specialized equipment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Galanthamine and Lycoramine using this advanced asymmetric methodology. These insights are derived directly from the patent specifications and are intended to clarify the feasibility and advantages of this route for potential partners and stakeholders. Understanding these details is crucial for evaluating the technology's fit within your existing manufacturing portfolio.
Q: How does this asymmetric synthesis method improve upon traditional extraction?
A: Traditional extraction from Amaryllidaceae plants yields extremely low quantities (approx. 1/10,000), making it unsustainable for large-scale demand. This patented asymmetric synthesis provides a consistent, high-yield alternative that is independent of seasonal plant availability and environmental constraints.
Q: What is the key advantage of the Ruthenium-catalyzed hydrogenation step?
A: The use of a chiral spiro bisphosphine ligand Ruthenium catalyst allows for the direct establishment of the critical chiral center early in the synthesis. This avoids the need for inefficient resolution of racemic mixtures, significantly improving overall atom economy and optical purity.
Q: Is this process suitable for industrial scale-up?
A: Yes, the patent explicitly highlights the method's suitability for large-scale production. The route utilizes robust reactions like reductive Heck coupling and standard oxidation/reduction steps that are well-understood in process chemistry, facilitating safe and efficient commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Galanthamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of securing a dependable supply of high-value neurological APIs like Galanthamine. Our team of expert process chemists has extensively analyzed the route described in CN102532149A and possesses the technical capability to implement this asymmetric synthesis at an industrial level. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project moves seamlessly from development to market. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of Galanthamine or Lycoramine meets the highest international pharmacopoeia standards. We are committed to delivering not just a product, but a partnership built on technical excellence and supply security.
We invite you to discuss how our manufacturing capabilities can support your specific requirements for these complex alkaloids. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating exactly how this synthetic route can optimize your budget. Please contact us today to request specific COA data and route feasibility assessments, and let us help you secure a competitive advantage in the global market for Alzheimer's therapeutics through reliable, high-quality chemical supply.
