Advanced Chiral Resolution Strategy for Commercial Ramosetron Hydrochloride Production
The pharmaceutical industry continuously seeks robust synthetic pathways that balance stereochemical purity with economic viability, particularly for potent antiemetic agents like Ramosetron Hydrochloride. Patent CN100486977C introduces a transformative methodology for the preparation of R-5-[(1-Methyl-1H-indole-3-yl) carbonyl]-4,5,6,7-tetrahydro-1H-benzimidazole hydrochloride, shifting the paradigm from traditional late-stage resolution to an innovative resolution-before-condensation strategy. This technical breakthrough addresses critical bottlenecks in chiral drug manufacturing by fundamentally restructuring the synthetic sequence to prioritize enantiomeric enrichment prior to the introduction of expensive side chains. By decoupling the resolution step from the final coupling reaction, the process achieves superior control over impurity profiles while drastically minimizing the consumption of high-value precursors. For global supply chain stakeholders, this represents a significant evolution in process chemistry, offering a reliable pharmaceutical intermediates supplier pathway that aligns with modern green chemistry principles and cost-efficiency mandates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Ramosetron has relied on a condensation-first approach where the racemic acid core is coupled with N-methylindole prior to chiral separation. As illustrated in the reaction scheme below, this conventional route necessitates the attachment of the costly N-methylindole moiety to the entire racemic mixture, meaning fifty percent of this valuable reagent is inevitably wasted on the unwanted S-enantiomer which is subsequently discarded during resolution. Furthermore, the resolution yield in these traditional processes is notoriously low, often falling below twenty percent, which severely impacts the overall mass balance and economic feasibility of the campaign. The complexity of solvent recovery in these multi-step sequences adds another layer of operational burden, making the process less attractive for large-scale cost reduction in pharmaceutical intermediates manufacturing. The accumulation of byproducts and the difficulty in purifying the final racemate before resolution often lead to extended processing times and increased environmental waste.
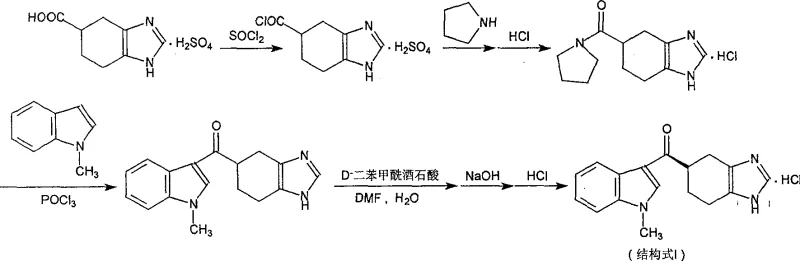
The Novel Approach
In stark contrast, the novel methodology disclosed in the patent employs a strategic resolution-before-condensation sequence that fundamentally alters the economic landscape of the synthesis. By introducing a temporary, easily removable chiral auxiliary group (indoline) prior to the resolution step, the process allows for the efficient isolation of the desired R-enantiomer before the expensive N-methylindole is ever introduced. This approach ensures that the high-value side chain is only reacted with the optically pure intermediate, effectively eliminating the waste associated with processing the wrong enantiomer. The reaction pathway depicted below demonstrates how the resolved intermediate is hydrolyzed back to the chiral acid, which is then activated for the final coupling, ensuring that every gram of N-methylindole contributes directly to the final product yield. This logical reordering of synthetic steps not only simplifies the purification protocols but also facilitates a more streamlined workflow that is inherently more suitable for commercial scale-up of complex pharmaceutical intermediates.
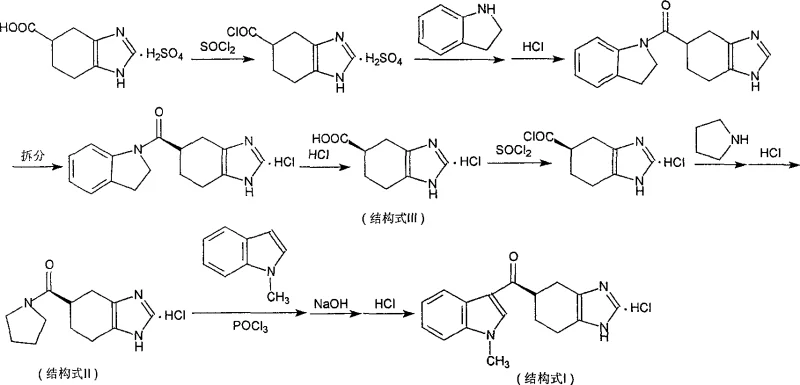
Mechanistic Insights into Chiral Resolution and Active Amide Formation
The core of this technological advancement lies in the precise manipulation of the chiral center through the formation of a diastereomeric salt with D-dibenzoyl tartaric acid or tartrate. Unlike direct resolution of the final product, resolving the indoline-amide intermediate leverages the steric bulk of the indoline group to enhance the crystallization efficiency of the diastereomeric salts. This mechanistic nuance allows for repeated recrystallization in alcoholic solvents until high optical purity is achieved, a critical parameter for meeting the stringent regulatory requirements of API production. Once the R-enantiomer is secured, the indoline group is cleaved via hydrolysis under acidic or alkaline conditions, regenerating the chiral carboxylic acid without racemization. This preservation of stereochemical integrity throughout the hydrolysis step is paramount, ensuring that the hard-won optical purity is not compromised before the final bond-forming event.
Following the regeneration of the chiral acid, the process utilizes a sophisticated activation strategy involving the formation of an active amide using tetramethyleneimine. This specific intermediate serves as a highly reactive acylating agent, significantly lowering the energy barrier for the subsequent Friedel-Crafts-type acylation with N-methylindole. The use of phosphorus oxychloride as a dehydrating agent in the final step facilitates the attack of the electron-rich indole ring on the activated carbonyl, driving the reaction to completion with high conversion rates. This mechanistic design effectively bypasses the need for harsh conditions that might degrade the sensitive benzimidazole core, thereby minimizing the formation of degradation impurities. The result is a robust chemical transformation that delivers the target molecule with exceptional purity, addressing the primary concerns of any R&D Director focused on impurity control and process robustness.
How to Synthesize Ramosetron Hydrochloride Efficiently
The synthesis of Ramosetron Hydrochloride via this novel route requires careful attention to reaction conditions, particularly during the chlorination and resolution phases. The process begins with the conversion of the racemic acid to its acid chloride using thionyl chloride, followed by immediate acylation with indoline to set the stage for resolution. Detailed standard operating procedures for temperature control, stoichiometry, and crystallization parameters are essential to replicate the high yields reported in the patent literature. Operators must ensure strict pH control during the hydrolysis and salification steps to prevent epimerization of the chiral center. For a comprehensive breakdown of the specific reaction conditions and workup procedures, please refer to the standardized guide below.
- Convert racemic tetrahydrobenzimidazole carboxylic acid to acid chloride and react with indoline to form a resolvable amide intermediate.
- Perform chiral resolution using D-dibenzoyl tartaric acid to isolate the R-enantiomer, followed by hydrolysis to recover the chiral acid.
- Activate the chiral acid with thionyl chloride, form the active amide with tetramethyleneimine, and finalize condensation with N-methylindole.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this novel synthetic route offers compelling advantages that directly translate to improved margins and supply security. The most significant benefit is the drastic reduction in the consumption of N-methylindole, a key raw material that represents a substantial portion of the bill of materials. By deferring the introduction of this reagent until after resolution, the process eliminates the inherent fifty percent loss associated with racemic coupling, leading to substantial cost savings without compromising quality. Additionally, the simplified operational flow reduces the demand for complex solvent systems and extensive purification columns, further lowering the variable costs associated with manufacturing. These efficiencies make the process highly attractive for reducing lead time for high-purity pharmaceutical intermediates while maintaining a competitive price point in the global market.
- Cost Reduction in Manufacturing: The elimination of wasted N-methylindole on the S-enantiomer creates a direct and significant reduction in raw material costs. Furthermore, the ability to recycle solvents more effectively due to the cleaner reaction profile reduces utility and waste disposal expenses. The higher overall yield means that less starting material is required to produce the same amount of final API, amplifying the economic benefits across the entire production volume. This efficiency allows manufacturers to offer more competitive pricing structures to downstream partners while maintaining healthy margins.
- Enhanced Supply Chain Reliability: The reliance on readily available reagents like indoline and tetramethyleneimine, rather than scarce chiral pool materials, ensures a stable and resilient supply chain. The robustness of the resolution step minimizes the risk of batch failures due to poor optical purity, guaranteeing consistent output volumes. This predictability is crucial for long-term supply agreements and helps mitigate the risks associated with raw material volatility. Suppliers adopting this method can provide more reliable delivery schedules, enhancing trust with global pharmaceutical clients.
- Scalability and Environmental Compliance: The process is designed with industrial scalability in mind, featuring steps that are easily adaptable to large-scale reactors and continuous flow systems. The reduction in solvent usage and the potential for 'one-pot' operations significantly lower the environmental footprint of the manufacturing process. This alignment with green chemistry principles simplifies regulatory compliance and reduces the burden of waste treatment. Consequently, the method supports sustainable growth and meets the increasingly stringent environmental standards imposed on chemical manufacturing facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis method. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation. Understanding these nuances is essential for evaluating the feasibility of technology transfer and integration into existing production lines. Stakeholders are encouraged to review these points to gain a clearer picture of the operational benefits.
Q: How does the new resolution method reduce raw material costs?
A: By resolving the chirality early using indoline instead of N-methylindole, the expensive side chain is only introduced to the correct R-enantiomer, avoiding waste on the discarded S-enantiomer.
Q: What is the advantage of the 'active amide' intermediate?
A: The active amide (tetramethyleneimine derivative) significantly enhances the electrophilicity of the carbonyl carbon, facilitating the final Friedel-Crafts acylation with N-methylindole under milder conditions.
Q: Is this process suitable for large-scale continuous production?
A: Yes, the patent highlights that the streamlined steps and improved solvent recovery allow for 'one-pot' continuous operations, making it highly adaptable for industrial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ramosetron Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to meet the evolving demands of the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this novel resolution strategy are fully realized in practice. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Ramosetron Hydrochloride meets the highest international standards. Our commitment to technical excellence allows us to navigate the complexities of chiral synthesis with precision, delivering products that empower our clients to bring life-saving medications to market faster.
We invite potential partners to engage with our technical procurement team to discuss how this optimized route can benefit your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic impact of switching to this more efficient manufacturing process. We are prepared to provide specific COA data and route feasibility assessments to support your decision-making process. Contact us today to explore a partnership that combines cutting-edge chemistry with reliable commercial execution.
