Advanced Rare Earth Catalysis for Scalable Secondary Amine Derivative Manufacturing
Advanced Rare Earth Catalysis for Scalable Secondary Amine Derivative Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to access critical nitrogen-containing scaffolds, particularly secondary amine derivatives which serve as pivotal building blocks in drug discovery and agrochemical development. A significant technological breakthrough in this domain is documented in patent CN110818576B, which details a novel preparation method for secondary amine derivatives synthesized via rare earth catalysis. This innovation represents a paradigm shift from classical reduction methodologies, offering a robust, high-yielding, and environmentally considerate route that addresses long-standing pain points in organic synthesis. By leveraging a specific yttrium-based catalyst system, this technology enables the direct transformation of secondary amides into valuable amines under remarkably mild conditions, eliminating the need for high-pressure hydrogenation or hazardous stoichiometric reducing agents. For R&D directors and procurement strategists, understanding the implications of this patent is crucial for optimizing supply chains and reducing the cost of goods sold for complex intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of secondary amines from carbonyl precursors or amides has relied heavily on classical reducing agents such as sodium borohydride (NaBH4), lithium aluminum hydride (LiAlH4), or diborane (B2H6). While these reagents are chemically effective, they present substantial drawbacks when evaluated through the lens of modern green chemistry and industrial scalability. The primary concern lies in the atom economy and waste generation; these reactions typically require a large excess of the reducing agent to drive completion, resulting in the production of significant quantities of metal salt waste that necessitates costly and complex disposal protocols. Furthermore, reagents like LiAlH4 are pyrophoric and moisture-sensitive, imposing stringent safety requirements on manufacturing facilities and increasing operational overhead. The post-reaction workup is often cumbersome, involving quenching steps that can be dangerous and time-consuming, ultimately extending the production lead time and reducing overall plant throughput efficiency.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN110818576B utilizes a rare earth catalytic system that fundamentally alters the reaction landscape. By employing bistrimethylsilylamino yttrium as a catalyst in conjunction with pinacol borane, the process achieves high conversion rates without the need for stoichiometric metal hydrides. 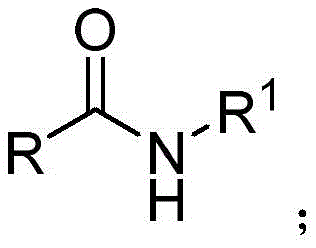 . This approach operates at atmospheric pressure, removing the need for specialized high-pressure autoclaves required for catalytic hydrogenation, thereby lowering capital expenditure barriers for adoption. The reaction conditions are notably mild, typically ranging between 100°C and 140°C, which enhances safety profiles and allows for the use of standard glass-lined or stainless steel reactors commonly found in multipurpose chemical plants. This transition from hazardous stoichiometric reduction to a catalytic cycle signifies a major step forward in process intensification and safety management.
. This approach operates at atmospheric pressure, removing the need for specialized high-pressure autoclaves required for catalytic hydrogenation, thereby lowering capital expenditure barriers for adoption. The reaction conditions are notably mild, typically ranging between 100°C and 140°C, which enhances safety profiles and allows for the use of standard glass-lined or stainless steel reactors commonly found in multipurpose chemical plants. This transition from hazardous stoichiometric reduction to a catalytic cycle signifies a major step forward in process intensification and safety management.
Mechanistic Insights into Rare Earth Catalyzed Amide Reduction
The core of this technological advancement lies in the unique mechanistic pathway facilitated by the yttrium catalyst. Unlike transition metals that might suffer from toxicity issues or difficult removal from the final API, the rare earth catalyst system described demonstrates strong selectivity and facilitates a clean transformation. The reaction initiates with the interaction between the rare earth catalyst and pinacol borane to generate a reactive rare earth hydride intermediate. This active species then inserts into the acyl group of the secondary amide substrate, forming a key intermediate that subsequently reacts with additional borane species. 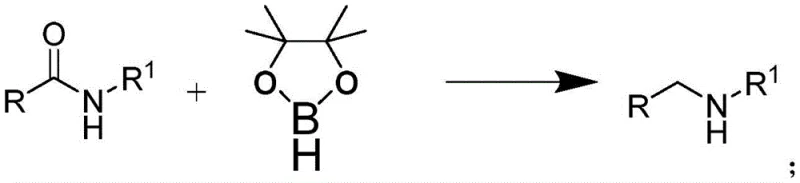 . This catalytic cycle continues through the formation of iminium and imine intermediates, eventually yielding the target amine after hydrolysis. The elegance of this mechanism is its ability to bypass the formation of stable byproducts that typically plague amide reductions, ensuring that the reaction proceeds with high fidelity towards the desired secondary amine structure.
. This catalytic cycle continues through the formation of iminium and imine intermediates, eventually yielding the target amine after hydrolysis. The elegance of this mechanism is its ability to bypass the formation of stable byproducts that typically plague amide reductions, ensuring that the reaction proceeds with high fidelity towards the desired secondary amine structure.
From an impurity control perspective, this mechanism offers distinct advantages for pharmaceutical manufacturing. The high selectivity of the rare earth catalyst minimizes side reactions such as over-reduction or the formation of tertiary amines, which are common impurities in less controlled reduction processes. The use of pinacol borane as the terminal reductant generates boron-containing byproducts that are generally easier to separate from the organic phase compared to the inorganic salts generated by aluminum or lithium hydrides. This inherent cleanliness of the reaction profile translates directly into simplified downstream processing, requiring fewer crystallization or chromatography steps to achieve the stringent purity specifications demanded by global regulatory bodies. For quality assurance teams, this means a more robust and predictable manufacturing process with a lower risk of batch failure due to impurity spikes.
How to Synthesize Secondary Amine Derivatives Efficiently
Implementing this synthesis route requires careful attention to the specific molar ratios and environmental controls outlined in the patent data to ensure optimal performance. The process begins with the preparation of the reaction vessel under an inert atmosphere, typically nitrogen, to prevent catalyst deactivation by moisture or oxygen. The detailed standardized synthesis steps are provided in the guide below. Operators must precisely measure the secondary amide substrate and mix it with pinacol borane in a molar ratio ranging from 1:5 to 1:8, ensuring an excess of the borane reagent to drive the equilibrium forward. The rare earth catalyst is then introduced at a loading of approximately 8% to 15% relative to the substrate, a concentration that balances catalytic activity with cost efficiency. The mixture is heated to the specified range of 100°C to 140°C and maintained for 20 to 25 hours, allowing the catalytic cycle to reach completion before proceeding to isolation.
- Prepare the reaction mixture by combining secondary amide, pinacol borane, and bistrimethylsilylamino yttrium catalyst in toluene or xylene solvent under nitrogen protection.
- Heat the reaction mixture to a temperature between 100°C and 140°C and maintain stirring for 20 to 25 hours to ensure complete conversion.
- Upon completion, isolate the target secondary amine derivative directly without the need for complex intermediate separation, achieving yields up to 90%.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this rare earth catalyzed methodology offers compelling economic and logistical benefits that extend beyond simple yield improvements. The elimination of hazardous, moisture-sensitive reducing agents like LiAlH4 significantly reduces the costs associated with special storage, handling, and disposal of dangerous chemicals. By shifting to a catalytic process that operates at atmospheric pressure, manufacturers can utilize existing standard reactor infrastructure without the need for costly upgrades to high-pressure systems, thereby accelerating the timeline from process development to commercial production. The stability of the raw materials, specifically the secondary amides and pinacol borane, ensures a reliable supply chain with reduced risk of raw material degradation during transport or storage, contributing to greater overall supply continuity.
- Cost Reduction in Manufacturing: The transition to a catalytic system inherently reduces the consumption of expensive reagents per kilogram of product produced. By avoiding the use of stoichiometric amounts of metal hydrides, the direct material costs are significantly lowered, and the burden on waste treatment facilities is drastically reduced. The simplified workup procedure, which does not require complex quenching of reactive metals, leads to substantial savings in labor hours and utility consumption during the production cycle. Furthermore, the high selectivity of the reaction minimizes the loss of valuable starting materials to byproducts, improving the overall mass balance and economic efficiency of the manufacturing process.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, including various substituted secondary amides and pinacol borane, are commercially available from multiple global suppliers, reducing dependency on single-source vendors. The robustness of the reaction conditions, which tolerate a wide range of functional groups as demonstrated by the substrate scope in the patent, allows for flexibility in sourcing different grade starting materials without compromising final product quality. This flexibility is critical for maintaining production schedules in the face of market fluctuations or raw material shortages, ensuring that delivery commitments to downstream pharmaceutical customers are consistently met.
- Scalability and Environmental Compliance: Operating at atmospheric pressure and moderate temperatures makes this process inherently safer and easier to scale from pilot plant to multi-ton commercial production. The reduction in hazardous waste generation aligns with increasingly stringent environmental regulations, lowering the compliance costs and potential liabilities associated with chemical manufacturing. The use of less toxic reagents and the generation of fewer pollutant emissions contribute to a greener manufacturing footprint, which is becoming a key differentiator for suppliers seeking to partner with sustainability-focused multinational corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this rare earth catalyzed synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this method into their existing manufacturing portfolios for pharmaceutical intermediates and fine chemicals.
Q: What are the advantages of using rare earth catalysts over traditional reducing agents?
A: Unlike traditional reducing agents like LiAlH4 which generate significant metal salt waste and require harsh conditions, the rare earth catalyzed method operates at atmospheric pressure with high atom economy and reduced hazardous waste.
Q: Is this synthesis method suitable for large-scale production?
A: Yes, the process uses stable raw materials, operates at normal pressure, and avoids sensitive organosilicon reagents, making it highly suitable for commercial scale-up and continuous manufacturing.
Q: What is the expected yield and purity profile?
A: The patent data indicates yields reaching up to 90% with strong reaction selectivity, minimizing byproduct formation and simplifying downstream purification processes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Secondary Amine Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the one described in CN110818576B for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of secondary amine derivatives meets the exacting standards required for drug substance manufacturing. We are committed to leveraging our technical expertise to optimize these rare earth catalyzed routes for maximum efficiency and cost-effectiveness.
We invite R&D directors and procurement leaders to collaborate with us to explore how this technology can enhance your supply chain resilience and reduce overall manufacturing costs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific project needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities align with your strategic goals for high-purity pharmaceutical intermediates.
