Advanced Catalytic Synthesis of PARP Inhibitor Intermediate for Commercial Scale-Up
Advanced Catalytic Synthesis of PARP Inhibitor Intermediate for Commercial Scale-Up
The pharmaceutical industry is constantly seeking more efficient pathways for the synthesis of complex oncology therapeutics, particularly poly (ADP-ribose) polymerase (PARP) inhibitors. Patent CN111171002B discloses a groundbreaking preparation method for a critical intermediate in the synthesis of Pamiparib, specifically methyl (R)-2-(1-(2-(tert-butyloxy)-2-oxoethyl)-2-methylpyrrolidin-2-yl)-6-fluoro-1-tosyl-1H-indole-4-carboxylate, often referred to as compound BG-11. This technical disclosure represents a significant leap forward in process chemistry, addressing long-standing challenges related to conversion rates and stereochemical integrity. By leveraging a specialized palladium catalyst system comprising Pd(dtbpf)Cl2 and cuprous salts, the invention offers a robust solution for producing high-purity intermediates essential for the development of next-generation cancer therapies. For R&D directors and procurement specialists, understanding the nuances of this catalytic system is vital for securing a reliable pharmaceutical intermediate supplier capable of meeting stringent quality demands.
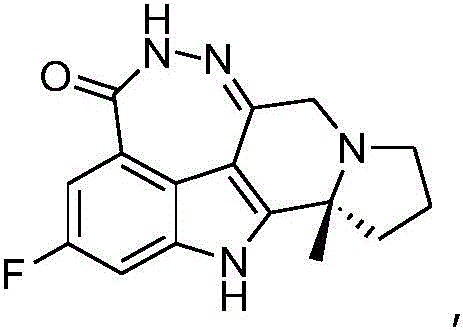
The core innovation lies in the optimization of the indole ring formation via a palladium-catalyzed cyclization. Unlike previous methods that struggled with incomplete reactions and racemization, this novel approach ensures that the chiral center at the pyrrolidine ring remains intact throughout the harsh reaction conditions. The ability to achieve chiral purity exceeding 99.0% directly from the reactor, without the need for subsequent chemical resolution, is a game-changer for cost reduction in API manufacturing. This level of efficiency not only streamlines the production timeline but also drastically reduces the material loss typically associated with purification steps, making it an attractive candidate for commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art processes, such as those disclosed in WO2017/032289A1, relied heavily on catalyst systems like Pd(PPh3)2Cl2 combined with copper iodide. While functional on a small scale, these conventional methods exhibit severe limitations when translated to industrial production. A primary drawback is the poor conversion rate of the starting material, BG-10. Experimental data indicates that using traditional triphenylphosphine-based catalysts can leave upwards of 31.6% of the raw material unreacted, creating a nightmare for downstream purification. Furthermore, these older catalytic systems often fail to maintain the stereochemical integrity of the molecule, leading to partial racemization where the chiral purity drops to unacceptable levels around 90%. This necessitates additional, expensive resolution steps to meet the rigorous specifications required for drug substance manufacturing, thereby inflating costs and extending lead times.
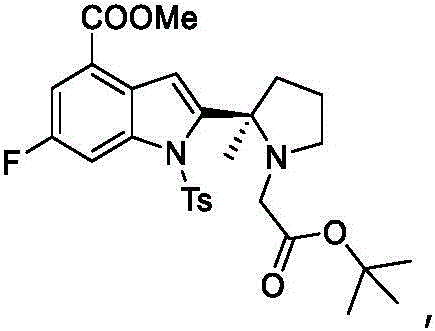
The Novel Approach
The inventive method described in CN111171002B overcomes these hurdles by introducing a more sophisticated ligand environment around the palladium center. By utilizing 1,1'-bis(di-tert-butylphosphino)ferrocene palladium dichloride (Pd(dtbpf)Cl2), the reaction kinetics are dramatically improved. This specific catalyst facilitates a much faster and more complete cyclization, reducing the residual starting material BG-10 to negligible levels, often below 3%. More importantly, the steric bulk and electronic properties of the dtbpf ligand protect the chiral center from epimerization during the high-temperature reaction phase. As a result, the process yields the target intermediate BG-11 with exceptional chiral purity, consistently above 99.0%. This advancement allows manufacturers to bypass resolution entirely, offering a direct path to high-quality material suitable for immediate use in subsequent synthetic steps.
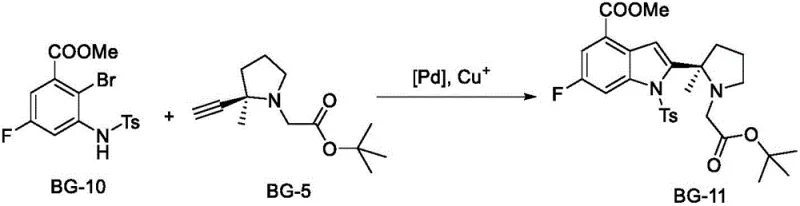
Mechanistic Insights into Palladium-Catalyzed Cyclization
The success of this synthesis hinges on a delicate balance of oxidative addition, alkyne insertion, and reductive elimination within the catalytic cycle. The reaction begins with the activation of the aryl bromide bond in BG-10 by the Pd(0) species generated in situ. This is followed by the coordination and insertion of the terminal alkyne from the BG-5 fragment. A critical mechanistic feature revealed in the patent data is the formation of a transient intermediate, designated as BG-11A. This species represents the open-chain alkyne insertion product prior to the final cyclization that forms the indole core. Monitoring the concentration of BG-11A is crucial, as its persistence indicates incomplete cyclization. The optimized conditions, specifically the use of tetramethylguanidine as a base and maintaining temperatures between 75°C and 80°C, ensure that BG-11A rapidly converts to the final cyclic product BG-11, preventing the accumulation of this key impurity.
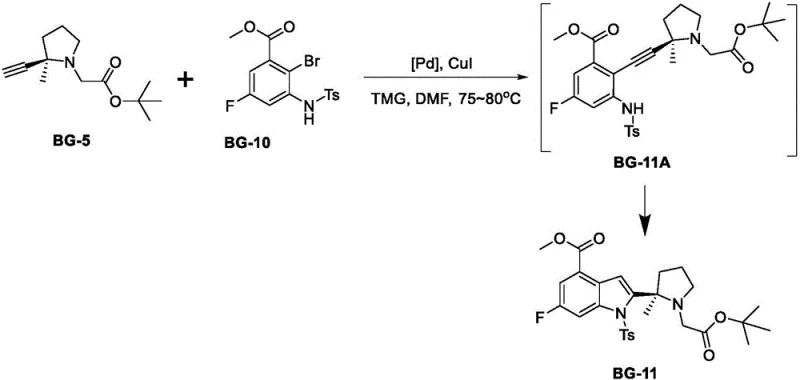
Furthermore, the choice of ligand plays a pivotal role in suppressing side reactions that lead to racemization. The bulky tert-butyl groups on the ferrocene backbone of the dtbpf ligand create a constrained coordination sphere that favors the desired reductive elimination pathway while disfavoring mechanisms that would scramble the stereocenter. This mechanistic control is what differentiates the novel process from earlier attempts using simpler phosphine ligands. By understanding these subtleties, process chemists can better appreciate why this specific catalyst system is superior for maintaining the optical purity required for biologically active PARP inhibitors. The result is a cleaner reaction profile with fewer byproducts, simplifying the isolation procedure and enhancing the overall yield of the process.
How to Synthesize Methyl (R)-2-(1-(2-(tert-butyloxy)-2-oxoethyl)-2-methylpyrrolidin-2-yl)-6-fluoro-1-tosyl-1H-indole-4-carboxylate Efficiently
Executing this synthesis requires careful attention to the preparation of the free base of BG-5 and the precise control of the catalytic coupling conditions. The process begins with the liberation of the BG-5 amine from its salt form using a biphasic system of n-heptane and water, ensuring a dry and reactive nucleophile for the subsequent step. The coupling reaction is then performed in a polar aprotic solvent like DMF, which solubilizes both the organic substrates and the inorganic catalyst components. Maintaining an inert nitrogen atmosphere is essential to prevent oxidation of the sensitive Pd(0) species. Following the reaction, a rigorous workup involving thiourea washing is employed to scavenge residual copper and palladium metals, ensuring the final product meets strict heavy metal specifications. For a detailed breakdown of the standardized operating procedures, please refer to the guide below.
- Preparation of BG-5 Free Base: Suspend BG-5 salt in a mixed solvent of n-heptane and water, treat with an alkaline solution such as potassium hydroxide to liberate the free base, separate the organic phase, and concentrate to obtain a DMF solution.
- Catalytic Coupling Reaction: React BG-10 with the prepared BG-5 free base in an organic solvent like DMF using a palladium catalyst system comprising Pd(dtbpf)Cl2 and cuprous iodide at elevated temperatures (75-80°C) under nitrogen atmosphere.
- Purification and Crystallization: After reaction completion, perform aqueous workup involving thiourea washing to remove metal residues, followed by solvent exchange to methanol and induced crystallization to isolate the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel catalytic process translates into tangible strategic benefits beyond mere chemical yield. The primary advantage lies in the drastic simplification of the manufacturing workflow. By eliminating the need for chiral resolution—a process that typically consumes significant amounts of resolving agents, solvents, and time—the overall production cost is significantly reduced. This efficiency gain allows for a more competitive pricing structure for the final API, providing a buffer against raw material price fluctuations. Additionally, the high conversion rate means that less raw material is wasted, improving the atom economy of the process and aligning with modern green chemistry principles that are increasingly important for regulatory compliance and corporate sustainability goals.
- Cost Reduction in Manufacturing: The elimination of downstream resolution steps removes a major cost center from the production budget. Traditional methods often require multiple crystallization cycles or chromatographic separations to upgrade chiral purity, which are resource-intensive. By achieving >99% chiral purity directly from the reactor, this method saves substantial amounts of solvents and processing time. Furthermore, the high conversion rate minimizes the loss of expensive starting materials, ensuring that every kilogram of input generates maximum output value.
- Enhanced Supply Chain Reliability: The robustness of the Pd(dtbpf)Cl2 catalyst system ensures consistent batch-to-batch quality, which is critical for maintaining a steady supply of clinical and commercial materials. The process uses widely available industrial solvents and reagents, reducing the risk of supply disruptions associated with exotic or specialized chemicals. This reliability allows supply chain planners to forecast production timelines with greater accuracy, reducing lead time for high-purity pharmaceutical intermediates and ensuring that downstream drug manufacturing schedules are met without delay.
- Scalability and Environmental Compliance: The reaction conditions are mild enough to be safely scaled from pilot plants to multi-ton commercial reactors without requiring specialized high-pressure or cryogenic equipment. The use of standard solvents like DMF and methanol facilitates easier solvent recovery and recycling, lowering the environmental footprint of the operation. This scalability ensures that the supply can grow in tandem with the clinical demand for the PARP inhibitor, supporting a seamless transition from Phase III trials to full commercial launch.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a clear picture of what partners can expect when adopting this technology. Understanding these details helps in making informed decisions regarding technology transfer and vendor qualification.
Q: Why is the Pd(dtbpf)Cl2 catalyst preferred over traditional Pd(PPh3)2Cl2 for this synthesis?
A: The use of Pd(dtbpf)Cl2 significantly improves both conversion rates and chiral purity. While traditional catalysts leave substantial unreacted starting material (over 30%) and result in lower optical purity requiring resolution, the dtbpf system achieves near-complete conversion and maintains chiral purity above 99%, eliminating the need for costly downstream resolution steps.
Q: How is the critical intermediate impurity BG-11A managed during production?
A: The process utilizes precise HPLC monitoring to track the transient formation of the alkyne insertion intermediate BG-11A. By optimizing reaction time and temperature (specifically maintaining 75-80°C), the process ensures that BG-11A fully cyclizes into the desired product BG-11, minimizing residual impurities in the final crude mixture.
Q: Is this synthetic route suitable for large-scale commercial manufacturing?
A: Yes, the method is designed for scalability. It employs industrially common solvents like DMF and n-heptane, avoids cryogenic conditions, and utilizes a robust catalyst loading that balances cost with performance. The crystallization protocol using methanol and water is also highly amenable to standard filtration and drying equipment found in GMP facilities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable PARP Inhibitor Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of life-saving oncology drugs. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of BG-11 or related PARP inhibitor intermediates meets the highest international standards. Our commitment to technical excellence allows us to navigate complex synthetic challenges, delivering materials that facilitate your drug development timeline.
We invite you to collaborate with us to leverage this advanced catalytic technology for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized processes can enhance your project's economic viability and operational efficiency.
