Advanced Metal-Free Synthesis of Benzothiazole C2 Arylation Derivatives for Commercial Scale Production
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the benzothiazole C2-aryl bond has relied heavily on transition metal catalysis, predominantly utilizing palladium or copper complexes to mediate the cross-coupling between benzothiazoles and aryl halides or boronic acids. While effective in laboratory settings, these conventional methodologies present significant hurdles for industrial pharmaceutical manufacturing, primarily due to the high cost of precious metal catalysts and the stringent regulatory requirements regarding residual heavy metals in active pharmaceutical ingredients (APIs). The necessity for pre-functionalized substrates, such as aryl halides or organoboron reagents, further inflates raw material costs and complicates the supply chain, as these intermediates often require multi-step synthesis themselves. Moreover, the removal of trace transition metals from the final product to meet ICH Q3D guidelines often necessitates additional purification steps, such as scavenging or recrystallization, which drastically reduce overall process efficiency and yield. These factors collectively create a bottleneck in the cost reduction in pharmaceutical intermediates manufacturing, driving the urgent need for more sustainable and economically viable synthetic alternatives.
The Novel Approach
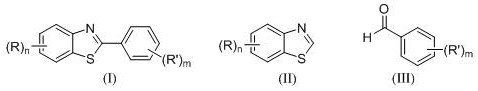
In stark contrast to traditional metal-catalyzed routes, the innovative methodology disclosed in patent CN111039893B introduces a transition metal-free oxidative coupling strategy that fundamentally reshapes the economic and operational landscape of benzothiazole derivative synthesis. This novel approach leverages a dual-component oxidation system comprising N-chlorosuccinimide (NCS) as a radical initiator and tert-butyl hydroperoxide (TBHP) as the terminal oxidant, operating efficiently within a dimethyl sulfoxide (DMSO) solvent matrix. By utilizing readily available and inexpensive substituted benzaldehydes as the arylating agents instead of pre-functionalized halides, the process significantly simplifies the starting material procurement and reduces the overall carbon footprint of the synthesis. The reaction proceeds smoothly under a nitrogen atmosphere at elevated temperatures, demonstrating remarkable tolerance to a wide variety of functional groups on both the benzothiazole and benzaldehyde scaffolds. This metal-free paradigm not only eliminates the risk of heavy metal contamination but also streamlines the downstream processing workflow, offering a robust pathway for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into NCS/TBHP-Mediated Radical Oxidative Coupling
The mechanistic underpinning of this transformation involves a sophisticated radical cascade initiated by the interaction between NCS and TBHP, which serves as the engine driving the C-H functionalization at the C2 position of the benzothiazole ring. Upon heating, NCS facilitates the homolytic cleavage of the O-O bond in TBHP, generating highly reactive tert-butoxy and hydroxyl radicals that are crucial for the subsequent activation of the benzaldehyde substrate. These radical species abstract the formyl hydrogen from the benzaldehyde, leading to the formation of an acyl radical intermediate, which acts as the key nucleophilic species in the coupling sequence. The electron-deficient nature of the benzothiazole ring, particularly at the C2 position, makes it an ideal electrophilic trap for the generated acyl radicals, facilitating the formation of a new carbon-carbon bond through a radical addition mechanism. Following the addition step, the intermediate undergoes oxidation and aromatization, likely mediated by the excess oxidant in the system, to restore the aromaticity of the benzothiazole core and deliver the final 2-arylbenzothiazole product with high structural fidelity.
Beyond the primary radical generation, the role of the solvent, DMSO, is pivotal in stabilizing the polar transition states and enhancing the solubility of the radical intermediates throughout the reaction coordinate. As a strong Lewis base, DMSO coordinates with the radical species and potentially assists in the proton transfer steps required for the final aromatization, thereby improving the overall reaction kinetics and yield. The careful balance of the NCS/TBHP ratio ensures a steady flux of radicals without causing excessive over-oxidation or decomposition of the sensitive heterocyclic substrate, which is a common pitfall in aggressive radical chemistries. Furthermore, the inert nitrogen atmosphere prevents the quenching of radical intermediates by atmospheric oxygen, ensuring that the oxidative potential of the system is directed exclusively towards the desired C-H activation pathway. This precise control over the radical environment allows for the synthesis of high-purity OLED material precursors and pharmaceutical intermediates with minimal byproduct formation, addressing critical quality concerns for R&D directors focused on impurity profiling.
How to Synthesize Substituted Benzothiazole C2 Arylate Derivatives Efficiently
The practical execution of this synthesis protocol is designed for operational simplicity while maintaining rigorous control over reaction parameters to ensure reproducibility and safety on scale. The process begins with the precise weighing of substituted benzothiazole and substituted benzaldehyde substrates, which are then charged into a reaction vessel along with the radical initiator NCS and the oxidant TBHP in the appropriate stoichiometric ratios. DMSO is added as the reaction medium, and the mixture is subjected to heating under a nitrogen blanket to initiate the radical cascade, with temperature and time being critical variables that dictate the conversion efficiency and product selectivity. Detailed standardized synthesis steps, including specific workup procedures and purification protocols via column chromatography, are essential for achieving the reported yields and purity levels across different substrate combinations.
- Mix substituted benzothiazole and substituted benzaldehyde with N-chlorosuccinimide (NCS) and tert-butyl hydroperoxide (TBHP) in DMSO solvent.
- Stir the reaction mixture under a nitrogen atmosphere at 120°C for approximately 12 hours to facilitate radical oxidative coupling.
- Upon completion, extract the reaction liquid with dichloromethane or ethyl acetate, concentrate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this metal-free oxidative coupling technology offers profound advantages that directly impact the bottom line and supply chain resilience for chemical manufacturers. The elimination of transition metal catalysts removes a major cost driver associated with precious metal procurement and the specialized waste treatment required for heavy metal disposal, leading to substantial cost savings in the overall production budget. Additionally, the reliance on commodity chemicals like benzaldehydes and DMSO, which are produced on a massive global scale, ensures a stable and reliable supply chain that is less susceptible to the geopolitical fluctuations often affecting specialized catalyst markets. The simplified purification process, devoid of metal scavenging steps, reduces the consumption of auxiliary materials and shortens the manufacturing cycle time, thereby enhancing the throughput capacity of existing production facilities. These operational efficiencies translate into a more competitive pricing structure for the final intermediates, making this technology an attractive option for cost reduction in pharmaceutical intermediates manufacturing without compromising on quality or regulatory compliance.
- Cost Reduction in Manufacturing: The removal of expensive palladium or copper catalysts from the process equation fundamentally alters the cost structure of benzothiazole derivative production, as it eradicates the need for costly ligand systems and metal recovery operations. By utilizing inexpensive oxidants like TBHP and NCS, which are widely available from bulk chemical suppliers, the raw material expenditure is significantly lowered compared to traditional cross-coupling methods. Furthermore, the avoidance of pre-functionalized aryl halides or boronic acids reduces the number of synthetic steps required to prepare starting materials, compounding the savings through reduced labor and utility consumption. This streamlined approach allows manufacturers to offer high-purity pharmaceutical intermediates at a more aggressive price point, providing a distinct competitive advantage in the global market.
- Enhanced Supply Chain Reliability: The reliance on universally available feedstocks such as substituted benzaldehydes and common solvents like DMSO mitigates the risk of supply disruptions that often plague specialized reagent markets. Unlike transition metal catalysts, which may face supply constraints due to mining limitations or refining bottlenecks, the reagents for this process are produced by a diverse network of chemical manufacturers worldwide, ensuring continuous availability. This diversification of the supply base enhances the robustness of the production schedule, allowing for consistent delivery timelines even during periods of market volatility. For supply chain heads, this translates to reduced lead time for high-purity pharmaceutical intermediates and greater flexibility in inventory management, securing the continuity of drug development pipelines.
- Scalability and Environmental Compliance: The metal-free nature of this synthesis aligns perfectly with modern green chemistry principles and increasingly stringent environmental regulations regarding heavy metal discharge in industrial effluents. Scaling this process from laboratory to commercial production does not require complex engineering controls for metal containment or specialized waste streams, simplifying the regulatory approval process for new manufacturing sites. The use of DMSO, a solvent with well-established recovery and recycling protocols, further minimizes the environmental footprint of the operation. Consequently, this technology facilitates the commercial scale-up of complex pharmaceutical intermediates with a lower barrier to entry for environmental compliance, ensuring long-term sustainability and operational license.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel benzothiazole arylation technology, derived directly from the experimental data and scope defined in the patent literature. These insights are intended to clarify the operational feasibility and strategic benefits for stakeholders evaluating this synthetic route for their specific applications. Understanding the nuances of substrate compatibility and reaction conditions is crucial for successful technology transfer and process optimization.
Q: Does this synthesis method require transition metal catalysts?
A: No, the method described in patent CN111039893B utilizes a metal-free NCS/TBHP dual oxidation system, eliminating the need for expensive palladium or copper catalysts and simplifying purification.
Q: What is the substrate scope for this benzothiazole arylation?
A: The process demonstrates a wide substrate range, accommodating various substituents on both the benzothiazole ring (e.g., chloro, nitro, methoxy) and the benzaldehyde phenyl ring (e.g., methyl, halo, trifluoromethyl).
Q: Is this process suitable for large-scale pharmaceutical manufacturing?
A: Yes, the use of common solvents like DMSO, inexpensive oxidants, and the absence of sensitive metal catalysts make this route highly scalable and compliant with stringent pharmaceutical impurity guidelines.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Substituted Benzothiazole C2 Arylate Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the metal-free NCS/TBHP oxidation system described in patent CN111039893B for the next generation of pharmaceutical intermediate production. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from bench-scale discovery to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of benzothiazole derivatives meets the highest industry standards for potency and impurity profiles. We are committed to leveraging this advanced chemistry to deliver superior value to our global partners, combining technical expertise with operational excellence.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project requirements and volume needs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this metal-free protocol for your supply chain. We encourage you to contact us today to obtain specific COA data for our benchmark batches and to initiate a dialogue regarding route feasibility assessments for your target molecules. Let us collaborate to optimize your synthesis strategy and secure a reliable, cost-effective supply of critical benzothiazole intermediates for your drug development programs.
