Advanced Visible Light Catalysis for High-Purity Chiral Amine Intermediates
The pharmaceutical industry is currently witnessing a paradigm shift towards greener, more sustainable synthetic methodologies, particularly in the realm of chiral amine production. Patent CN110194735B introduces a groundbreaking visible light asymmetric catalytic synthesis method for chiral 3-(2-pyridine)-3-aryl substituted amine compounds, addressing critical inefficiencies in traditional manufacturing. This technology leverages a dual-catalyst system comprising an organic photocatalyst (DPZ) and a chiral spiro phosphonic acid to drive conjugate addition-protonation reactions under mild conditions. For R&D directors and procurement specialists, this represents a significant opportunity to enhance the purity profile of key intermediates while simultaneously reducing the environmental footprint of the synthesis process. The ability to generate high-value chiral scaffolds without transition metals aligns perfectly with modern regulatory demands for cleaner drug substance production.
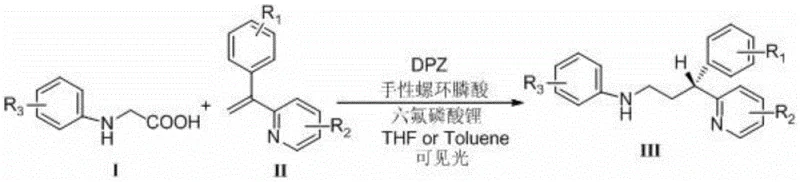
Historically, the synthesis of chiral 3-(2-pyridine)-3-aryl substituted amines has relied heavily on transition metal-catalyzed asymmetric hydrogenation of allylamines. Literature references, such as those by Botteghi and Marchetti, indicate that these conventional methods often suffer from significant limitations, including moderate yields around 50% and suboptimal optical purity frequently below 60% ee. Furthermore, the reliance on precious metals introduces complex downstream processing requirements to ensure residual metal levels meet strict pharmacopeial standards. In stark contrast, the novel approach detailed in the patent utilizes visible light irradiation to activate the photocatalyst, facilitating a radical mechanism that proceeds with exceptional stereoselectivity. This metal-free strategy not only simplifies the purification workflow but also drastically improves the overall atom economy and operational safety by avoiding high-pressure hydrogenation equipment.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
The traditional reliance on transition metal catalysis for constructing chiral amine bonds presents multifaceted challenges for large-scale manufacturing operations. Primary among these is the inherent instability and cost associated with noble metal catalysts, which can lead to batch-to-batch variability and inflated raw material costs. Additionally, the removal of trace metal contaminants from the final active pharmaceutical ingredient (API) requires specialized scavenging resins or extensive recrystallization processes, adding time and expense to the production timeline. The moderate enantioselectivity observed in older hydrogenation protocols often necessitates additional resolution steps, effectively halving the theoretical yield and generating substantial chemical waste. These factors collectively create a bottleneck for supply chain managers seeking reliable, cost-effective sources of high-purity chiral intermediates for antihistamine and other therapeutic classes.
The Novel Approach
The visible light catalytic method described in CN110194735B offers a robust solution by employing an organic photocatalyst, DPZ, in conjunction with a chiral Brønsted acid catalyst. This system operates under ambient pressure and relatively low temperatures ranging from -35°C to -40°C, utilizing inexpensive blue LED light sources to drive the reaction. The mechanism involves the generation of radical intermediates that undergo highly stereocontrolled addition to the olefinic bond, guided by the chiral environment of the phosphoric acid. This results in products with excellent enantiomeric excess values, consistently exceeding 90% ee across a broad range of substrates. By eliminating heavy metals and harsh reaction conditions, this approach streamlines the manufacturing process, reduces waste generation, and ensures a cleaner impurity profile for the final drug substance.
Mechanistic Insights into Visible Light Asymmetric Catalysis
The core of this innovative synthesis lies in the synergistic interaction between the DPZ photocatalyst and the chiral spiro phosphonic acid. Upon irradiation with visible light, the DPZ molecule absorbs photons to reach an excited state, enabling it to participate in single-electron transfer processes with the N-aryl substituted glycine substrate. This generates an alpha-amino radical species which subsequently adds to the electron-deficient double bond of the alpha-aryl-alpha-(2-pyridine) substituted terminal olefin. The chiral phosphoric acid plays a dual role: it activates the imine or olefin substrate through hydrogen bonding interactions and provides a chiral pocket that dictates the facial selectivity of the radical addition. This precise control over the transition state geometry is what allows for the high levels of stereoinduction observed in the experimental data.
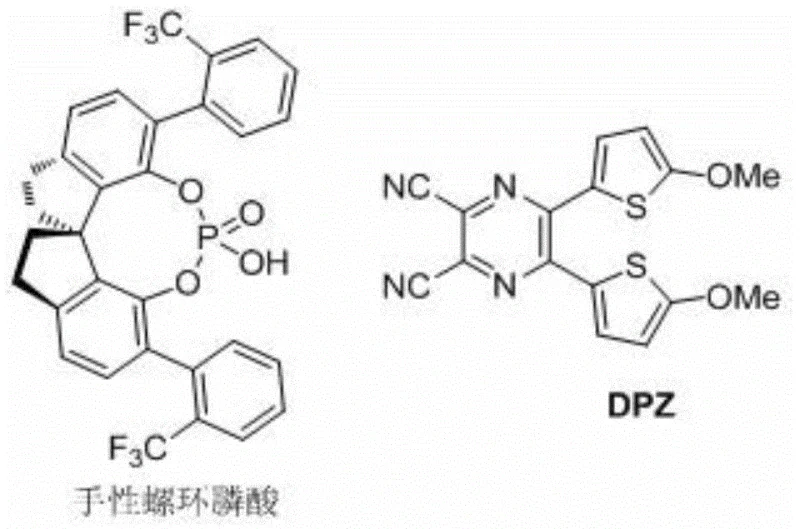
Impurity control in this system is inherently superior due to the mildness of the reaction conditions and the specificity of the radical pathway. Unlike thermal radical reactions which can lead to indiscriminate bond formation and polymerization, the photo-induced process is tightly regulated by the catalyst concentration and light intensity. The use of lithium hexafluorophosphate as an additive further stabilizes the ionic intermediates and enhances the solubility of the catalyst species in organic solvents like THF or toluene. This stabilization minimizes side reactions such as homocoupling or over-reduction, ensuring that the crude reaction mixture is dominated by the desired chiral product. Consequently, the downstream purification via standard column chromatography is highly efficient, yielding products with minimal contamination from structural analogs or degradation byproducts.
How to Synthesize Chiral 3-(2-pyridine)-3-aryl Substituted Amines Efficiently
Implementing this synthesis route requires careful attention to reaction parameters, particularly regarding the exclusion of oxygen and moisture which can quench the radical intermediates. The protocol typically involves dissolving the DPZ catalyst in toluene, removing the solvent to form a thin film, and then adding the substrates and chiral acid in dry THF. The reaction vessel must be rigorously degassed and maintained under an inert argon atmosphere throughout the irradiation period. While the general procedure is straightforward, optimization of the light source distance and temperature control is critical for maximizing yield and enantioselectivity. Detailed standardized synthesis steps for replicating this high-efficiency process are provided in the guide below.
- Prepare the reaction mixture by dissolving DPZ photocatalyst in toluene, then adding N-aryl substituted glycine, alpha-aryl-alpha-(2-pyridine) substituted terminal olefin, chiral spiro phosphonic acid, and lithium hexafluorophosphate in THF.
- Degas the Schlenk tube by vacuumizing and filling with argon three times, then freeze the mixture at -80°C before restoring to room temperature and filling with argon protection.
- Stir the reaction at -35°C to -40°C under 3W blue LED irradiation (450-455nm) for approximately 60 hours, then purify the product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this visible light catalytic technology translates into tangible operational improvements and risk mitigation. The elimination of transition metals removes a major variable from the supply chain, reducing dependency on volatile precious metal markets and simplifying vendor qualification processes. Furthermore, the mild reaction conditions allow for the use of standard glass-lined reactors equipped with LED arrays, avoiding the need for specialized high-pressure hydrogenation vessels. This flexibility enhances manufacturing agility, allowing facilities to switch between different product campaigns with minimal retooling downtime. The overall process robustness ensures consistent supply continuity, which is paramount for meeting the demanding delivery schedules of global pharmaceutical clients.
- Cost Reduction in Manufacturing: The most significant economic driver for this technology is the complete removal of expensive transition metal catalysts and the associated purification costs. By utilizing organic photocatalysts and readily available chiral acids, the raw material cost profile is significantly optimized. Additionally, the high yields and excellent stereoselectivity reduce the need for recycling mother liquors or performing chiral resolutions, which are traditionally resource-intensive operations. The simplified workup procedure also lowers utility consumption and labor hours per kilogram of product, contributing to a leaner cost structure for API intermediate manufacturing.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, namely N-aryl substituted glycines and alpha-aryl-alpha-(2-pyridine) substituted terminal olefins, are commercially available and chemically stable. This ensures a secure upstream supply base that is less susceptible to geopolitical disruptions compared to rare earth metals. The reaction's tolerance to various functional groups means that a single platform technology can be adapted to produce a wide library of analogues, providing supply chain resilience against demand fluctuations for specific drug candidates. This versatility allows manufacturers to maintain strategic stockpiles of key intermediates with confidence.
- Scalability and Environmental Compliance: Scaling photochemical reactions has historically been challenging, but advancements in flow chemistry and LED reactor design have made kilogram-scale production feasible. The absence of heavy metal waste streams simplifies environmental compliance and wastewater treatment, aligning with increasingly stringent global regulations on industrial effluents. The energy efficiency of LED lighting combined with low-temperature operation further reduces the carbon footprint of the manufacturing process. These factors collectively position this technology as a future-proof solution for sustainable chemical production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this visible light catalytic method. Understanding these details is crucial for evaluating the feasibility of integrating this process into existing manufacturing pipelines. The answers are derived directly from the experimental data and technical specifications outlined in the patent documentation.
Q: What are the optical purity and yield advantages of this visible light method?
A: According to patent CN110194735B, this method achieves optical purity (ee values) up to 94% and yields ranging from 73% to 91%, significantly outperforming traditional transition metal hydrogenation methods which often yield less than 60% ee.
Q: Does this process involve heavy metal catalysts?
A: No, the process utilizes an organic photocatalyst (DPZ) and a chiral spiro phosphonic acid, eliminating the need for transition metals and the subsequent costly removal steps associated with heavy metal residues.
Q: What is the substrate scope for this synthesis?
A: The method demonstrates broad substrate tolerance, accommodating various substituents such as F, Cl, Br, Me, and OMe on the aryl rings of both the glycine and the olefin components, making it versatile for diverse API intermediate production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral 3-(2-pyridine)-3-aryl Substituted Amine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of visible light catalysis in modern pharmaceutical synthesis. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like this are successfully translated into robust industrial processes. Our state-of-the-art facilities are equipped with advanced photochemical reactors and rigorous QC labs capable of verifying stringent purity specifications, including precise enantiomeric excess measurements. We are committed to delivering high-quality chiral intermediates that meet the exacting standards of the global pharmaceutical industry.
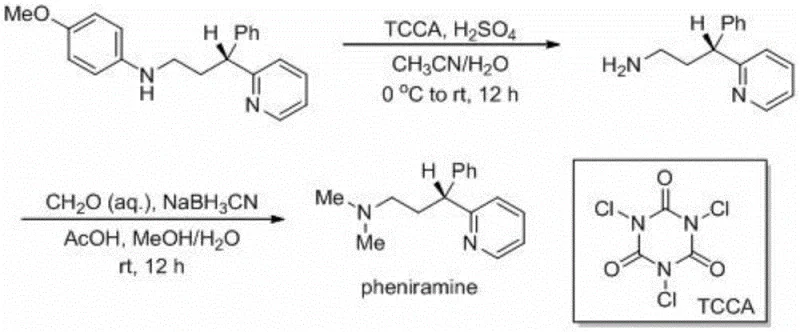
We invite potential partners to engage with our technical team to explore how this cost-effective synthesis route can be tailored to your specific project needs. By leveraging our expertise in process optimization, we can provide a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this metal-free methodology. Please contact our technical procurement team to request specific COA data for relevant intermediates and comprehensive route feasibility assessments. Let us collaborate to accelerate your drug development timeline while achieving superior cost and quality outcomes.
