Advanced Manufacturing of Progesterone via Lewis Acid Elimination and Catalytic Hydrogenation
The pharmaceutical industry continuously seeks robust synthetic pathways for critical steroid hormones, and the novel process detailed in patent CN103087136A represents a significant advancement in the manufacturing of Progesterone. This technology shifts the paradigm from traditional extraction-dependent methods to a streamlined semi-synthetic approach utilizing 17-Alpha-Hydroxy Progesterone as the key starting material. By leveraging Lewis acid-mediated elimination followed by catalytic hydrogenation, this method addresses longstanding inefficiencies in yield and operational complexity. The structural integrity of the final product is paramount, as Progesterone serves as a pivotal intermediate for all steroid hormones and a vital therapeutic agent for reproductive health. 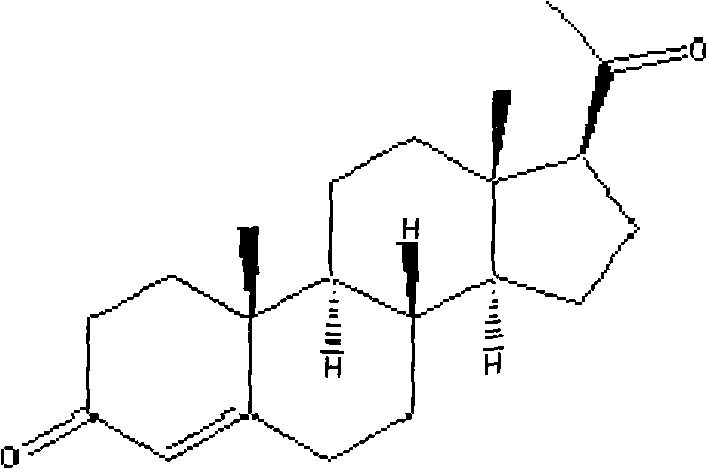 This report analyzes the technical merits and commercial viability of this innovation for global supply chains.
This report analyzes the technical merits and commercial viability of this innovation for global supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of Progesterone has relied heavily on natural precursors such as diosgenin, extracted from wild yams, or cholesterol derived from animal sources. As illustrated in the traditional diosgenin pathway, the synthesis involves multiple cumbersome steps including acetylation, oxidation, and complex ring modifications. 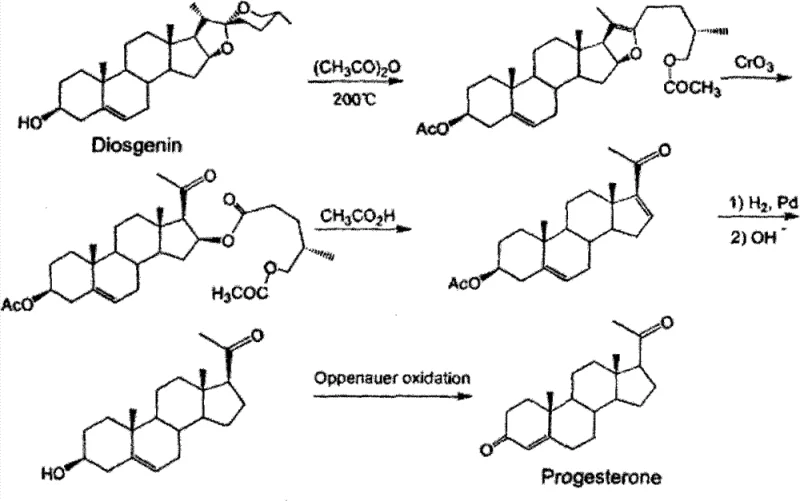 These legacy routes suffer from inherent volatility due to the agricultural dependency of raw materials, leading to inconsistent supply and fluctuating costs. Furthermore, prior art methods, such as those disclosed in CN201110007898.1, often necessitate rigorous anhydrous conditions and energy-intensive steam distillation for product isolation. This steam distillation step is particularly problematic; excessive steam can carry away the product, drastically reducing yield, while insufficient steam prevents proper crystallization, resulting in oily residues that are difficult to purify. Such operational sensitivities create significant bottlenecks for reliable pharmaceutical intermediates supplier networks aiming for consistent quality.
These legacy routes suffer from inherent volatility due to the agricultural dependency of raw materials, leading to inconsistent supply and fluctuating costs. Furthermore, prior art methods, such as those disclosed in CN201110007898.1, often necessitate rigorous anhydrous conditions and energy-intensive steam distillation for product isolation. This steam distillation step is particularly problematic; excessive steam can carry away the product, drastically reducing yield, while insufficient steam prevents proper crystallization, resulting in oily residues that are difficult to purify. Such operational sensitivities create significant bottlenecks for reliable pharmaceutical intermediates supplier networks aiming for consistent quality.
The Novel Approach
In stark contrast, the novel methodology presented in CN103087136A simplifies the synthetic architecture by initiating the sequence from 17-Alpha-Hydroxy Progesterone, a compound readily available via microbial fermentation of 4-AD (Androst-4-ene-3,17-dione). This strategic choice eliminates the lengthy plant growth cycles and extraction inefficiencies associated with diosgenin. The core innovation lies in a direct elimination reaction followed by a selective reduction, bypassing the need for complex protecting group strategies or harsh oxidative cleavage of side chains found in cholesterol-based routes. 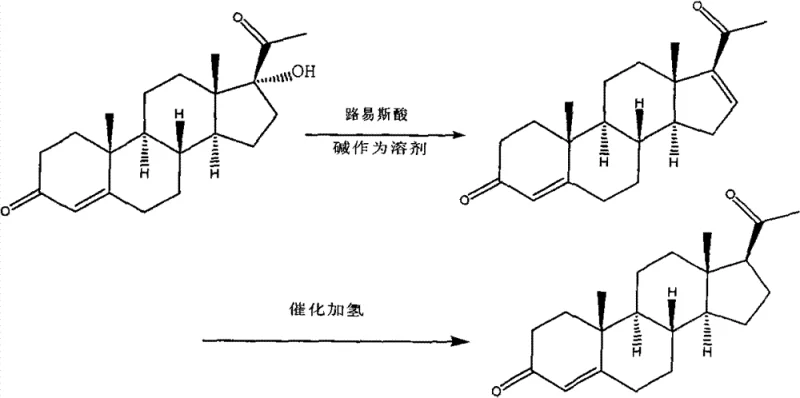 By avoiding the problematic steam distillation entirely and utilizing standard recrystallization techniques, this approach ensures a more stable and predictable manufacturing profile. The result is a process that is not only chemically elegant but also operationally superior, offering a viable solution for cost reduction in pharmaceutical intermediates manufacturing through simplified unit operations and reduced energy consumption.
By avoiding the problematic steam distillation entirely and utilizing standard recrystallization techniques, this approach ensures a more stable and predictable manufacturing profile. The result is a process that is not only chemically elegant but also operationally superior, offering a viable solution for cost reduction in pharmaceutical intermediates manufacturing through simplified unit operations and reduced energy consumption.
Mechanistic Insights into Lewis Acid-Mediated Elimination and Hydrogenation
The chemical elegance of this process is rooted in the precise control of the elimination step, where the 17-alpha-hydroxyl group is removed to establish the necessary double bond geometry or prepare the molecule for subsequent reduction. The patent specifies the use of Lewis acids such as phosphorus oxychloride (POCl3) in conjunction with basic solvents like pyridine or DMF. In this mechanism, the Lewis acid activates the hydroxyl group, facilitating its departure as a leaving group, while the base scavenges the generated protons. This reaction is typically conducted at elevated temperatures, around 80°C, to drive the equilibrium towards the elimination product. The choice of solvent is critical; anhydrous conditions are maintained to prevent hydrolysis of the Lewis acid, ensuring high conversion rates. Following this, the intermediate undergoes catalytic hydrogenation using heterogeneous catalysts such as Raney Nickel or Palladium on Carbon. This step selectively reduces specific unsaturations without affecting the sensitive ketone functionalities at positions 3 and 20, preserving the biological activity of the steroid nucleus.
Impurity control is a defining feature of this synthetic route, directly addressing the needs of R&D directors focused on purity profiles. The patent reports High Performance Liquid Chromatography (HPLC) purity levels exceeding 99%, achieved through a combination of selective catalysis and effective downstream processing. The hydrogenation step is carefully monitored, often at room temperature and moderate hydrogen pressure (1 kg/cm²), to prevent over-reduction or stereochemical scrambling. Post-reaction, the crude solid is subjected to decolorization using activated carbon in hot ethanol, which effectively removes trace organic impurities and catalyst residues. The final recrystallization from ethanol yields off-white crystals with a sharp melting point range of 128-130°C, indicative of high crystalline purity. This rigorous control over the reaction environment and purification sequence ensures that the final high-purity Progesterone meets stringent pharmacopeial standards, minimizing the risk of downstream formulation issues.
How to Synthesize Progesterone Efficiently
The synthesis of Progesterone via this novel route is designed for operational simplicity, making it accessible for both pilot-scale validation and full-scale commercial production. The process begins with the dissolution of 17-Alpha-Hydroxy Progesterone in an anhydrous alkaline solvent, followed by the controlled addition of the Lewis acid reagent. After the elimination phase is complete, the reaction mixture is quenched into acid, neutralized, and the intermediate is isolated. This intermediate is then subjected to hydrogenation in an autoclave, followed by a straightforward workup involving filtration and concentration. The detailed standardized synthetic steps, including specific molar ratios, temperature gradients, and safety protocols for handling Lewis acids and hydrogen gas, are outlined below for technical reference.
- Perform elimination reaction using 17-Alpha-Hydroxy Progesterone with Lewis acid (e.g., POCl3) in alkaline solvent (e.g., Pyridine) at elevated temperatures.
- Execute catalytic hydrogenation of the elimination product using Raney Nickel or Palladium on Carbon under controlled hydrogen pressure.
- Purify the crude product via decolorization with activated carbon and recrystallization from ethanol to obtain off-white Progesterone crystals.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this synthetic route offers tangible strategic benefits beyond mere chemical efficiency. The primary advantage lies in the decoupling of production from agricultural variables. By utilizing fermentation-derived starting materials, manufacturers can secure a consistent supply of raw materials regardless of seasonal harvest fluctuations or geopolitical instability affecting plant-based sources. This stability is crucial for maintaining continuous production schedules and meeting the rigid delivery timelines demanded by global pharmaceutical clients. Furthermore, the elimination of energy-intensive steps like prolonged steam distillation translates directly into lower utility costs and a reduced carbon footprint, aligning with modern sustainability goals in commercial scale-up of complex pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The streamlined nature of this process significantly lowers the cost of goods sold (COGS) by reducing the number of unit operations and eliminating the need for exotic reagents. The avoidance of steam distillation not only saves energy but also prevents product loss due to volatilization, thereby improving the overall mass balance. Additionally, the use of common industrial solvents like ethanol and pyridine, along with recoverable catalysts like Raney Nickel, minimizes raw material expenditure. These factors collectively contribute to substantial cost savings without compromising the quality of the final active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: Relying on 17-Alpha-Hydroxy Progesterone, which is produced via established fermentation technologies, ensures a robust and scalable supply chain. Unlike wild-crafted diosgenin, which is subject to ecological constraints and supply shocks, fermentation substrates can be produced in controlled bioreactors year-round. This reliability reduces lead time for high-purity Progesterone deliveries, allowing downstream drug manufacturers to optimize their inventory levels and reduce safety stock requirements. The predictability of this supply chain is a critical asset for long-term strategic planning in the volatile pharmaceutical market.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated in embodiments ranging from gram to multi-gram scales with consistent results. The reaction conditions, such as moderate temperatures and atmospheric or low-pressure hydrogenation, are easily adaptable to large-scale reactors without requiring specialized high-pressure equipment. Moreover, the simplified waste stream, devoid of heavy metal contaminants from complex oxidation steps, facilitates easier compliance with environmental regulations. This ease of waste treatment and the potential for solvent recovery further enhance the environmental profile of the manufacturing process.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel Progesterone synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on process capabilities and product specifications. Understanding these details is essential for technical teams evaluating the feasibility of adopting this methodology for their specific production needs.
Q: What are the primary advantages of using 17-Alpha-Hydroxy Progesterone as a starting material?
A: Using 17-Alpha-Hydroxy Progesterone bypasses the long growth cycles associated with natural plant extracts like diosgenin. It allows for a shorter synthetic route with higher overall yields and avoids complex steam distillation steps required in older methodologies.
Q: How does this process ensure high purity levels suitable for pharmaceutical applications?
A: The process utilizes specific Lewis acid conditions and controlled hydrogenation to minimize byproduct formation. Final purification through ethanol recrystallization and activated carbon decolorization consistently achieves HPLC purity greater than 99%.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the methodology employs standard industrial reagents like phosphorus oxychloride and Raney Nickel, and operates at manageable temperatures and pressures, making it highly suitable for commercial scale-up from pilot to multi-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Progesterone Supplier
The technological advancements described in CN103087136A underscore the potential for more efficient and sustainable hormone manufacturing. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate such innovative laboratory processes into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot to plant is seamless and compliant. We maintain stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of Progesterone meets the highest international standards for pharmaceutical application.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic impact of switching to this method. We encourage you to contact us for specific COA data and route feasibility assessments tailored to your volume requirements, ensuring a partnership built on transparency, quality, and mutual growth in the global healthcare market.
