Advanced Two-Step Synthesis of High-Purity Ioversol Hydrolysate for Commercial Scale-Up
Advanced Two-Step Synthesis of High-Purity Ioversol Hydrolysate for Commercial Scale-Up
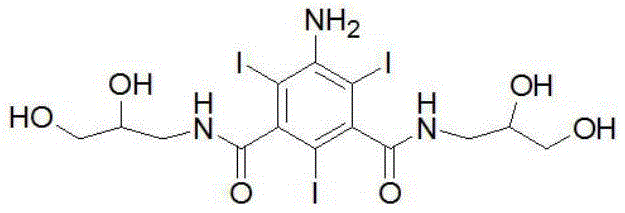
The pharmaceutical industry constantly seeks more efficient pathways for producing high-value contrast agents, and the recent disclosure in patent CN114409566A represents a significant leap forward in the synthesis of ioversol hydrolysate. This critical intermediate, chemically known as 5-(N-2-hydroxyethyl)hydroxyacetamido-N,N'-bis(2,3-dihydroxypropyl)-2,4,6-triiodo-1,3-benzenedicarboxamide, serves as the foundational building block for ioversol, a widely used non-ionic X-ray contrast medium. The patented methodology addresses long-standing challenges regarding impurity profiles and process complexity, offering a route that achieves a remarkable purity of greater than 99.5% and a molar yield exceeding 90%. By leveraging a streamlined acetoxyacetylation strategy followed by controlled hydrolysis, this technology provides a robust framework for industrial manufacturing. For R&D directors and procurement specialists, understanding the nuances of this synthesis is vital for securing a reliable pharmaceutical intermediate supplier capable of meeting stringent regulatory standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of ioversol intermediates has been plagued by inefficient multi-step sequences that compromise both yield and economic viability. Prior art, such as that described in patents US4396598 and WO2008/104853, typically necessitates three to four distinct reaction steps, often involving the preparation of unstable acyl chlorides from scratch. These legacy processes frequently result in significant accumulation of impurities, particularly residual iodide starting materials, which are notoriously difficult to remove without resorting to expensive and low-throughput preparative chromatography. Furthermore, earlier attempts using ethoxyacetyl chloride under N,N-dimethylpyridine catalysis, as noted in WO93/16981, suffered from incomplete reactions, leaving substantial amounts of unreacted iodide in the final hydrolysate. This lack of conversion efficiency not only drives up raw material costs but also creates a heavy burden on downstream purification units, making large-scale industrial application economically unfeasible for many manufacturers seeking cost reduction in API manufacturing.
The Novel Approach
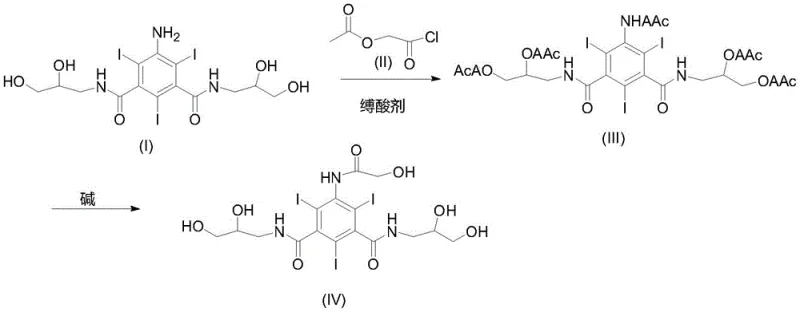
In stark contrast to these cumbersome legacy routes, the method disclosed in CN114409566A introduces a highly efficient two-step one-pot reaction sequence that dramatically simplifies the operational workflow. The core innovation lies in the direct ethoxylation and acetylation of the iodide starting material using commercially available ethoxyacetyl chloride in the presence of a specific acid-binding agent. This eliminates the need for a separate acyl chloride preparation stage, thereby reducing the overall step count and minimizing potential points of failure. The reaction proceeds smoothly to form the acetylated intermediate (III), which is subsequently hydrolyzed in situ without isolation. This telescoped approach not only accelerates the production timeline but also ensures that the final product precipitates directly from the aqueous solution upon pH adjustment, facilitating easy filtration and drying. The result is a white solid with exceptional purity, validating the process as a superior choice for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Acetoxyacetylation and Hydrolysis
The success of this synthesis hinges on the precise control of the acylation mechanism, where the nucleophilic amine groups of the iodide substrate attack the electrophilic carbonyl carbon of the ethoxyacetyl chloride. To drive this equilibrium forward and neutralize the generated hydrogen chloride, the protocol mandates the use of an acid-binding agent, preferably triethylamine, in a molar ratio of 3.0-4.2:1 relative to the iodide. This stoichiometric excess is critical; insufficient base leads to incomplete conversion and residual starting material, while excessive base can promote side reactions. The reaction is conducted in polar aprotic solvents such as N,N-dimethylacetamide (DMAC) or N,N-dimethylformamide (DMF) at temperatures ranging from 0°C to 40°C. These conditions optimize the kinetic energy of the molecules while preventing thermal degradation of the sensitive triiodobenzene core, ensuring that the acetylated intermediate (III) is formed with high fidelity before the subsequent hydrolysis step commences.
Following the acylation, the mechanism shifts to a base-catalyzed hydrolysis designed to cleave the acetyl protecting groups and reveal the free hydroxyl functionalities essential for the biological activity of the final contrast agent. An alkaline solution, typically sodium hydroxide with a concentration of 10%-35%, is added to raise the pH to between 12 and 14. This strong basic environment facilitates the rapid saponification of the ester linkages within a short timeframe of 0.5 to 2 hours. Crucially, the process concludes with a careful acidification using concentrated hydrochloric acid to adjust the pH to a range of 4-7. This specific pH window induces the precipitation of the target ioversol hydrolysate while keeping soluble impurities in the mother liquor. This "crystallization in aqueous solution" technique is a masterstroke of process chemistry, leveraging solubility differences to achieve high purity without the need for organic solvent recrystallization or column chromatography.
How to Synthesize Ioversol Hydrolysate Efficiently
Implementing this synthesis requires strict adherence to the specified stoichiometric ratios and temperature profiles to ensure reproducibility and safety on a kilogram or tonne scale. The process begins with the uniform mixing of the iodide substrate and ethoxyacetyl chloride in a suitable solvent, followed by the controlled addition of the acid scavenger to manage exotherms. Once the acetylation is complete, the reaction mixture is treated with alkali for hydrolysis, and the product is recovered via simple solid-liquid separation. The detailed standardized synthetic steps, including specific reagent quantities and workup procedures derived from the patent examples, are outlined below to guide process engineers in replicating this high-yield route.
- Mix iodide and ethoxyacetyl chloride in a polar aprotic solvent with an acid-binding agent at 0-40°C to form the acetylated intermediate.
- Add alkaline solution to adjust pH to 12-14 for hydrolysis, followed by acidification to pH 4-7 to precipitate the product.
- Separate solids via filtration and dry at 60-80°C to obtain white ioversol hydrolysate powder with >99.5% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthesis route offers compelling economic and logistical benefits that extend far beyond simple yield improvements. By utilizing commercially available raw materials like ethoxyacetyl chloride and avoiding the capital-intensive setup for in-situ acyl chloride generation, the barrier to entry for production is significantly lowered. The elimination of complex purification steps such as preparative chromatography translates directly into reduced operational expenditures (OPEX) and lower waste disposal costs. Furthermore, the ability to crystallize the product directly from an aqueous solution minimizes the consumption of volatile organic compounds (VOCs), aligning the manufacturing process with increasingly stringent environmental regulations and sustainability goals. This green chemistry approach not only mitigates regulatory risk but also enhances the overall safety profile of the production facility.
- Cost Reduction in Manufacturing: The streamlined two-step process drastically reduces the consumption of solvents and reagents compared to traditional three or four-step methods. By removing the need for intermediate isolation and purification, manufacturers can achieve substantial cost savings in labor, energy, and material usage. The high conversion rate ensures that expensive iodinated starting materials are utilized efficiently, minimizing the financial loss associated with unreacted feedstock. Additionally, the simplified workflow reduces the requirement for specialized equipment, allowing for production in standard glass-lined or stainless steel reactors, which further optimizes capital expenditure.
- Enhanced Supply Chain Reliability: Since the key reagents, including the iodide substrate and ethoxyacetyl chloride, are commercially available commodities, the supply chain is less vulnerable to bottlenecks associated with custom-synthesized precursors. The robustness of the reaction conditions, which tolerate a range of temperatures and solvent choices, provides flexibility in sourcing and inventory management. This reliability ensures consistent production schedules and shorter lead times for high-purity pharmaceutical intermediates, enabling downstream API manufacturers to maintain steady inventory levels and meet market demand without interruption.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated effectively from gram scale in the lab to multi-kilogram batches in the patent examples. The use of aqueous crystallization significantly reduces the volume of organic waste streams, simplifying wastewater treatment and lowering the environmental footprint of the facility. This ease of scale-up, combined with the high purity of the final product (>99.5%), means that the process can be rapidly transferred to commercial production lines with minimal validation hurdles, ensuring a continuous and compliant supply of this critical medical intermediate.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this preparation method. These answers are derived directly from the experimental data and technical disclosures found in the patent literature, providing clarity on reaction parameters, impurity control, and scalability. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for their own manufacturing portfolios.
Q: What are the critical reaction conditions for maximizing yield in ioversol hydrolysate synthesis?
A: According to patent CN114409566A, maintaining the ethoxyacetylation temperature between 0-40°C and using a molar ratio of acid-binding agent to iodide of 3.0-4.2:1 is crucial. Additionally, controlling the hydrolysis pH between 12-14 ensures complete conversion before final acidification.
Q: How does this new method improve upon traditional ioversol intermediate production?
A: Traditional methods often require 3-4 steps and complex chromatographic purification. This novel approach simplifies the process to just two steps in a one-pot reaction, eliminating the need for intermediate isolation and achieving yields over 90% with purity exceeding 99.5%.
Q: Is the raw material ethoxyacetyl chloride commercially viable for large-scale production?
A: Yes, the process utilizes commercially available ethoxyacetyl chloride and iodide. This avoids the need for in-situ preparation of unstable acyl chlorides, significantly reducing raw material costs and supply chain complexity compared to older patents like US4396598.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ioversol Hydrolysate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the production of life-saving diagnostic agents. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We are committed to delivering ioversol hydrolysate that meets stringent purity specifications, backed by our rigorous QC labs and state-of-the-art analytical capabilities. Our facility is equipped to handle the specific solvent systems and temperature controls required by this advanced synthesis, guaranteeing a product that consistently exceeds the >99.5% purity benchmark demanded by global regulatory bodies.
We invite pharmaceutical partners to collaborate with us to leverage this cutting-edge technology for their supply chains. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing processes can enhance your operational efficiency and reduce your overall cost of goods sold.
