Scalable Manufacturing of 5-Bromo-2,4-dichloropyridine via Novel Diazotization Route
Scalable Manufacturing of 5-Bromo-2,4-dichloropyridine via Novel Diazotization Route
The pharmaceutical industry constantly seeks reliable pathways for complex heterocyclic intermediates that balance high purity with economic viability. Patent CN103420903A introduces a transformative method for synthesizing 5-Bromo-2,4-dichloropyridine, a critical building block in medicinal chemistry. This technology addresses the longstanding challenges associated with poly-halogenated pyridine production by replacing hazardous cryogenic processes with a manageable, high-yield sequence starting from inexpensive 2-amino-4-chloropyridine. By leveraging regioselective bromination followed by a classic Sandmeyer-type transformation, this approach eliminates the need for extreme low-temperature infrastructure and dangerous organometallic reagents. For R&D directors and procurement strategists, this represents a pivotal shift towards safer, more cost-effective manufacturing of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of poly-halogenated pyridines has relied heavily on direct lithiation strategies that impose severe operational burdens on manufacturing facilities. Traditional routes often necessitate the use of strong bases like Lithium Diisopropylamide (LDA) at cryogenic temperatures reaching -100°C, as illustrated in prior art methodologies. These conditions not only require specialized equipment capable of maintaining extreme cold but also introduce significant safety risks associated with handling pyrophoric reagents on a large scale. Furthermore, the selectivity of such lithiation reactions can be unpredictable, often leading to complex impurity profiles that are difficult and costly to purify. The reliance on expensive starting materials and the inherent difficulty in controlling exothermic events at such low temperatures make these conventional methods economically unviable for commercial production.
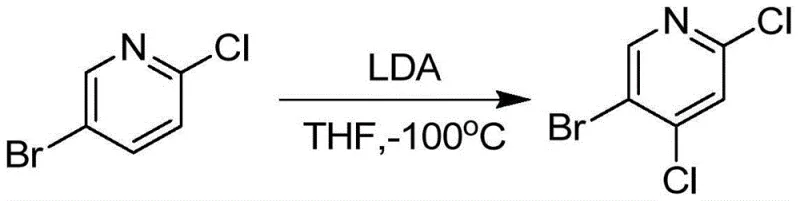
In addition to lithiation, other historical approaches have utilized harsh halogenating agents or fluorinated solvents under similarly restrictive low-temperature conditions, such as -75°C. These methods suffer from poor atom economy and generate substantial waste streams, complicating environmental compliance and disposal protocols. The combination of high raw material costs, energy-intensive cooling requirements, and low overall yields creates a bottleneck for supply chains attempting to secure consistent volumes of 5-Bromo-2,4-dichloropyridine. Consequently, manufacturers have struggled to scale these processes beyond laboratory quantities without incurring prohibitive costs or compromising on product quality.
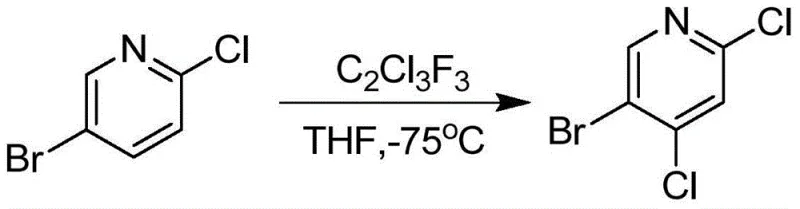
The Novel Approach
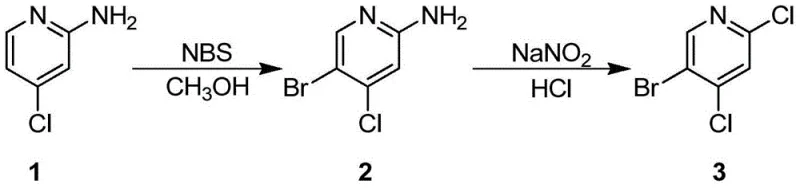
The patented methodology offers a robust alternative by utilizing 2-amino-4-chloropyridine as a readily available and cost-effective starting scaffold. This route bypasses the need for cryogenic lithiation entirely, instead employing N-bromo-succinimide (NBS) for a highly regioselective bromination at the 5-position of the pyridine ring. The reaction proceeds smoothly at 0°C in dichloromethane, achieving yields exceeding 80% with minimal byproduct formation. Following isolation, the amino group serves as a versatile handle for conversion to the chloro substituent via diazotization and subsequent treatment with cuprous chloride. This two-step sequence operates at moderate temperatures (-30°C to room temperature), drastically reducing energy consumption and equipment complexity compared to legacy methods.
Mechanistic Insights into Regioselective Bromination and Diazotization
The success of this synthesis hinges on the electronic activation of the pyridine ring by the amino group, which directs the electrophilic bromination specifically to the 5-position. In the presence of NBS, the electron-rich nature of the aminopyridine facilitates an electrophilic aromatic substitution that avoids the formation of undesired isomers, a common pitfall in non-directed halogenation. The use of dichloromethane as a solvent ensures optimal solubility and heat dissipation during the exothermic bromination event. Following the bromination, the transformation of the amine to a chloride via the diazonium salt is a classic example of nucleophilic aromatic substitution mediated by a copper catalyst. The generation of the diazonium ion at -30°C ensures stability, preventing premature decomposition, while the subsequent addition of cuprous chloride facilitates the replacement of the diazo group with chlorine through a radical or ionic mechanism depending on the specific conditions.
From an impurity control perspective, this route is superior because each step generates distinct, separable byproducts. The succinimide byproduct from the NBS reaction is easily removed during the aqueous workup, while the copper salts from the chlorination step are eliminated during the final extraction and purification. This clarity in the impurity profile simplifies the downstream processing, allowing for the attainment of high-purity specifications required for pharmaceutical applications without the need for extensive chromatographic purification. The mechanistic predictability of these well-understood reactions provides R&D teams with confidence in the reproducibility of the process across different batch sizes.
How to Synthesize 5-Bromo-2,4-dichloropyridine Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production, emphasizing precise temperature control and stoichiometric balance. The process begins with the dissolution of the starting material in an inert solvent, followed by the controlled addition of the brominating agent to manage heat release. After isolating the bromo-intermediate, the diazotization step requires careful monitoring of pH and temperature to ensure complete conversion before the final chlorination. For detailed operational parameters and specific molar ratios, please refer to the standardized synthesis guide below.
- Dissolve 2-amino-4-chloropyridine in dichloromethane, cool to 0°C, and react with N-bromo-succinimide (NBS) to obtain the 5-bromo intermediate.
- Perform diazotization of the intermediate using sodium nitrite and concentrated hydrochloric acid at -30°C.
- Add cuprous chloride to the diazonium salt solution, warm to room temperature, and isolate the final 5-Bromo-2,4-dichloropyridine product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthetic route translates directly into enhanced supply security and reduced operational risk. By shifting away from exotic, low-temperature chemistries to standard unit operations, manufacturers can utilize existing infrastructure without capital-intensive upgrades. The reliance on commodity chemicals like 2-amino-4-chloropyridine and NBS ensures a stable supply of raw materials, insulating the production schedule from the volatility often seen with specialized organometallic reagents. This stability is crucial for maintaining continuous production lines and meeting the rigorous delivery timelines demanded by global pharmaceutical clients.
- Cost Reduction in Manufacturing: The elimination of cryogenic cooling systems operating at -100°C results in substantial energy savings and reduces the wear and tear on specialized reactor vessels. Furthermore, the avoidance of expensive organolithium bases and the use of high-yielding steps significantly lowers the cost of goods sold (COGS). The simplified purification process also reduces solvent consumption and waste disposal costs, contributing to a leaner and more profitable manufacturing model.
- Enhanced Supply Chain Reliability: Sourcing 2-amino-4-chloropyridine is far more reliable than procuring custom-synthesized halogenated precursors, as it is a widely produced bulk chemical. This abundance ensures that production schedules are not held hostage by single-source suppliers or long lead times for niche reagents. Additionally, the robustness of the reaction conditions minimizes the risk of batch failures due to sensitive parameters, thereby guaranteeing consistent output and on-time delivery performance.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated effectively from gram to kilogram scales without loss of efficiency. The absence of highly toxic phosphorus oxychloride or pyrophoric butyllithium simplifies environmental health and safety (EHS) compliance, reducing the regulatory burden on the manufacturing site. Waste streams are easier to treat, and the overall greener profile of the synthesis aligns with modern sustainability goals, making it an attractive option for environmentally conscious partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 5-Bromo-2,4-dichloropyridine using this patented methodology. These answers are derived directly from the experimental data and technical specifications provided in the source documentation to ensure accuracy and relevance for potential partners.
Q: What are the critical temperature controls for the diazotization step?
A: The diazotization reaction must be strictly maintained at -30°C during the addition of sodium nitrite to ensure stability of the diazonium salt and prevent side reactions, before warming to room temperature for the Sandmeyer chlorination.
Q: Why is this route preferred over cryogenic lithiation methods?
A: Unlike traditional methods requiring -100°C and hazardous organolithium reagents like LDA, this patented route operates at mild temperatures (0°C and -30°C) using stable reagents like NBS, significantly improving operational safety and scalability.
Q: What is the expected yield profile for this synthesis?
A: The bromination step typically achieves yields greater than 80% (up to 87% in examples), while the subsequent diazotization-chlorination sequence provides yields around 68%, resulting in a robust total yield exceeding 50%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Bromo-2,4-dichloropyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the drug development lifecycle. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with precision. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. By adopting advanced synthetic routes like the one described in CN103420903A, we optimize our processes to provide you with the best balance of quality and value.
We invite you to collaborate with us to explore how this efficient synthesis can benefit your specific project needs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to support your supply chain strategy and accelerate your time to market.
