Advanced Manufacturing of Pimavanserin Intermediates via Safe Carbon Dioxide Fixation Technology
Advanced Manufacturing of Pimavanserin Intermediates via Safe Carbon Dioxide Fixation Technology
The pharmaceutical industry is constantly seeking robust, scalable, and safe synthetic routes for complex active pharmaceutical ingredients (APIs), particularly for neurological treatments where purity is paramount. Pimavanserin, a selective inverse agonist of the serotonin 5-HT2A receptor, represents a critical therapy for treating hallucinations and delusions associated with Parkinson’s disease psychosis. The structural complexity of this molecule, specifically its hemitartrate salt form shown in Formula I, demands a synthesis strategy that balances high yield with stringent impurity control. 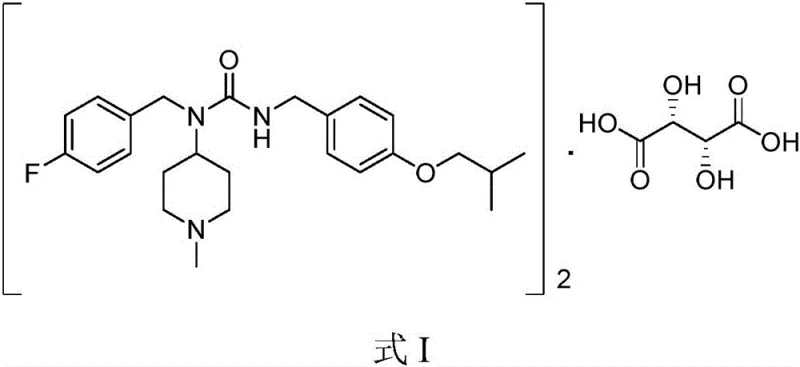 Recent advancements detailed in patent CN110894186B introduce a groundbreaking preparation method that leverages carbon dioxide fixation to construct the core urea linkage. This innovation addresses long-standing safety and purity challenges, offering a viable pathway for reliable pharmaceutical intermediate supplier networks to enhance their production capabilities while adhering to green chemistry principles.
Recent advancements detailed in patent CN110894186B introduce a groundbreaking preparation method that leverages carbon dioxide fixation to construct the core urea linkage. This innovation addresses long-standing safety and purity challenges, offering a viable pathway for reliable pharmaceutical intermediate supplier networks to enhance their production capabilities while adhering to green chemistry principles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of pimavanserin and its key urea intermediates has relied on hazardous reagents and multi-step sequences that pose significant risks to both operational safety and product quality. Early methodologies, such as those disclosed in WO2006036874A1, utilized virulent phosgene gas to generate isocyanate intermediates, necessitating specialized containment equipment and creating severe safety liabilities for manufacturing facilities. 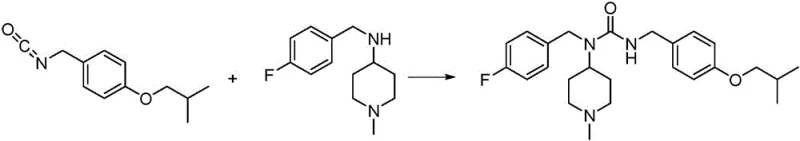 Alternative approaches attempted to mitigate phosgene risks through Curtius rearrangement using diphenyl phosphoryl azide (DPPA), but this reagent is prohibitively expensive and generates stoichiometric amounts of phosphorus waste, making it unsuitable for cost reduction in pharmaceutical manufacturing. Furthermore, routes employing carbonyldiimidazole (CDI), as seen in patents like CN105820110A and CN105153016A, suffer from persistent impurity issues; specifically, the formation of Imidazole residues and a specific urea byproduct known as Impurity 1 is prevalent.
Alternative approaches attempted to mitigate phosgene risks through Curtius rearrangement using diphenyl phosphoryl azide (DPPA), but this reagent is prohibitively expensive and generates stoichiometric amounts of phosphorus waste, making it unsuitable for cost reduction in pharmaceutical manufacturing. Furthermore, routes employing carbonyldiimidazole (CDI), as seen in patents like CN105820110A and CN105153016A, suffer from persistent impurity issues; specifically, the formation of Imidazole residues and a specific urea byproduct known as Impurity 1 is prevalent. 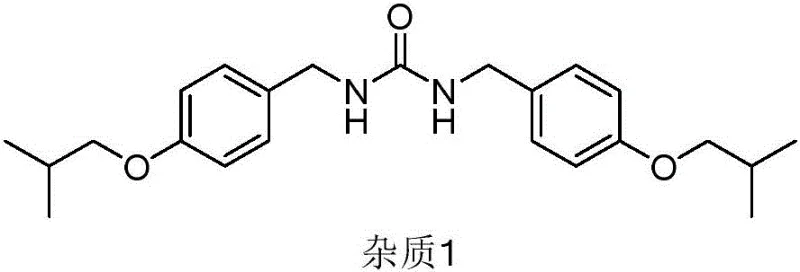 Literature indicates that these traditional CDI-mediated pathways often result in Impurity 1 levels ranging from 8% to 15%, which are notoriously difficult to remove during purification, thereby compromising the overall yield and requiring extensive downstream processing.
Literature indicates that these traditional CDI-mediated pathways often result in Impurity 1 levels ranging from 8% to 15%, which are notoriously difficult to remove during purification, thereby compromising the overall yield and requiring extensive downstream processing.
The Novel Approach
In stark contrast to these legacy methods, the technology protected under CN110894186B introduces a streamlined, one-pot strategy for generating the critical carbamate intermediate using carbon dioxide as a C1 building block. This novel approach reacts 4-isobutoxybenzylamine (Compound M) directly with an alkyl halide (Compound SM) and CO2 in the presence of a base to yield the stable carbamate (Compound A). 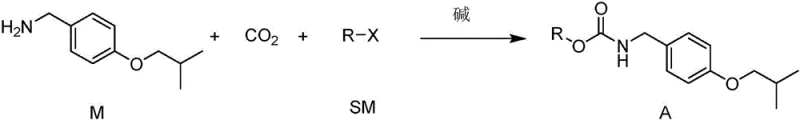 This transformation occurs under mild conditions, typically between 0°C and 50°C, avoiding the extreme temperatures and pressures associated with phosgene chemistry. By bypassing the formation of free isocyanates and avoiding imidazole-based activators entirely, this method fundamentally alters the impurity profile of the synthesis. The result is a process that is not only safer for operators but also inherently cleaner, producing intermediates with negligible levels of the problematic Impurity 1, thus facilitating easier purification and higher overall throughput for commercial scale-up of complex pharmaceutical intermediates.
This transformation occurs under mild conditions, typically between 0°C and 50°C, avoiding the extreme temperatures and pressures associated with phosgene chemistry. By bypassing the formation of free isocyanates and avoiding imidazole-based activators entirely, this method fundamentally alters the impurity profile of the synthesis. The result is a process that is not only safer for operators but also inherently cleaner, producing intermediates with negligible levels of the problematic Impurity 1, thus facilitating easier purification and higher overall throughput for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into CO2-Mediated Carbamate Formation
The core innovation of this synthesis lies in the efficient fixation of carbon dioxide onto the primary amine of Compound M to form a carbamate salt, which is subsequently trapped by an alkyl halide. Mechanistically, the reaction initiates with the nucleophilic attack of the amine nitrogen on the electrophilic carbon of CO2, facilitated by a strong organic base such as DBU or an inorganic carbonate like potassium carbonate. This generates a zwitterionic carbamate species that is stabilized in the polar aprotic solvent medium. The addition of the alkyl halide (where X is Cl, Br, or I) then triggers an SN2 substitution, alkylating the carbamate oxygen to lock in the protecting group and form the stable N-alkoxycarbonyl derivative (Compound A). This mechanism is highly advantageous because it utilizes CO2, an abundant and non-toxic gas, effectively turning a greenhouse gas into a valuable synthetic reagent while avoiding the generation of hazardous leaving groups associated with chloroformates.
Following the formation of Intermediate A, the synthesis proceeds to the final urea assembly through a transurethanization-like coupling with the piperidine amine (Compound N). 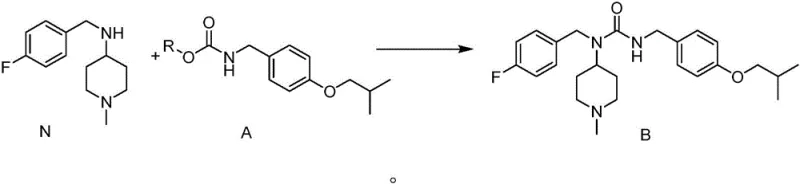 In this step, a strong base such as potassium tert-butoxide or sodium hydride deprotonates the piperidine nitrogen, enhancing its nucleophilicity to attack the carbonyl carbon of the carbamate. This displaces the alkoxide leaving group (RO-) and forms the desired unsymmetrical urea linkage found in Pimavanserin (Compound B). The choice of base and solvent in this step is critical; the patent specifies conditions (reflux in toluene or THF) that drive the equilibrium toward the urea product while minimizing side reactions. This two-step sequence (Carbamate formation -> Coupling) ensures that the highly reactive isocyanate species is never generated in bulk, significantly reducing the risk of polymerization or hydrolysis side products that typically plague direct isocyanate-amine couplings.
In this step, a strong base such as potassium tert-butoxide or sodium hydride deprotonates the piperidine nitrogen, enhancing its nucleophilicity to attack the carbonyl carbon of the carbamate. This displaces the alkoxide leaving group (RO-) and forms the desired unsymmetrical urea linkage found in Pimavanserin (Compound B). The choice of base and solvent in this step is critical; the patent specifies conditions (reflux in toluene or THF) that drive the equilibrium toward the urea product while minimizing side reactions. This two-step sequence (Carbamate formation -> Coupling) ensures that the highly reactive isocyanate species is never generated in bulk, significantly reducing the risk of polymerization or hydrolysis side products that typically plague direct isocyanate-amine couplings.
How to Synthesize Pimavanserin Efficiently
The implementation of this CO2 fixation technology offers a practical and robust protocol for laboratories and pilot plants aiming to produce high-purity pimavanserin intermediates. The process begins with the careful control of CO2 pressure and base stoichiometry to ensure complete conversion of the starting amine, followed by a straightforward aqueous workup to isolate the carbamate. Detailed standard operating procedures regarding specific molar ratios, solvent choices, and temperature ramps are essential to replicate the high yields reported in the patent examples. For process chemists looking to adopt this methodology, the following guide outlines the critical operational phases required to achieve optimal results.
- React 4-isobutoxybenzylamine (Compound M) with an alkyl halide (SM) and carbon dioxide in the presence of a base to form the carbamate intermediate (Compound A).
- Couple the carbamate intermediate (Compound A) with N-(4-fluorobenzyl)-1-methyl-4-piperidinamine (Compound N) under basic conditions to generate the free base Pimavanserin (Compound B).
- Perform salt formation by reacting Pimavanserin (Compound B) with tartaric acid in an alcoholic solvent to obtain the final Pimavanserin Hemitartrate (Compound I).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the shift from phosgene or CDI-based chemistry to this CO2 fixation route represents a strategic opportunity to optimize both cost structures and supply reliability. Traditional methods rely on reagents that are either heavily regulated due to toxicity (phosgene, chloroformates) or subject to volatile pricing and supply constraints (DPPA, CDI). By transitioning to a process that utilizes commodity chemicals like carbon dioxide, simple alkyl halides, and commercially available amines, manufacturers can decouple their production costs from the fluctuations of specialty reagent markets. This stability is crucial for maintaining consistent margins in the competitive generic and branded pharmaceutical sectors.
- Cost Reduction in Manufacturing: The elimination of expensive coupling agents like DPPA and the avoidance of toxic phosgene infrastructure leads to substantial cost savings in raw material procurement and capital expenditure. Furthermore, the high selectivity of the CO2 fixation reaction minimizes the formation of hard-to-remove impurities, which reduces the consumption of solvents and silica gel during purification steps. This efficiency translates directly into a lower cost of goods sold (COGS) without compromising the quality of the final API intermediate.
- Enhanced Supply Chain Reliability: Relying on widely available starting materials such as 4-isobutoxybenzylamine and simple alkyl halides mitigates the risk of supply disruptions often associated with specialized hazardous reagents. The robustness of the reaction conditions, which tolerate mild temperatures and standard pressure, allows for flexible scheduling and easier integration into existing multipurpose manufacturing suites. This flexibility ensures a more reliable supply of high-purity pharmaceutical intermediates, enabling downstream API manufacturers to meet their production targets consistently.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, replacing virulent phosgene with carbon dioxide significantly lowers the regulatory burden and safety risks associated with production. The process generates less hazardous waste and avoids the creation of imidazole-containing byproducts that require complex disposal protocols. This alignment with green chemistry principles facilitates smoother regulatory approvals and supports the sustainability goals of modern pharmaceutical supply chains, making it easier to scale from pilot batches to multi-ton commercial production.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this new synthetic route is essential for stakeholders evaluating its adoption for commercial manufacturing. The following questions address common concerns regarding impurity control, scalability, and the specific advantages of the CO2 fixation methodology over traditional approaches. These insights are derived directly from the experimental data and technical disclosures within the patent literature.
Q: How does this new CO2 fixation method improve impurity profiles compared to CDI routes?
A: Traditional routes using carbonyldiimidazole (CDI) often result in difficult-to-remove imidazole impurities and specific urea byproducts (Impurity 1) at levels of 8-15%. The novel CO2 fixation method described in patent CN110894186B completely avoids imidazole reagents, resulting in a cleaner reaction profile with negligible Impurity 1 content.
Q: Is the carbon dioxide fixation process scalable for industrial production?
A: Yes, the process is highly scalable. It utilizes mild reaction conditions (0-50°C for the fixation step) and common organic solvents like DMF or dioxane. The use of gaseous or solid CO2 eliminates the need for handling hazardous phosgene gas, significantly simplifying equipment requirements and safety protocols for large-scale manufacturing.
Q: What are the cost advantages of using alkyl halides over isocyanates?
A: The new route replaces expensive and unstable isocyanate precursors or costly coupling agents like DPPA with inexpensive alkyl halides (e.g., bromopropane, iodopropane) and carbon dioxide. This substitution drastically reduces raw material costs and simplifies the supply chain by relying on commodity chemicals.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pimavanserin Intermediate Supplier
As the demand for safer and more efficient neurological therapies grows, the ability to manufacture key intermediates like those for Pimavanserin with high purity and consistency becomes a critical competitive advantage. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, leveraging advanced synthetic technologies such as the CO2 fixation route to deliver superior quality intermediates. With extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, our team ensures that every batch meets stringent purity specifications through our rigorous QC labs and state-of-the-art analytical capabilities.
We invite global partners to collaborate with us to optimize their supply chains and reduce manufacturing costs through innovative chemistry. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our advanced manufacturing capabilities can support your long-term production goals.
