Scalable Synthesis of 2,1-Benzisoxazole Derivatives for Commercial Anticancer Applications
Introduction to Novel 2,1-Benzisoxazole Synthetic Technology
The pharmaceutical industry is constantly seeking robust and economically viable pathways to access complex heterocyclic scaffolds that serve as critical intermediates for next-generation therapeutics. Patent CN110590760B introduces a groundbreaking methodology for the construction of 2,1-benzisoxazole derivatives fused with 1,2,3-triazole moieties, which are structurally analogous to the potent antimitotic agent Combretastatin. This intellectual property represents a significant leap forward in organic synthesis by replacing multi-step, hazardous protocols with a streamlined one-pot cascade reaction that utilizes readily available starting materials such as o-bromoacetophenone, various aldehydes, and sodium azide. The strategic design of this synthetic route not only addresses the longstanding challenges associated with the instability of azide intermediates but also ensures high atom economy and operational simplicity, making it an ideal candidate for industrial adoption by reliable pharmaceutical intermediate suppliers seeking to optimize their manufacturing portfolios.
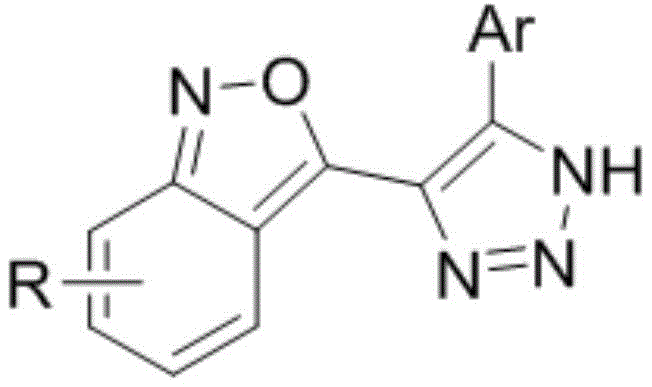
Furthermore, the biological implications of these novel biheterocyclic compounds extend far beyond mere structural novelty, as the incorporation of the 1,2,3-triazole ring system effectively locks the molecular conformation, thereby preventing cis-trans isomerization that often plagues stilbene-based anticancer drugs. This structural rigidity enhances the drug-likeness and metabolic stability of the resulting analogs, potentially leading to superior pharmacokinetic profiles in vivo. By leveraging the versatility of this patented approach, research and development teams can rapidly generate diverse libraries of compounds to explore structure-activity relationships, accelerating the discovery of new candidates with enhanced anti-tumor and anticancer activities. The convergence of synthetic efficiency and biological potential positions this technology as a cornerstone for future oncology drug development programs globally.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of benzisoxazole cores has been fraught with significant technical and economic hurdles that have hindered their widespread adoption in large-scale manufacturing processes. Traditional methodologies predominantly rely on the use of o-nitrobenzaldehyde derivatives as key starting materials, which necessitate rigorous reduction steps to convert the nitro group into the requisite amine or hydroxylamine intermediates prior to cyclization. These reduction processes often demand the use of expensive precious metal catalysts such as palladium or platinum, or alternatively, stoichiometric amounts of hazardous reducing agents that generate substantial quantities of toxic waste, thereby increasing both the environmental footprint and the overall cost of goods. Additionally, alternative routes utilizing o-azidobenzaldehyde suffer from inherently low yields due to the thermal instability of the azide functionality, which poses serious safety risks during handling and limits the scope of compatible substrates to only the most robust chemical entities.
The Novel Approach
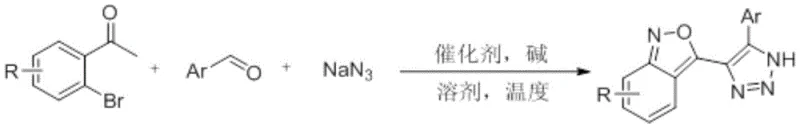
In stark contrast to these legacy methods, the innovative protocol disclosed in CN110590760B circumvents these bottlenecks by employing a direct oxidative cyclization strategy that transforms simple o-bromoacetophenones into the target benzisoxazole framework in a single operational step. This transformative approach eliminates the need for pre-functionalized nitro or azide precursors, instead generating the reactive azide species in situ from inexpensive sodium azide, which significantly mitigates safety concerns associated with isolating energetic intermediates. The reaction proceeds through a sophisticated cascade mechanism involving aldol condensation, azido addition, and subsequent copper-catalyzed oxidative cyclization, all occurring within the same reaction vessel without the need for intermediate isolation or purification. This telescoped process not only drastically reduces the total processing time and solvent consumption but also enhances the overall yield and purity of the final product, offering a compelling value proposition for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into CuO-Catalyzed Oxidative Cyclization
A deep understanding of the catalytic cycle underpinning this transformation is essential for R&D directors aiming to optimize process parameters and ensure consistent quality control during scale-up. The reaction initiates with a base-mediated aldol condensation between the o-bromoacetophenone and the aromatic aldehyde, forming an alpha,beta-unsaturated ketone intermediate that serves as the electrophilic acceptor for the subsequent nucleophilic attack by the azide anion. Following the azido-Michael addition, the system undergoes an intramolecular nucleophilic substitution where the azide nitrogen displaces the ortho-bromine atom, constructing the triazole ring while simultaneously setting the stage for the final oxidative closure. The presence of copper(II) oxide is critical at this juncture, as it facilitates the dehydrogenative aromatization of the dihydro-intermediate to the fully aromatic benzisoxazole system, likely proceeding through a single-electron transfer mechanism that regenerates the active catalytic species without requiring external oxidants.
From an impurity control perspective, the selection of potassium carbonate as the base plays a pivotal role in suppressing side reactions such as the hydrolysis of the nitrile or the formation of polymeric by-products that often complicate downstream purification. The mild basicity of K2CO3 ensures that the reaction environment remains sufficiently nucleophilic to drive the azide addition forward while avoiding the harsh conditions that could lead to the decomposition of the sensitive triazole moiety. Furthermore, the use of dimethyl sulfoxide (DMSO) as the solvent provides a high dielectric constant medium that stabilizes the charged transition states and enhances the solubility of the inorganic salts, thereby ensuring homogeneous reaction kinetics throughout the process. This precise tuning of reaction conditions results in a clean impurity profile, minimizing the burden on analytical laboratories and reducing the number of recrystallization steps required to meet stringent purity specifications for clinical grade materials.
How to Synthesize 2,1-Benzisoxazole Derivatives Efficiently
Implementing this synthetic route in a laboratory or pilot plant setting requires strict adherence to the optimized parameters defined in the patent to maximize yield and reproducibility. The process begins by charging a reactor with the specified molar ratios of o-bromoacetophenone, the chosen aldehyde variant, and sodium azide, followed by the addition of the copper catalyst and base in DMSO. It is imperative to maintain the reaction temperature within the narrow window of 100-120°C to balance the kinetic energy required for cyclization against the thermal stability limits of the azide components. Detailed standardized operating procedures regarding mixing speeds, heating ramps, and quenching protocols are essential to ensure that the exothermic nature of the initial condensation does not lead to runaway scenarios, and the complete step-by-step guide for executing this synthesis is provided below for technical reference.
- Combine o-bromoacetophenone, aromatic aldehyde, and sodium azide in DMSO solvent with CuO catalyst and K2CO3 base.
- Heat the reaction mixture to 100-120°C with magnetic stirring for approximately 24 hours to facilitate cascade cyclization.
- Perform aqueous workup with ethyl acetate extraction, followed by column chromatography to isolate the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic methodology offers tangible benefits that directly impact the bottom line and operational resilience of the organization. By shifting away from dependency on scarce and volatile noble metal catalysts towards abundant and inexpensive copper salts, manufacturers can achieve substantial cost savings in raw material procurement while insulating themselves from the price fluctuations associated with the precious metals market. The elimination of separate reduction and protection/deprotection steps further contributes to cost reduction in manufacturing by reducing the total number of unit operations, which in turn lowers labor costs, energy consumption, and equipment occupancy time, allowing for higher throughput within existing facility footprints.
- Cost Reduction in Manufacturing: The economic advantage of this process is primarily driven by the replacement of expensive reagents with commodity chemicals that are available in bulk quantities from multiple global suppliers, ensuring competitive pricing and negotiation leverage. The one-pot nature of the reaction significantly reduces solvent usage and waste disposal costs, as there is no need to isolate and dry unstable intermediates, which traditionally requires additional filtration and drying equipment. Moreover, the high selectivity of the copper-catalyzed system minimizes the formation of difficult-to-remove impurities, thereby increasing the overall recovery rate of the final product and reducing the loss of valuable materials during purification stages.
- Enhanced Supply Chain Reliability: Sourcing reliability is greatly improved because the key starting materials, such as o-bromoacetophenone and substituted benzaldehydes, are established commodity chemicals with mature and diversified supply chains that are less prone to disruption compared to specialized fine chemical intermediates. The robustness of the reaction conditions allows for flexibility in sourcing, as the process tolerates minor variations in reagent quality without compromising the final product specification, reducing the risk of batch failures due to raw material inconsistencies. This resilience ensures continuous production schedules and reliable delivery timelines for downstream customers who depend on a steady flow of high-quality intermediates for their own drug development pipelines.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, this method aligns perfectly with green chemistry principles by avoiding the use of toxic heavy metals and minimizing the generation of hazardous waste streams that require costly treatment. The scalability of the process is demonstrated by its ability to function effectively in standard stainless steel reactors without the need for specialized pressure vessels or cryogenic cooling systems, facilitating a smooth transition from gram-scale laboratory experiments to multi-ton commercial production. This ease of scale-up reduces the capital expenditure required for new capacity expansion and accelerates the time-to-market for new products derived from this versatile chemical platform.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and potential of this patented synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear picture of what partners can expect when adopting this methodology. Understanding these nuances is critical for making informed decisions about process integration and long-term strategic planning.
Q: What are the primary advantages of this one-pot synthesis over traditional nitro-reduction methods?
A: This method eliminates the need for hazardous reducing agents and expensive noble metal catalysts, utilizing inexpensive copper oxide instead, which drastically simplifies purification and lowers raw material costs.
Q: Is this synthetic route suitable for large-scale commercial production?
A: Yes, the use of thermally stable solvents like DMSO and moderate temperatures (100-120°C) ensures excellent process safety and scalability for metric-ton manufacturing.
Q: What is the substrate scope for the aldehyde component in this reaction?
A: The protocol demonstrates wide compatibility with various substituents including electron-donating methoxy groups, electron-withdrawing nitro and cyano groups, and heterocyclic furan rings.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,1-Benzisoxazole Derivatives Supplier
As the global demand for advanced oncology intermediates continues to rise, partnering with an experienced CDMO like NINGBO INNO PHARMCHEM ensures that your project benefits from our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team possesses the expertise to adapt this patented one-pot protocol to your specific quality requirements, ensuring stringent purity specifications are met through our rigorous QC labs equipped with state-of-the-art analytical instrumentation. We understand that consistency is key in pharmaceutical supply, and our robust quality management systems guarantee that every batch of 2,1-benzisoxazole derivatives delivered meets the highest international standards for safety and efficacy.
We invite you to engage with our technical procurement team to discuss how we can tailor this synthesis to your specific needs and provide a Customized Cost-Saving Analysis for your project. By collaborating with us, you gain access to specific COA data and route feasibility assessments that will empower you to make confident decisions about your supply chain strategy. Let us help you accelerate your drug development timeline with a reliable, cost-effective, and scalable supply of these critical anticancer intermediates.
