Advanced Catalytic Synthesis of 4-Polyfluoroalkyl-2,4-Disubstituted Pyrrole Derivatives for Commercial Scale-up
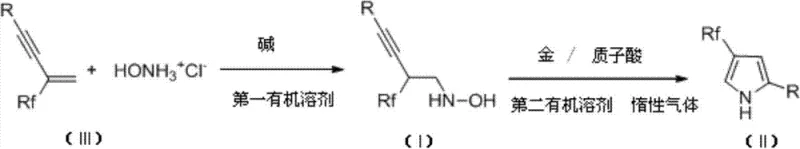
The pharmaceutical and agrochemical industries are constantly seeking robust methodologies to access fluorinated heterocycles, given their profound impact on metabolic stability and bioavailability. Patent CN103755587A introduces a groundbreaking approach for the preparation of 4-polyfluoroalkyl-2,4-disubstituted pyrrole derivatives, a structural motif highly valued in modern drug discovery. This technology leverages a sophisticated dual-catalytic system involving gold complexes and protonic acids to achieve cyclization under remarkably mild conditions. By utilizing fluorine-containing conjugated enynes and hydroxylamine hydrochloride as primary building blocks, the process circumvents the limitations of classical heterocyclic synthesis. For R&D directors and procurement specialists alike, this represents a significant advancement in the reliable supply of high-purity pharmaceutical intermediates, offering a pathway to complex molecular architectures that was previously difficult to navigate efficiently.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trifluoromethyl-substituted pyrroles has been fraught with synthetic challenges that hinder large-scale production. Traditional strategies often rely on the condensation of 1,4-dicarbonyl precursors via the Paal-Knorr reaction, which necessitates multi-step sequences to generate the requisite diketones, thereby reducing overall atom economy. Alternative approaches involving 1,3-dipolar cycloadditions or the use of electron-deficient olefins with isocyanide ylides frequently suffer from poor regioselectivity and limited substrate scope. Furthermore, some contemporary methods depend on unstable enolate substrates and require cryogenic conditions with expensive organolithium reagents like butyllithium, posing significant safety hazards and cost barriers for industrial application. These constraints severely limit the accessibility of diverse fluorinated pyrrole libraries needed for comprehensive structure-activity relationship studies.
The Novel Approach
In stark contrast, the methodology disclosed in CN103755587A offers a streamlined, two-step sequence that dramatically simplifies the construction of the pyrrole core. The process initiates with the formation of a fluorine-containing alkynyl hydroxylamine intermediate under basic conditions, followed by a gold-catalyzed cyclization that proceeds efficiently at room temperature. This novel route eliminates the need for cryogenic operations and highly reactive organometallics, replacing them with stable, commercially available reagents. The use of a gold catalyst in conjunction with a protonic acid allows for precise activation of the alkyne moiety, facilitating ring closure with exceptional chemoselectivity. This shift not only enhances operational safety but also significantly reduces the complexity of the manufacturing process, making it an ideal candidate for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Gold-Catalyzed Cyclization
The core innovation of this technology lies in the synergistic action of the gold catalyst and the protonic acid during the cyclization step. The gold species, typically a cationic gold complex such as IPrAuNTf2 or gold trichloride, acts as a potent pi-acid to activate the carbon-carbon triple bond of the hydroxylamine intermediate. This activation increases the electrophilicity of the alkyne, rendering it susceptible to nucleophilic attack by the proximal nitrogen atom of the hydroxylamine group. Simultaneously, the protonic acid, such as HNTf2 or methanesulfonic acid, assists in the protonation of the hydroxyl group, facilitating the elimination of water or rearrangement necessary for aromatization. This dual-catalytic mechanism ensures that the reaction proceeds through a low-energy pathway, avoiding the formation of polymeric byproducts often seen in harsher acidic conditions.
From an impurity control perspective, the mildness of the reaction conditions is paramount. By operating at room temperature rather than elevated heats, the process minimizes thermal degradation of sensitive functional groups such as esters, nitriles, and nitro groups, which are frequently present in advanced intermediates. The high chemoselectivity of the gold catalyst ensures that side reactions, such as polymerization of the enyne or over-oxidation, are suppressed. This results in a cleaner crude reaction profile, which simplifies downstream purification via column chromatography. For quality assurance teams, this translates to a more consistent impurity profile and higher batch-to-batch reproducibility, critical factors when scaling up the commercial production of complex pharmaceutical intermediates.
How to Synthesize 4-Polyfluoroalkyl-2,4-Disubstituted Pyrrole Derivatives Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production. The process begins with the preparation of the key hydroxylamine intermediate by reacting a fluorinated conjugated enyne with hydroxylamine hydrochloride in a solvent like dichloromethane, using a base such as triethylamine to scavenge the generated acid. Once the intermediate is isolated and purified, it is subjected to the cyclization conditions in a polar aprotic solvent like DMF or acetonitrile. The detailed standardized synthesis steps below outline the specific molar ratios, solvent choices, and workup procedures required to achieve the high yields reported in the patent examples.
- React fluorine-containing conjugated enyne with hydroxylamine hydrochloride and a base in an organic solvent at 0°C to room temperature to form the hydroxylamine intermediate.
- Purify the intermediate via column chromatography using petroleum ether and ethyl acetate.
- Dissolve the intermediate in a second organic solvent and add a gold catalyst and protonic acid catalyst to induce cyclization at room temperature, yielding the final pyrrole derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible strategic benefits beyond mere chemical elegance. The reliance on commercially available starting materials, such as fluorinated enynes and hydroxylamine salts, mitigates the risk of supply chain disruptions associated with custom-synthesized or exotic reagents. Furthermore, the elimination of hazardous reagents like butyllithium reduces the regulatory burden and safety infrastructure costs required for manufacturing facilities. The ability to run reactions at room temperature also leads to substantial energy savings compared to processes requiring prolonged heating or cryogenic cooling, contributing to a more sustainable and cost-effective production model.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by removing the need for expensive transition metal removal steps often associated with palladium or nickel catalysis, as gold loading can be kept low while maintaining high efficiency. Additionally, the high yields observed across a broad range of substrates mean less raw material waste and lower cost per kilogram of the final active pharmaceutical ingredient precursor. The simplified workup procedure, which often involves direct column chromatography without complex extractions, further reduces labor and solvent consumption costs.
- Enhanced Supply Chain Reliability: By utilizing robust and stable intermediates, the manufacturing timeline is shortened, reducing lead time for high-purity pharmaceutical intermediates. The tolerance for various functional groups means that a single platform technology can be used to produce a wide library of derivatives, allowing for flexible inventory management and rapid response to changing market demands. This versatility ensures a steady supply of diverse building blocks for drug development pipelines without the need for multiple distinct synthetic routes.
- Scalability and Environmental Compliance: The mild reaction conditions and use of standard organic solvents facilitate easier scale-up from gram to ton quantities without significant re-engineering of the process. The reduction in hazardous waste generation, particularly the avoidance of heavy metal contaminants and reactive organolithium byproducts, aligns with increasingly stringent environmental regulations. This compliance not only avoids potential fines but also enhances the corporate sustainability profile, a key metric for modern supply chain evaluations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this gold-catalyzed pyrrole synthesis. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the feasibility and advantages of adopting this technology for your specific application needs.
Q: What are the advantages of this gold-catalyzed method over traditional Paal-Knorr synthesis?
A: Unlike traditional methods that often require harsh conditions or unstable precursors like 1,4-dicarbonyls, this gold-catalyzed route operates under mild room temperature conditions with high chemoselectivity and avoids expensive reagents like butyllithium.
Q: Is this process scalable for industrial production of pharmaceutical intermediates?
A: Yes, the process utilizes commercially available raw materials and standard organic solvents like dichloromethane and DMF. The mild reaction temperatures and high yields demonstrated in the patent examples indicate strong potential for commercial scale-up.
Q: What types of substituents are compatible with this synthesis route?
A: The method exhibits excellent functional group tolerance, accommodating alkyl, alkenyl, aryl, heteroaryl, ester, and amide groups, making it versatile for synthesizing diverse fluorinated pyrrole libraries.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Polyfluoroalkyl-2,4-Disubstituted Pyrrole Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced heterocyclic intermediates play in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless. We are equipped with rigorous QC labs and adhere to stringent purity specifications to guarantee that every batch of 4-polyfluoroalkyl-2,4-disubstituted pyrrole derivatives meets the highest international standards. Our commitment to technical excellence allows us to navigate the complexities of fluorinated chemistry with precision and reliability.
We invite you to collaborate with us to leverage this innovative synthetic technology for your projects. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can accelerate your development timeline and optimize your supply chain efficiency.
