Advanced Aqueous Synthesis of Unsymmetrical Bis(indolyl)methanes for Neurodegenerative Drug Development
The pharmaceutical industry is constantly seeking robust, scalable, and environmentally benign methodologies for constructing complex heterocyclic scaffolds, particularly those with proven neuroprotective potential. Patent CN101585799B introduces a groundbreaking approach for the preparation of unsymmetrical bis-indolyl substituted methane compounds, a class of molecules increasingly recognized for their significant physiological activities, including anti-tumor, anti-viral, and notably, anti-Alzheimer's properties. This technical insight report analyzes the novel Indium(III)-catalyzed Friedel-Crafts reaction detailed in the patent, which utilizes water as a green solvent to achieve high yields under mild conditions. For R&D directors and procurement managers alike, this technology represents a pivotal shift away from harsh, organic-solvent-dependent processes toward a more sustainable and cost-efficient manufacturing paradigm. By leveraging the unique Lewis acidity of InX3 (where X is Br, Cl, or OTf), the process enables the efficient coupling of phthalide derivatives with various indoles, overcoming the historical limitations associated with synthesizing unsymmetrical variants of these bioactive cores.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of bis-indolylmethane derivatives has relied heavily on a variety of catalysts such as Montmorillonite K-10, Lithium Perchlorate (LiClO4), Iodine (I2), or Michaelis acid, often necessitating the use of volatile organic compounds (VOCs) as reaction media. These traditional protocols frequently suffer from significant drawbacks, including the requirement for stringent anhydrous conditions, prolonged reaction times, and the generation of substantial chemical waste due to difficult work-up procedures. Furthermore, existing literature predominantly focuses on the formation of symmetrical bis-indolylmethanes, leaving a critical gap in the efficient production of unsymmetrical analogs which are often essential for fine-tuning biological activity and pharmacokinetic profiles. The reliance on moisture-sensitive catalysts also complicates the supply chain, requiring specialized storage and handling that inflates operational costs and increases the risk of batch-to-batch variability in large-scale production environments.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN101585799B employs a highly atom-economical Friedel-Crafts reaction catalyzed by Indium salts in an aqueous medium. This innovative strategy eliminates the need for toxic organic solvents, utilizing water not merely as a passive medium but as a facilitator that enhances the reaction rate through hydrophobic effects. The process operates under remarkably mild conditions, typically ranging from room temperature to 80°C, and achieves conversion rates exceeding 70% with excellent substrate adaptability. By shifting to a water-based system, the method drastically simplifies the isolation of the final product, often requiring only simple extraction and chromatography, thereby reducing the overall process mass intensity (PMI). This transition to aqueous chemistry not only aligns with modern green chemistry principles but also offers a tangible pathway for cost reduction in pharmaceutical manufacturing by minimizing solvent recovery costs and waste disposal fees.
Mechanistic Insights into InX3-Catalyzed Friedel-Crafts Alkylation
The core of this synthetic breakthrough lies in the unique catalytic behavior of Indium(III) species, which act as potent yet water-tolerant Lewis acids. In the reaction mechanism, the InX3 catalyst coordinates with the carbonyl oxygen of the phthalide derivative (substrate 1), thereby increasing the electrophilicity of the adjacent carbon center. This activation facilitates a nucleophilic attack by the C3-position of the indole ring (substrate 2), which is the most electron-rich site on the heterocycle. Unlike traditional Lewis acids like Aluminum Chloride or Boron Trifluoride, which are instantly quenched by water, Indium salts maintain their catalytic activity in aqueous environments, allowing the reaction to proceed smoothly without the need for rigorous drying of reagents. This tolerance is mechanistically attributed to the high oxophilicity of Indium combined with its stability against hydrolysis under the specific reaction conditions employed.
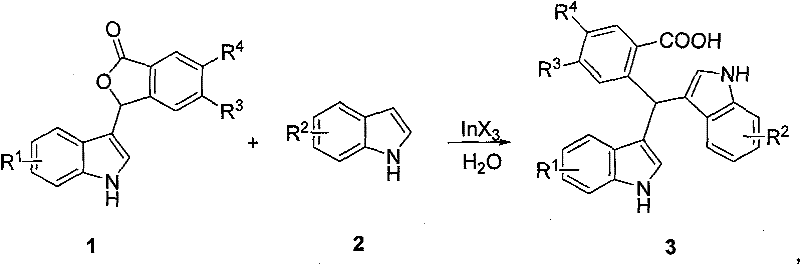
Following the initial alkylation, the intermediate undergoes further transformation to yield the stable unsymmetrical bis(indolyl)methane structure. The presence of water likely assists in the proton transfer steps necessary to regenerate the aromaticity of the indole ring and release the product from the catalyst complex. This mechanistic pathway ensures high regioselectivity, predominantly favoring the 3,3'-disubstituted products observed in the patent examples. The ability to tolerate diverse functional groups on both the indole and phthalide rings—such as methoxy, halogen, nitro, and benzyloxy substituents—demonstrates the robustness of the catalytic cycle. For process chemists, understanding this mechanism is vital for optimizing reaction parameters, as it confirms that the rate-limiting step is likely the electrophilic activation rather than the nucleophilic attack, allowing for fine-tuning of catalyst loading to maximize efficiency without compromising purity.
How to Synthesize Unsymmetrical Bis(indolyl)methane Efficiently
The practical implementation of this synthesis is straightforward and designed for scalability, making it an ideal candidate for technology transfer from the laboratory to pilot plant operations. The standard protocol involves charging a reaction vessel with water and a catalytic amount of InX3 (typically 0.05 equivalents), followed by the addition of the phthalide and indole substrates in a slight molar excess to drive the equilibrium forward. The mixture is then stirred at controlled temperatures between ambient and 80°C for a duration of 5 to 48 hours, depending on the electronic nature of the substituents. Upon completion, the product is isolated via ethyl acetate extraction, dried over anhydrous sodium sulfate, and purified using flash silica gel column chromatography.
- Prepare the reaction vessel by adding water (0.5-500 mL) and the indium catalyst (InX3, 0.01-10 mmol) to a dry flask.
- Introduce the phthalide derivative substrate (0.2-200 mmol) and the indole substrate (0.3-300 mmol) into the aqueous mixture.
- Stir the reaction at temperatures ranging from room temperature to 80°C for 5 to 48 hours, then extract with ethyl acetate and purify via silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this aqueous Indium-catalyzed process offers distinct strategic advantages over conventional organic synthesis routes. The elimination of expensive, flammable, and toxic organic solvents directly translates to lower raw material costs and reduced regulatory burdens associated with VOC emissions. Furthermore, the simplicity of the work-up procedure—relying on basic liquid-liquid extraction rather than complex distillation or crystallization sequences—shortens the overall production cycle time. This efficiency gain is critical for maintaining a reliable supply of high-purity pharmaceutical intermediates in a competitive market where speed-to-market is often the differentiator between success and failure. The robustness of the reaction also implies fewer failed batches, ensuring consistent availability for downstream drug development programs.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the substitution of costly organic solvents with inexpensive tap water, which serves as the reaction medium. Additionally, the low catalyst loading (0.05 equivalents) minimizes the consumption of Indium salts, which, while precious, are used in such small quantities that their impact on the Bill of Materials is negligible compared to solvent savings. The simplified purification process reduces the consumption of silica gel and eluents, further driving down the cost of goods sold (COGS). By removing the need for specialized anhydrous infrastructure, capital expenditure for new production lines is also significantly reduced, offering a clear path to cost reduction in pharmaceutical intermediate manufacturing.
- Enhanced Supply Chain Reliability: Utilizing water as a solvent inherently improves process safety by eliminating fire hazards associated with volatile organic compounds, thereby reducing insurance premiums and safety compliance costs. The mild reaction conditions (up to 80°C) allow for the use of standard glass-lined or stainless steel reactors without the need for exotic materials capable of withstanding extreme temperatures or corrosive acidic environments. This compatibility with standard equipment ensures that the process can be easily integrated into existing manufacturing facilities, mitigating supply chain disruptions caused by equipment bottlenecks. Moreover, the broad substrate scope means that a single platform technology can produce a wide library of derivatives, enhancing supply flexibility for diverse R&D pipelines.
- Scalability and Environmental Compliance: From an environmental perspective, this method aligns perfectly with global sustainability goals, as the aqueous waste stream is far easier to treat than mixed organic waste. The high atom economy of the Friedel-Crafts reaction ensures that the majority of reactant mass is incorporated into the final product, minimizing waste generation at the source. This green profile facilitates smoother regulatory approvals in regions with strict environmental laws, such as the EU and North America. The process has been demonstrated to scale effectively from milligram to multi-gram levels in the patent examples, indicating strong potential for commercial scale-up of complex pharmaceutical intermediates without the typical teething problems associated with solvent exchange or heat transfer issues in viscous organic media.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Indium-catalyzed synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the process capabilities and limitations for potential partners.
Q: What are the primary advantages of using InX3 catalysts over traditional Lewis acids?
A: InX3 catalysts offer superior water tolerance, allowing the reaction to proceed in pure water without organic co-solvents, which significantly simplifies downstream processing and reduces environmental impact compared to moisture-sensitive traditional catalysts.
Q: Can this synthesis method be scaled for industrial production of Alzheimer's drug intermediates?
A: Yes, the method utilizes mild reaction conditions (room temperature to 80°C) and inexpensive, non-toxic solvents (water), making it highly suitable for large-scale commercial manufacturing with minimal safety hazards.
Q: What is the biological significance of the synthesized unsymmetrical bis(indolyl)methanes?
A: Preliminary biological assays indicate that specific derivatives synthesized via this method exhibit potent activity in restoring memory deficits in Drosophila models, suggesting strong potential as therapeutic agents for Alzheimer's disease.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bis(indolyl)methane Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the InX3-catalyzed aqueous synthesis route for producing next-generation neurodegenerative disease therapeutics. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from discovery to market is seamless and efficient. Our state-of-the-art facilities are equipped to handle water-based chemistries with the highest standards of safety and quality, guaranteeing stringent purity specifications for every batch of bis(indolyl)methane intermediates we produce. With our rigorous QC labs and dedicated process development teams, we are uniquely positioned to optimize this green chemistry protocol for your specific API requirements, delivering a reliable supply chain solution that meets the demanding timelines of the global pharmaceutical industry.
We invite you to collaborate with us to leverage this advanced synthetic methodology for your Alzheimer's drug development programs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your project's volume and purity needs. Please contact us today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary targets, and let us help you accelerate your journey from bench to bedside with confidence and efficiency.
