Scaling High-Performance Dithienopyrrole Oligomers for Next-Gen Organic Electronics
Scaling High-Performance Dithienopyrrole Oligomers for Next-Gen Organic Electronics
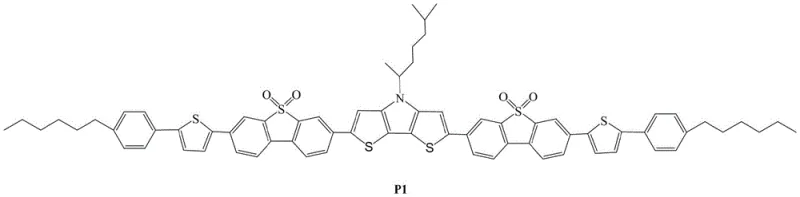
The rapid evolution of the organic electronics sector demands materials that offer not only superior optoelectronic performance but also robust thermal stability and processability. A significant advancement in this domain is documented in Chinese Patent CN109206436B, which discloses a novel oligomeric thiophene derivative utilizing dithienopyrrole as a central electron-donating unit. This specific molecular architecture represents a strategic leap forward for manufacturers of Organic Light-Emitting Diodes (OLED) and Organic Solar Cells (OSC), addressing critical challenges related to energy level alignment and intermolecular packing. By integrating a rigid S,S-dioxo-dibenzothiophene electron-accepting unit with a hexylphenylthienyl end-group, the resulting material achieves a balanced energy gap and enhanced planarity. For R&D directors and procurement specialists alike, understanding the synthesis and commercial viability of this compound is paramount for securing a competitive edge in the high-value display and energy markets.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of conjugated oligomers for semiconductor applications has been plagued by issues regarding structural irregularity and insufficient thermal resilience. Conventional polythiophenes often suffer from batch-to-batch variability in molecular weight distribution, which directly translates to inconsistent device performance and yield losses during mass production. Furthermore, many legacy synthetic routes rely on harsh reaction conditions that can degrade sensitive functional groups or introduce difficult-to-remove metallic impurities, necessitating costly and time-consuming purification steps. The lack of rigid planar structures in older generation materials often results in poor π-π stacking, limiting charge carrier mobility and ultimately reducing the efficiency of the final electronic device. These inherent limitations create significant bottlenecks for supply chain managers who require consistent, high-purity feedstocks to maintain uninterrupted production lines.
The Novel Approach
The methodology outlined in the patent introduces a sophisticated Donor-Acceptor-Donor (D-A-D) strategy that fundamentally overcomes these historical constraints. By employing a Stille cross-coupling reaction to link a specifically designed dithienopyrrole core with dibenzothiophene-S,S-dioxide units, the process ensures a well-defined molecular weight and a highly planar backbone. This structural precision is critical because it facilitates strong intermolecular π-π interactions, which are essential for efficient charge transport in solid-state films. Moreover, the incorporation of the sulfone group within the dibenzothiophene unit acts as a powerful electron-withdrawing moiety, effectively lowering the HOMO energy level to approximately -5.48 eV. This tuning of electronic properties is achieved without compromising thermal stability, as the material demonstrates resilience up to 300°C, thereby offering a reliable solution for high-temperature processing environments common in industrial manufacturing.
Mechanistic Insights into Stille Cross-Coupling and D-A Architecture
The core of this synthesis lies in the palladium-catalyzed Stille cross-coupling reaction, a transformative tool in organic synthesis known for its tolerance of various functional groups and mild reaction conditions. In this specific application, the mechanism involves the oxidative addition of a palladium(0) catalyst, specifically tetrakis(triphenylphosphine)palladium, into the carbon-bromine bond of the 3-bromo-7-[5-(4-n-hexylphenyl)-2-thienyl]dibenzothiophene-S,S-dioxide intermediate. This is followed by transmetallation with the organotin species, 2,6-bis(trimethyltin)-N-(1,5-dimethylhexyl)dithieno[3,2-b:2',3'-d]pyrrole, which serves as the electron-rich donor center. The final reductive elimination step forge the carbon-carbon bonds that connect the donor and acceptor units, regenerating the active palladium catalyst. The choice of solvent, typically anhydrous toluene or N,N-dimethylformamide, plays a crucial role in solubilizing these large, conjugated intermediates while maintaining the stability of the catalytic cycle at temperatures ranging from 100°C to 120°C.
Beyond the coupling mechanism itself, the resulting molecular architecture is engineered for optimal electronic performance. The central dithienopyrrole unit provides a rich source of electrons, while the flanking S,S-dioxo-dibenzothiophene units act as electron sinks, creating an internal push-pull system that narrows the optical band gap to a desirable 2.24 eV. This D-A arrangement promotes intramolecular charge transfer, which is vital for light absorption and emission processes in optoelectronic devices. Additionally, the presence of long alkyl chains, such as the 1,5-dimethylhexyl group on the pyrrole nitrogen and the n-hexyl chains on the terminal phenyl rings, ensures sufficient solubility in common organic solvents. This solubility is a key factor for solution-processable manufacturing techniques like spin-coating or inkjet printing, allowing for the formation of uniform thin films with minimal defects.
How to Synthesize Dithienopyrrole-Based Oligomer Efficiently
The preparation of this high-performance oligomer requires strict adherence to inert atmosphere conditions and precise stoichiometric control to maximize yield and purity. The process begins with the careful drying of solvents and reagents to prevent catalyst deactivation, followed by a prolonged heating period to drive the coupling reaction to completion. While the laboratory-scale examples utilize standard Schlenk techniques, the underlying chemistry is robust enough to be adapted for larger scale reactors with appropriate engineering controls. The purification protocol described, involving extraction, gradient column chromatography, and a final precipitation step, is particularly noteworthy for its ability to remove tin residues and homocoupling byproducts. For a detailed breakdown of the standardized operating procedures and safety protocols required for this synthesis, please refer to the technical guide below.
- Prepare the reaction vessel under inert atmosphere (nitrogen or argon) and dissolve the brominated acceptor intermediate and stannylated donor core in anhydrous toluene or DMF.
- Add tetrakis(triphenylphosphine)palladium catalyst and heat the mixture to 100-120°C, maintaining stirring for 12 to 72 hours to ensure complete cross-coupling.
- Upon completion, extract the crude product with dichloromethane, purify via silica gel column chromatography using petroleum ether/dichloromethane, and finalize purity through precipitation in methanol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel dithienopyrrole derivative offers substantial strategic benefits beyond mere performance metrics. The synthetic route described eliminates the need for exotic or prohibitively expensive catalysts, relying instead on established palladium systems that are widely available in the global chemical market. This accessibility reduces the risk of supply disruptions and allows for more predictable budgeting and sourcing strategies. Furthermore, the thermal stability of the final product implies a longer shelf life and reduced sensitivity to storage conditions, minimizing waste due to degradation during transit or warehousing. The ability to purify the material via precipitation rather than solely relying on preparative HPLC suggests a pathway to significantly lower production costs at scale, making high-end organic semiconductors more economically viable for mass-market applications.
- Cost Reduction in Manufacturing: The synthetic pathway leverages a one-pot coupling strategy that minimizes the number of isolation steps required for intermediates, thereby reducing solvent consumption and labor hours. By utilizing a precipitation method for final purification using methanol and tetrachloroethane, the process avoids the extensive use of high-cost chromatographic media typically associated with oligomer purification. This streamlined approach translates directly into a lower cost of goods sold (COGS), allowing downstream device manufacturers to achieve better margins without sacrificing material quality. Additionally, the high thermal stability reduces the likelihood of batch rejection due to thermal degradation during device fabrication, further enhancing overall process economics.
- Enhanced Supply Chain Reliability: The starting materials, including the brominated dibenzothiophene derivatives and stannylated pyrroles, are constructed from commercially available building blocks, ensuring a stable and diversified supply base. The robustness of the Stille coupling reaction means that the process is less susceptible to minor fluctuations in reaction parameters, leading to higher batch-to-batch consistency and reliability. This consistency is critical for supply chain planners who need to guarantee continuous delivery schedules to their own customers in the electronics sector. The reduced sensitivity to moisture and oxygen compared to more reactive organometallic methods also simplifies logistics and storage requirements.
- Scalability and Environmental Compliance: The use of standard solvents like toluene and dichloromethane, combined with a clear workup procedure, facilitates easier scale-up from kilogram to multi-ton production capacities. The process design inherently supports efficient solvent recovery and recycling systems, which aligns with increasingly stringent environmental regulations and corporate sustainability goals. By minimizing the generation of heavy metal waste through optimized catalyst loading and effective purification, the manufacturing footprint is significantly reduced. This environmental compatibility not only mitigates regulatory risks but also enhances the brand value of the final electronic products as being produced through greener chemical processes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this dithienopyrrole-based oligomer. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a factual basis for decision-making. Understanding these nuances is essential for evaluating the feasibility of integrating this material into existing product lines or developing new device architectures.
Q: What are the thermal stability characteristics of this dithienopyrrole oligomer?
A: According to patent data, the oligomer exhibits excellent thermal stability with an initial melting point around 251°C and no decomposition observed up to 300°C, making it suitable for high-temperature device processing.
Q: Why is the Stille cross-coupling reaction preferred for this synthesis?
A: Stille coupling allows for the precise linkage of the electron-rich dithienopyrrole core with the electron-deficient dibenzothiophene dioxide units under relatively mild conditions, ensuring high structural fidelity.
Q: How does the molecular architecture affect electronic properties?
A: The Donor-Acceptor-Donor architecture creates a low HOMO energy level (-5.48 eV) and moderate band gap, facilitating efficient charge transport and π-π stacking interactions essential for OLED and OSC performance.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dithienopyrrole Derivative Supplier
The potential of this dithienopyrrole-based oligomer to revolutionize OLED and OSC technologies is immense, yet realizing this potential requires a manufacturing partner with deep technical expertise and scalable infrastructure. NINGBO INNO PHARMCHEM stands ready to support your development efforts, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped with state-of-the-art reactors capable of handling air-sensitive reactions like Stille couplings under strict inert atmospheres, ensuring the highest quality standards. We maintain stringent purity specifications and operate rigorous QC labs to verify that every batch meets the exacting requirements of the organic electronics industry, from HOMO/LUMO levels to trace metal content.
We invite you to collaborate with us to optimize this synthesis for your specific volume requirements and cost targets. Our technical team is prepared to provide a Customized Cost-Saving Analysis that identifies opportunities to further streamline the supply chain and reduce overall expenditure. Please contact our technical procurement team today to request specific COA data, discuss route feasibility assessments, and explore how we can jointly bring this advanced material to market efficiently.
