Advanced Cobalt-Catalyzed Synthesis of Difluoroalkyl Heterocycles for Commercial Scale-Up
Introduction to Next-Generation Fluorination Technology
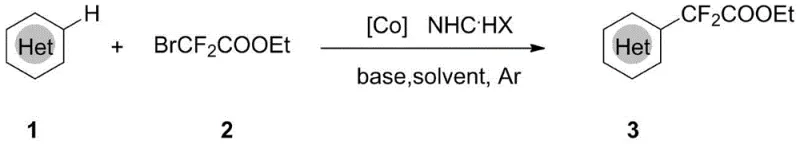
The strategic incorporation of fluorine atoms into organic scaffolds has become a cornerstone of modern medicinal chemistry and agrochemical design, fundamentally altering the physicochemical properties of bioactive molecules. Patent CN108997193B introduces a groundbreaking synthetic methodology that addresses the longstanding challenges associated with introducing difluoromethylene groups into heterocyclic systems. This innovation leverages a cobalt-catalyzed C-H activation strategy, utilizing ethyl difluorobromoacetate as a versatile building block to construct complex organofluorine architectures. By shifting away from traditional noble metal catalysts and hazardous fluorinating agents, this technology offers a sustainable and economically viable pathway for the production of high-value intermediates. For R&D directors and procurement specialists alike, this patent represents a significant opportunity to optimize supply chains for fluorinated pharmaceutical and agrochemical ingredients. The method's reliance on earth-abundant cobalt and simple operational protocols underscores its potential for seamless integration into large-scale manufacturing environments.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of difluoroalkylated heterocycles has been plagued by significant technical and safety hurdles that impede efficient commercial production. Traditional approaches often rely on dangerous deoxofluorination reagents such as DAST (diethylaminosulfur trifluoride), SF4, or XeF2, which require extremely harsh reaction conditions and pose severe safety risks during handling and storage. Alternative strategies involving transition metal-catalyzed coupling reactions typically necessitate the use of pre-functionalized aromatic substrates, such as iodoaromatics or arylboronic acids, which dramatically increases the step count and overall cost of goods. Furthermore, existing methods utilizing palladium catalysts, while effective, suffer from the economic burden of using precious metals and often require high temperatures up to 110°C with specialized ligands like Xantphos. These legacy processes frequently exhibit limited atom economy and generate substantial waste streams, creating bottlenecks for reliable agrochemical intermediate suppliers aiming for green chemistry compliance. The cumulative effect of these limitations is a supply chain vulnerable to raw material volatility and regulatory scrutiny regarding hazardous waste disposal.
The Novel Approach
In stark contrast to these cumbersome legacy protocols, the method disclosed in CN108997193B utilizes a direct C-H functionalization strategy that bypasses the need for substrate pre-functionalization entirely. By employing a catalytic system composed of inexpensive cobalt salts and nitrogen-heterocyclic carbene (NHC) precursors, the reaction achieves high efficiency under relatively mild thermal conditions. The use of ethyl difluorobromoacetate as the difluoroalkylating agent provides a commercially accessible and stable alternative to exotic fluorine sources, significantly simplifying the procurement process. This novel approach not only enhances the regioselectivity of the substitution at the C-H position adjacent to the heteroatom but also drastically reduces the environmental footprint of the synthesis. The operational simplicity, characterized by a straightforward workup involving filtration and concentration, makes this technology particularly attractive for cost reduction in pharmaceutical intermediates manufacturing. Ultimately, this shift towards base-metal catalysis represents a paradigm change in how complex fluorinated motifs are accessed industrially.
Mechanistic Insights into Cobalt-Catalyzed C-H Activation
The core of this technological breakthrough lies in the synergistic interaction between the cobalt center and the N-heterocyclic carbene ligand, which facilitates the activation of inert C-H bonds. The NHC ligand acts as a strong sigma-donor, stabilizing the low-valent cobalt species and enhancing its nucleophilicity towards the electrophilic difluoroalkyl radical generated in situ. Mechanistic studies suggest that the reaction proceeds through a radical pathway where the cobalt catalyst mediates the homolytic cleavage of the carbon-bromine bond in ethyl difluorobromoacetate. This generates a reactive difluoroalkyl radical species that selectively attacks the electron-rich positions of the heterocyclic substrate, such as the C2 or C3 positions of indoles and pyrroles. The robustness of the Co-NHC complex ensures that the catalytic cycle remains active throughout the reaction duration, minimizing catalyst deactivation and metal leaching issues common in other base-metal systems. For technical teams, understanding this mechanism is crucial for troubleshooting potential side reactions and optimizing ligand loading for specific substrate classes.
From an impurity control perspective, the high regioselectivity of this cobalt-catalyzed system is a major advantage for producing high-purity OLED material or API precursors. Unlike non-catalytic radical fluorinations which often yield complex mixtures of poly-fluorinated byproducts, this method directs the difluoroalkyl group specifically to the desired position adjacent to the heteroatom. The absence of pre-functionalized halides eliminates the risk of homocoupling impurities that are typical in cross-coupling reactions. Furthermore, the mild basic conditions employed, utilizing bases like potassium carbonate or triethylamine, prevent the degradation of sensitive functional groups that might be present on the heterocyclic core. This precision in bond formation translates directly to simplified downstream purification processes, reducing the need for extensive recrystallization or preparative HPLC steps. Consequently, the final product profile is cleaner, meeting the stringent purity specifications required by global regulatory bodies for clinical trial materials.
How to Synthesize Ethyl Difluoro(2-indolyl)acetate Efficiently
The practical implementation of this synthesis is designed to be accessible for both laboratory-scale optimization and pilot-plant operations without requiring specialized high-pressure equipment. The protocol begins with the preparation of the reaction mixture under an inert argon atmosphere to prevent oxidation of the sensitive cobalt catalyst and radical intermediates. Operators simply combine the heterocyclic substrate, ethyl difluorobromoacetate, cobalt bromide, the NHC precursor, and a base in a solvent like 1,4-dioxane or toluene. The detailed standardized synthesis steps see the guide below, which outlines the precise molar ratios and thermal profiles necessary to achieve optimal yields exceeding 80% for model substrates. Adhering to these parameters ensures consistent batch-to-batch reproducibility, a critical factor for contract development and manufacturing organizations (CDMOs).
- Under an argon atmosphere, dissolve the heterocyclic substrate and ethyl difluorobromoacetate in a solvent such as 1,4-dioxane, then add the cobalt catalyst, N-heterocyclic carbene precursor, and a base like triethylamine.
- Heat the reaction mixture to a temperature between 100°C and 130°C and stir continuously for approximately 6 hours to ensure complete conversion.
- Filter the cooled reaction mixture through diatomite, concentrate the filtrate under reduced pressure, and purify the crude residue via column chromatography using petroleum ether and ethyl acetate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this cobalt-catalyzed technology offers tangible benefits that extend far beyond simple reaction yield improvements. The elimination of expensive palladium catalysts and the avoidance of pre-functionalized starting materials result in a drastically simplified bill of materials. This structural simplification of the synthesis route inherently lowers the cost of goods sold (COGS) by removing multiple synthetic steps and the associated labor and utility costs. Moreover, the use of commercially available ethyl difluorobromoacetate ensures a stable and reliable supply of key raw materials, mitigating the risk of shortages often associated with custom-synthesized fluorinated reagents. The environmental friendliness of the process also aligns with increasingly strict corporate sustainability goals, potentially reducing waste disposal fees and regulatory compliance burdens.
- Cost Reduction in Manufacturing: The substitution of precious palladium catalysts with earth-abundant cobalt salts represents a direct and significant reduction in raw material costs for large-scale production. By eliminating the need for pre-functionalized substrates like aryl iodides or boronic acids, the process removes entire upstream synthesis stages, thereby compounding the savings in time and resources. The simplified workup procedure, which involves basic filtration and concentration rather than complex aqueous extractions or metal scavenging, further reduces operational expenditures. These cumulative efficiencies allow for a much more competitive pricing structure for the final difluoroalkylated intermediates in the global market.
- Enhanced Supply Chain Reliability: Sourcing strategies are greatly improved by the reliance on commodity chemicals such as cobalt bromide and triethylamine, which are widely available from multiple global vendors. The robustness of the reaction conditions means that the process is less susceptible to minor fluctuations in temperature or reagent quality, ensuring consistent output even with varying raw material batches. This stability is crucial for maintaining continuous production schedules and meeting tight delivery deadlines for downstream pharmaceutical clients. Additionally, the reduced hazard profile of the reagents simplifies logistics and storage requirements, lowering the total cost of ownership for inventory management.
- Scalability and Environmental Compliance: The transition from batch to continuous flow or larger batch reactors is facilitated by the homogeneous nature of the catalytic system and the absence of gas-evolving hazardous reagents. The method's alignment with green chemistry principles, specifically atom economy and the use of safer solvents, positions manufacturers favorably against tightening environmental regulations. Waste streams are significantly less toxic compared to traditional fluorination methods, simplifying effluent treatment and reducing the environmental impact of the manufacturing site. This scalability ensures that the technology can grow with demand, supporting the commercial scale-up of complex polymer additives or drug candidates without technical bottlenecks.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this difluoroalkylation technology in industrial settings. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation to ensure accuracy. Understanding these nuances helps stakeholders make informed decisions about integrating this method into their existing portfolios. The responses cover catalyst selection, substrate compatibility, and safety protocols essential for safe operation.
Q: What are the primary advantages of using cobalt over palladium for this difluoroalkylation?
A: Cobalt is an earth-abundant base metal that offers significant cost reductions compared to precious palladium catalysts. Furthermore, the cobalt-NHC system described in CN108997193B operates efficiently without the need for pre-functionalized substrates, improving atom economy.
Q: Which heterocyclic substrates are compatible with this synthetic method?
A: The method demonstrates broad substrate scope, successfully functionalizing 2-position unsubstituted indoles, benzofurans, pyrroles, thiophenes, benzimidazoles, and azaindoles with high regioselectivity.
Q: Does this process require hazardous fluorinating reagents like DAST?
A: No, this method avoids dangerous deoxofluorination reagents such as DAST or SF4. Instead, it utilizes commercially available and safer ethyl difluorobromoacetate as the difluoroalkyl source.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Difluoroalkyl Heterocyclic Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of cobalt-catalyzed C-H activation in reshaping the landscape of fluorinated intermediate production. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to market supply is seamless. Our facilities are equipped with rigorous QC labs capable of verifying the structural integrity and purity of these complex organofluorine molecules, adhering to stringent purity specifications required by top-tier pharmaceutical companies. We are committed to leveraging this patented technology to deliver high-quality intermediates that accelerate your drug development timelines while maintaining cost efficiency.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this cobalt-based methodology for your supply chain. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments for your target difluoroalkyl heterocycles. Let us collaborate to build a more resilient and cost-effective supply chain for your next-generation therapeutic or agrochemical products.
