Revolutionizing Prozac Intermediate Production: A Green Photochemical Catalysis Approach for Commercial Scale-Up
Revolutionizing Prozac Intermediate Production: A Green Photochemical Catalysis Approach for Commercial Scale-Up
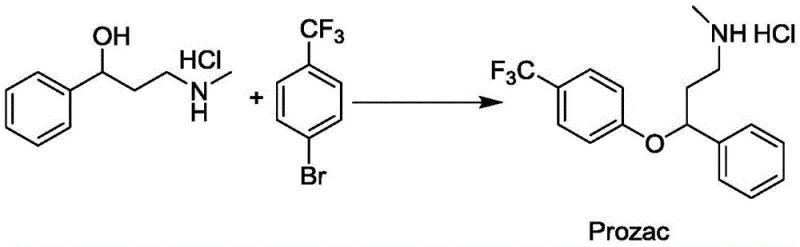
The pharmaceutical industry is currently undergoing a paradigm shift towards sustainable manufacturing, driven by the urgent need to reduce environmental footprints while maintaining high efficiency in the production of active pharmaceutical ingredients (APIs). A groundbreaking development in this sector is detailed in patent CN111423331B, which discloses a novel photochemical catalytic synthesis method for aryl alkyl ether compounds. This technology represents a significant leap forward from traditional stoichiometric methods, offering a robust pathway for synthesizing critical drug intermediates, including the antidepressant Fluoxetine, commonly known as Prozac. By leveraging the synergistic power of heterogeneous titanium dioxide (TiO2) photocatalysis and nickel co-catalysis under light irradiation, this method achieves exceptional conversion rates with minimal environmental impact. For R&D directors and supply chain strategists, understanding the implications of this patent is crucial for future-proofing production lines against tightening regulatory pressures and rising raw material costs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the aryl alkyl ether motif, a ubiquitous structural fragment in medicinal chemistry, has relied heavily on classical methodologies such as the Williamson ether synthesis, Ullmann coupling, and Mitsunobu reactions. While these methods are well-established in academic literature, they present substantial drawbacks when applied to modern industrial scale-up. The Williamson ether synthesis, for instance, typically necessitates the use of strong bases and alkyl halides, often leading to competitive elimination reactions and poor atom economy. Furthermore, transition-metal catalyzed approaches like Ullmann coupling frequently employ expensive and toxic palladium or copper catalysts that require rigorous removal steps to meet stringent pharmaceutical purity standards. These conventional processes are not only energy-intensive due to high-temperature requirements but also generate significant quantities of hazardous waste, complicating disposal and increasing the overall cost of goods sold (COGS) for manufacturers.
The Novel Approach
In stark contrast, the methodology outlined in patent CN111423331B introduces a transformative green chemistry solution that circumvents these historical bottlenecks. This novel approach utilizes a dual catalytic system comprising a semiconductor photocatalyst (TiO2) and a nickel-based co-catalyst to facilitate the C-O bond coupling between halogenated aromatic hydrocarbons and fatty alcohols. The reaction proceeds under mild conditions, typically at ambient temperatures ranging from 10°C to 50°C, driven solely by light energy from sources such as Xenon lamps or 395nm LEDs. This eliminates the need for thermal activation, thereby reducing energy consumption and enhancing process safety. Moreover, the use of TiO2, a material renowned for its low biotoxicity, chemical stability, and abundance, ensures that the catalytic system is both economically viable and environmentally benign. The ability to achieve GC yields as high as 90% under these gentle conditions underscores the efficiency and selectivity of this photochemical protocol.
Mechanistic Insights into TiO2-Ni Dual Photocatalytic C-O Coupling
To fully appreciate the technical sophistication of this invention, one must delve into the mechanistic intricacies of the TiO2-Ni dual catalytic cycle. Upon irradiation with light of appropriate wavelength, the TiO2 semiconductor undergoes charge separation, generating electron-hole pairs within its conduction and valence bands. These photogenerated charge carriers are pivotal in activating the substrate molecules; specifically, the electrons can reduce the nickel cocatalyst to a lower oxidation state capable of undergoing oxidative addition with the aryl halide. Simultaneously, the holes or secondary radical species generated at the TiO2 surface may assist in the activation of the alcohol nucleophile or the regeneration of the catalytic species. This cooperative mechanism ensures that the reaction proceeds through a controlled radical or organometallic pathway, rather than the uncontrolled free-radical processes often seen in non-selective photocatalysis. The presence of specific ligands, such as bipyridine derivatives, further stabilizes the nickel center, preventing catalyst deactivation and promoting the desired reductive elimination step to form the C-O bond.
From an impurity control perspective, this mechanism offers distinct advantages over thermal methods. Traditional high-temperature couplings often suffer from thermal degradation of sensitive functional groups or non-selective side reactions like dehalogenation. However, the photochemical activation provided by the TiO2 system is highly specific, targeting the halogen-carbon bond without affecting other susceptible moieties within the complex molecular architecture of drug intermediates. The heterogeneous nature of the TiO2 catalyst also plays a critical role in purity profiles; since the photocatalyst exists as a solid suspension, it can be physically separated from the reaction mixture via simple filtration or centrifugation post-reaction. This physical separation prevents metal leaching into the final product, a common issue with homogeneous palladium catalysts, thereby simplifying the downstream purification process and ensuring the final API intermediate meets rigorous quality specifications without the need for expensive scavenging resins.
How to Synthesize Prozac Intermediate Efficiently
The practical implementation of this photochemical strategy for the synthesis of high-value intermediates like Prozac involves a streamlined operational protocol that balances reaction efficiency with ease of handling. The process begins with the precise formulation of the reaction mixture, where the halogenated aromatic precursor and the alcohol substrate are combined with the nickel cocatalyst, ligand, and an inorganic base in a suitable solvent system. The selection of the solvent is flexible, ranging from polar aprotic solvents like DMF and DMSO to the alcohol substrate itself acting as the solvent, providing versatility for different solubility profiles. Once the homogeneous components are mixed, the heterogeneous TiO2 photocatalyst is introduced, and the system is purged with an inert gas to prevent oxygen quenching of the excited states. Detailed standardized synthetic steps for optimizing this reaction are provided in the guide below.
- Prepare the reaction system by mixing the halogenated aromatic hydrocarbon (e.g., p-trifluoromethylbromobenzene), fatty alcohol, Ni cocatalyst, ligand, and inorganic base in an inert organic solvent or excess alcohol.
- Add the heterogeneous TiO2 photocatalyst (e.g., P25, Anatase, or Rutile) to the transparent reactor and purge with inert gas (Nitrogen or Argon) to establish an oxygen-free environment.
- Irradiate the mixture with a Xenon lamp or 395nm LED light source at ambient temperature (10-50°C) for 8-20 hours, then separate the product via column chromatography and recover the TiO2 catalyst by centrifugation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this photochemical technology translates into tangible strategic benefits that extend beyond mere technical feasibility. The shift from precious metal catalysts to earth-abundant materials fundamentally alters the cost structure of the manufacturing process. By eliminating the dependency on volatile palladium markets and removing the need for complex metal removal units, manufacturers can achieve significant cost reductions in pharmaceutical intermediate manufacturing. Furthermore, the mild reaction conditions enhance operational safety, reducing the risk of thermal runaways and allowing for the use of simpler, less expensive reactor materials. This robustness ensures a more reliable supply chain, as the process is less susceptible to disruptions caused by utility failures or strict safety shutdowns often associated with high-pressure or high-temperature chemistry.
- Cost Reduction in Manufacturing: The economic argument for this technology is compelling, primarily driven by the substitution of expensive homogeneous catalysts with inexpensive, heterogeneous TiO2. Unlike palladium or rhodium catalysts which represent a significant portion of raw material costs and require costly recovery processes, TiO2 is abundant and cheap. Additionally, the patent data indicates that the catalyst can be recycled and reused multiple times without significant loss in activity, which drastically lowers the recurring cost of catalyst consumption. The elimination of stoichiometric reagents found in traditional methods like the Mitsunobu reaction further reduces waste disposal costs, contributing to a leaner and more profitable production model.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the scarcity of specialized reagents and the complexity of multi-step syntheses. This photochemical method simplifies the supply chain by utilizing readily available starting materials such as commercially sourced halogenated aromatics and common alcohols. The reaction's tolerance to various functional groups means that fewer protection and deprotection steps are required, shortening the overall synthesis timeline. Moreover, the use of standard light sources like LEDs ensures that the energy input is consistent and easily scalable, removing the bottleneck of specialized heating equipment and allowing for rapid ramp-up of production capacity to meet market demand fluctuations.
- Scalability and Environmental Compliance: As global regulations on chemical emissions become increasingly stringent, the environmental profile of a manufacturing process is a critical factor for long-term viability. This photochemical protocol aligns perfectly with green chemistry principles by operating at ambient temperatures and generating minimal hazardous waste. The heterogeneous catalyst system facilitates easy separation, reducing the load on wastewater treatment facilities. From a scalability standpoint, the reaction can be adapted to continuous flow photoreactors, which offer superior light penetration and heat transfer compared to batch processes. This adaptability ensures that the process can be seamlessly scaled from pilot plant quantities to multi-ton commercial production while maintaining high purity and yield standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photochemical synthesis method. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear understanding of the technology's capabilities and limitations for potential adopters.
Q: What are the primary advantages of using TiO2 photocatalysis over traditional Williamson ether synthesis?
A: Unlike the Williamson ether synthesis which often requires harsh basic conditions and stoichiometric reagents generating significant waste, the TiO2 photocatalytic method operates under mild, neutral conditions. Furthermore, TiO2 is a heterogeneous catalyst that can be easily separated via centrifugation and recycled multiple times without loss of activity, drastically reducing material costs and environmental impact.
Q: Can this photochemical method be scaled for industrial production of Prozac intermediates?
A: Yes, the process is highly scalable. The use of inexpensive light sources such as 395nm LEDs or Xenon lamps, combined with ambient temperature operation (25°C), eliminates the need for complex heating or cooling infrastructure. The robustness of the TiO2 catalyst allows for continuous flow processing or large batch reactors, ensuring consistent supply chain reliability.
Q: How does the Nickel-TiO2 dual catalytic system ensure high selectivity?
A: The system utilizes a synergistic mechanism where TiO2 generates charge carriers under light to activate the substrates, while the Nickel cocatalyst facilitates the specific C-O bond formation. This dual approach minimizes side reactions such as homocoupling or dehalogenation, resulting in high GC yields (up to 90%) and simplifying downstream purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Prozac Intermediate Supplier
The technological potential of the TiO2-mediated photochemical synthesis of aryl alkyl ethers represents a significant opportunity for pharmaceutical manufacturers seeking to optimize their production pipelines. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our facility is equipped with state-of-the-art photoreactors and rigorous QC labs capable of handling complex photochemical reactions while adhering to stringent purity specifications required for global pharmaceutical markets. We understand the nuances of managing heterogeneous catalytic systems and can guarantee the consistent quality necessary for regulatory approval.
We invite you to collaborate with us to leverage this advanced synthesis route for your supply chain needs. Our technical team is prepared to conduct a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this green chemistry approach can improve your margins. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments, allowing you to make informed decisions about integrating this efficient and sustainable manufacturing method into your portfolio.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
