Revolutionizing Azoxystrobin Intermediate Production via Selective N-Hydroxy Catalysis
Revolutionizing Azoxystrobin Intermediate Production via Selective N-Hydroxy Catalysis
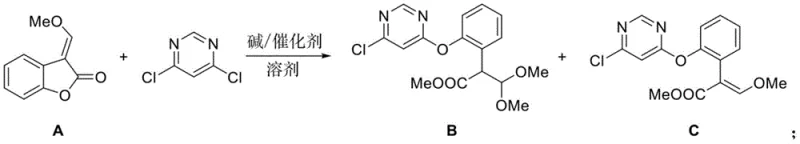
The global demand for high-efficiency fungicides continues to drive innovation in agrochemical intermediate manufacturing, particularly for strobilurin-class compounds like azoxystrobin. A pivotal advancement in this sector is detailed in patent CN112174897B, which discloses a highly selective preparation method for the critical azoxystrobin intermediate, (E)-2-[2-(6-chloropyrimidin-4-yloxy)phenyl]-3-methoxyacrylate. This technology addresses long-standing bottlenecks in yield and purity that have plagued conventional synthetic routes. By employing specific N-hydroxy catalysts in conjunction with mild carbonate bases, the process achieves superior chemoselectivity, effectively suppressing the formation of difficult-to-remove byproducts. For R&D directors and procurement specialists seeking a reliable agrochemical intermediate supplier, understanding the mechanistic advantages of this patented route is essential for securing a competitive supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of this key intermediate has relied on methods exemplified by earlier patents, such as those from Chengda Corporation, which utilize sodium methoxide in methanol under rigorous low-temperature conditions. These traditional protocols suffer from inherent inefficiencies, primarily due to the extreme nucleophilicity of the methoxide ion, which lacks the necessary selectivity to distinguish between the desired ring-opening pathway and competing Michael addition reactions. Consequently, the reaction generates a significant quantity of byproduct B, a structural isomer that is chemically similar to the target compound C and notoriously difficult to separate via standard crystallization or distillation techniques. Furthermore, the requirement for continuous low-temperature control over extended reaction periods, often exceeding 22 hours, results in substantial energy consumption and limits the throughput capacity of manufacturing facilities. The cumulative effect of these factors is a process with variable yields, typically hovering between 60% and 70%, and a final product profile that requires extensive downstream purification to meet the stringent quality standards demanded by modern agrochemical formulators.
The Novel Approach
In stark contrast, the methodology outlined in patent CN112174897B introduces a paradigm shift by replacing harsh alkoxide bases with mild inorganic carbonates and introducing specialized N-hydroxy catalysts. This novel approach fundamentally alters the reaction landscape, enabling the transformation to proceed under much milder thermal conditions, specifically between 20°C and 50°C, thereby drastically reducing energy overheads. The core innovation lies in the dual-stage addition of the base and the specific catalytic activation of the lactone substrate, which directs the reaction exclusively towards the desired ring-opening etherification. This strategic modification not only accelerates the reaction kinetics, reducing total processing time to approximately 6 to 8 hours, but also inherently suppresses the formation of the problematic Michael addition byproduct. For supply chain managers, this translates to a more robust and predictable manufacturing process that minimizes waste generation and maximizes the output of high-purity material per batch, directly addressing the need for cost reduction in pesticide manufacturing.
Mechanistic Insights into N-Hydroxy Catalyzed Ring-Opening
The efficacy of this synthetic route is rooted in the unique electronic properties of the selected N-hydroxy catalysts, such as N-hydroxysuccinimide (HOSu) and 1-hydroxybenzotriazole (OHBT). These molecules function by activating the carbonyl group of the methoxymethylene benzofuranone substrate through hydrogen bonding or transient acyl intermediate formation, rendering the lactone ring significantly more susceptible to nucleophilic attack by the pyrimidine moiety. Unlike traditional amine catalysts like DABCO or trimethylamine, which often struggle with separation and exhibit lower turnover frequencies, these N-hydroxy compounds provide a precise steric and electronic environment that favors the formation of the ether linkage over the conjugate addition pathway. This selective activation ensures that the nucleophilic oxygen of the pyrimidine attacks the correct position on the benzofuranone ring, effectively bypassing the thermodynamic trap that leads to byproduct B. The result is a reaction profile characterized by high conversion rates and exceptional selectivity, allowing for the direct isolation of the target intermediate with minimal impurity burden.
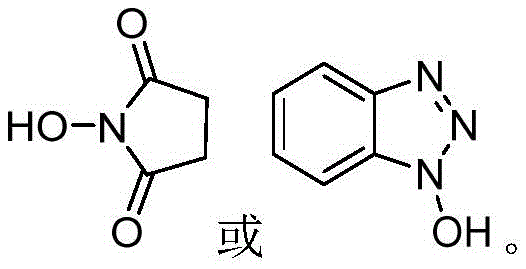
Furthermore, the choice of base plays a critical role in the overall mechanism and subsequent workup efficiency. By utilizing sodium carbonate or potassium carbonate instead of sodium methoxide, the system avoids the generation of highly reactive methoxide ions that indiscriminately attack electrophilic centers. The carbonate anion acts as a sufficient proton scavenger to drive the equilibrium forward without inducing the aggressive side reactions associated with strong alkoxides. This subtle yet profound change in reagent selection simplifies the post-reaction processing significantly. Since the N-hydroxy catalysts and the inorganic carbonate salts possess high water solubility, the crude reaction mixture can be subjected to a straightforward aqueous extraction protocol. The organic phase retains the lipophilic product, while the catalyst and base residues are washed away into the aqueous layer. This eliminates the need for complex acid-base extractions or column chromatography, streamlining the path from reactor to final crystal and ensuring a cleaner impurity profile for the high-purity agrochemical intermediate.
How to Synthesize (E)-2-[2-(6-chloropyrimidin-4-yloxy)phenyl]-3-methoxyacrylate Efficiently
The implementation of this catalytic protocol requires precise control over stoichiometry and temperature gradients to maximize the benefits of the N-hydroxy activation. The process begins with the dissolution of the starting materials in a polar solvent such as methanol or dimethylformamide, followed by the controlled addition of the base and catalyst under cooling to manage the initial exotherm. Detailed operational parameters, including specific molar ratios and heating ramps, are critical for reproducing the high yields reported in the patent examples. For process chemists looking to adopt this technology, adhering to the standardized synthesis steps is vital for ensuring consistent quality and safety during scale-up operations.
- Charge methoxymethylene benzofuranone and 4,6-dichloropyrimidine into a reactor with solvent (methanol or DMF) under inert atmosphere.
- Add carbonate base (sodium or potassium carbonate) and N-hydroxy catalyst (HOSu or OHBT) at low temperature, then warm to 20-30°C for ring-opening.
- Add remaining base, heat to 40-50°C for etherification, then isolate product via extraction and recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this catalytic technology offers compelling advantages that extend far beyond simple yield improvements, directly impacting the bottom line for agrochemical producers. The shift from expensive and hazardous sodium methoxide to commodity carbonate bases represents a tangible reduction in raw material costs, while the elimination of complex purification steps reduces solvent consumption and waste disposal fees. For procurement managers, this means a more stable cost structure that is less susceptible to fluctuations in specialty reagent pricing. Additionally, the robustness of the reaction conditions allows for greater flexibility in manufacturing scheduling, as the process is less sensitive to minor temperature deviations compared to the cryogenic requirements of legacy methods. This operational resilience is a key factor in ensuring commercial scale-up of complex agrochemical intermediates without compromising on delivery timelines or product specifications.
- Cost Reduction in Manufacturing: The replacement of sodium methoxide with inexpensive sodium or potassium carbonate significantly lowers the direct material cost per kilogram of product. Moreover, the high selectivity of the N-hydroxy catalysts minimizes the loss of valuable starting materials to byproduct formation, effectively increasing the mass balance efficiency of the entire process. The simplified workup procedure, which relies on aqueous washing rather than intensive chromatographic separation, further drives down operational expenditures by reducing labor hours and solvent usage. These cumulative efficiencies create a leaner manufacturing model that delivers substantial economic value without sacrificing chemical quality.
- Enhanced Supply Chain Reliability: The use of readily available, non-hazardous reagents like carbonates and common N-hydroxy compounds mitigates supply chain risks associated with regulated or dangerous chemicals. The shortened reaction cycle time, dropping from over 20 hours to under 10 hours, increases the asset utilization rate of production reactors, allowing manufacturers to respond more agilely to market demand spikes. This increased throughput capacity ensures a steady flow of material, reducing lead time for high-purity agrochemical intermediates and providing a buffer against potential logistical disruptions in the global supply network.
- Scalability and Environmental Compliance: The mild reaction temperatures and absence of highly corrosive reagents make this process inherently safer and easier to scale from pilot plant to multi-ton production volumes. The aqueous nature of the waste stream, containing primarily dissolved salts and water-soluble catalysts, simplifies effluent treatment and aligns with increasingly stringent environmental regulations regarding organic solvent discharge. This green chemistry profile not only reduces the environmental footprint of the manufacturing site but also future-proofs the supply chain against evolving regulatory pressures, ensuring long-term operational continuity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthetic route. These insights are derived directly from the experimental data and beneficial effects described in the patent literature, providing a clear picture of the technology's practical application. Understanding these nuances is crucial for stakeholders evaluating the feasibility of integrating this method into their existing production portfolios.
Q: How does the new catalytic method improve yield compared to traditional sodium methoxide routes?
A: The new method utilizes N-hydroxy catalysts like HOSu to selectively activate the lactone ring, suppressing the Michael addition side reaction that forms impurity B. This shifts the yield from the traditional 60-70% range to over 80-89%.
Q: What are the advantages of using carbonate bases over sodium methoxide in this synthesis?
A: Carbonate bases such as sodium carbonate are significantly less nucleophilic than sodium methoxide, reducing unwanted side reactions. Furthermore, they are cheaper, safer to handle, and allow for easier aqueous workup procedures.
Q: Is the catalyst easy to remove from the final agrochemical intermediate?
A: Yes, catalysts like HOSu and OHBT possess excellent water solubility. Post-reaction, they can be effectively removed through simple aqueous washing steps, eliminating the need for complex chromatographic purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Azoxystrobin Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthetic routes in the competitive agrochemical landscape. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN112174897B can be seamlessly translated into industrial reality. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of azoxystrobin intermediate meets the exacting standards required for final fungicide formulation. Our commitment to process excellence allows us to offer partners a secure and high-quality supply of critical intermediates.
We invite global agrochemical manufacturers to collaborate with us on optimizing their supply chains through advanced catalytic technologies. By leveraging our expertise, you can achieve significant operational efficiencies and secure a competitive edge in the market. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to support your strategic sourcing decisions.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
