Advanced Manufacturing of Iopamidol Intermediates for Global Pharmaceutical Supply Chains
The pharmaceutical industry continuously seeks robust synthetic routes for high-volume diagnostic agents, particularly non-ionic iodinated X-ray contrast media like Iopamidol. A significant technological breakthrough in this domain is detailed in patent CN103781758A, which discloses a novel process for the production of triiodinated contrast agents via a unique chemical intermediate. This patent introduces (S)-5-(2-acetoxypropionylamino)-2,4,6-triiodoisophthalic acid, a compound that serves as a pivotal precursor, enabling a more efficient and economically viable manufacturing pathway. By fundamentally restructuring the sequence of functional group introductions, this methodology addresses long-standing challenges regarding atom economy and impurity control. For R&D directors and procurement specialists, understanding this shift is critical, as it represents a move away from protection-heavy strategies toward a more direct acylation-first approach. The ability to produce high-purity intermediates with minimal downstream purification requirements offers a compelling value proposition for large-scale API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Iopamidol has been plagued by inefficiencies inherent in early-stage chiral introduction or excessive protection-deprotection sequences. Conventional methods, such as those described in earlier literature, often involve reacting 5-amino-2,4,6-triiodoisophthaloyl dichloride with chiral reagents at the very beginning of the synthesis. While chemically feasible, this approach necessitates the use of expensive reagents like 2-amino-1,3-propanediol (serinol) at a stage where yield losses are most costly. Alternatively, other routes attempt to introduce the chiral center later but rely heavily on protecting groups for primary alcohols. These protection strategies inevitably increase the number of synthetic steps, generate substantial chemical waste, and lower the overall atom usage. From a supply chain perspective, these multi-step processes introduce multiple points of failure, increasing the risk of batch variability and extending lead times for critical raw materials.
The Novel Approach
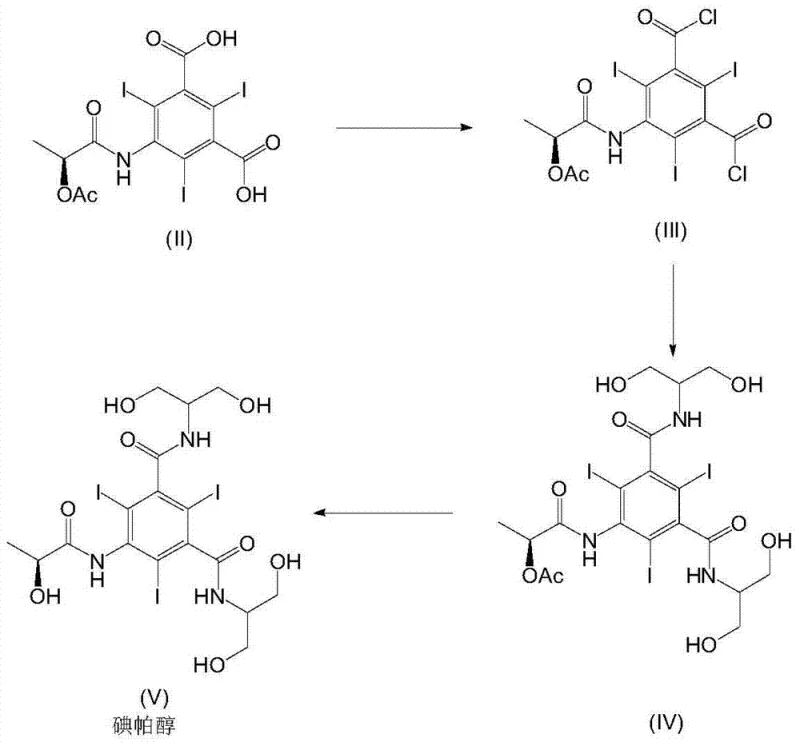
The innovative strategy outlined in the patent data circumvents these bottlenecks by utilizing 5-amino-2,4,6-triiodoisophthalic acid as the starting material and performing an acylation with (S)-1-chloro-1-oxopropan-2-yl acetate in the initial stages. This creates the stable Formula II intermediate, which can subsequently be converted into the acid dichloride (Formula III) before the final amidation. This sequence allows for the delayed introduction of the expensive serinol reagent until the penultimate steps, significantly optimizing cost efficiency. Furthermore, the process avoids the need for complex protecting group manipulations, thereby streamlining the workflow and reducing environmental impact. The reaction scheme below illustrates this streamlined progression from the novel acid intermediate to the final Iopamidol product, highlighting the logical flow that minimizes side reactions.
Mechanistic Insights into Acylation and Chlorination Chemistry
The core of this technological advancement lies in the precise control of the acylation reaction between the triiodinated amine and the chiral acyl chloride. The process utilizes dimethylacetamide (DMA) as a polar aprotic solvent, which is essential for solubilizing the highly iodinated aromatic starting material. The reaction is typically conducted at elevated temperatures, specifically around 50°C, to ensure complete conversion while maintaining the integrity of the chiral center. The stoichiometry is carefully managed, often employing an excess of the acylating agent (at least 1.5:1 equivalents) to drive the reaction to completion. Following the formation of the Formula II compound, the subsequent chlorination step employs phosphorus pentachloride (PCl5) rather than thionyl chloride. This choice is mechanistically significant as PCl5 offers a cleaner conversion to the acid dichloride with fewer gaseous by-products and reduced risk of side reactions that could compromise the stereochemical purity of the molecule.
Impurity control is a paramount concern for any regulatory submission, and this route demonstrates superior performance in minimizing related substances. The specific reaction conditions prevent the formation of common impurities such as acetyl and hydroxyacetyl analogues, which are often difficult to remove in traditional syntheses. By isolating the intermediate as a high-purity solid (up to 99.9% purity by HPLC) through controlled precipitation in water, the process effectively purges soluble impurities before they can propagate to the final API. The structural integrity of the intermediate is confirmed through rigorous spectroscopic analysis, ensuring that the chiral configuration is preserved throughout the harsh chlorination and amidation steps. The image below details the specific transformation from the amino acid starting material to the key acylated intermediate, showcasing the precise structural modification that enables this efficiency.
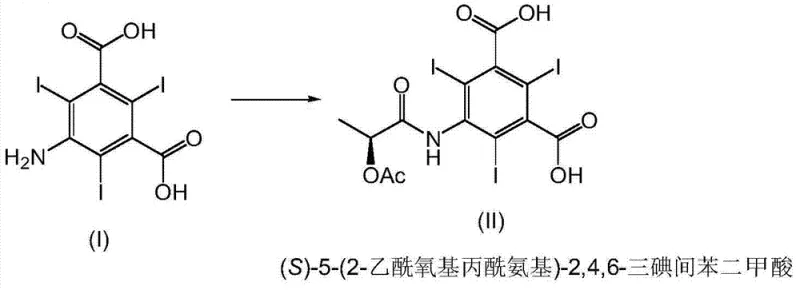
How to Synthesize (S)-5-(2-acetoxypropionylamino)-2,4,6-triiodoisophthalic Acid Efficiently
Executing this synthesis requires strict adherence to temperature controls and solvent ratios to maximize yield and purity. The process begins with the dissolution of the triiodinated amine in DMA, followed by the controlled addition of the chiral acyl chloride. Maintaining the reaction mixture at approximately 50°C for a defined period, typically around 8 hours, is crucial for achieving full conversion without degradation. Once the reaction is complete, the product is precipitated by pouring the mixture into water, a step that leverages the low solubility of the iodinated product in aqueous media to effect purification. The resulting solid is then filtered and dried under vacuum at moderate temperatures to remove residual solvent. For detailed operational parameters and safety considerations regarding the handling of chlorinating agents, please refer to the standardized guide below.
- Acylation of 5-amino-2,4,6-triiodoisophthalic acid with (S)-1-chloro-1-oxopropan-2-yl acetate in DMA at 50°C to form the novel Formula II compound.
- Conversion of the Formula II acid to the corresponding acid dichloride (Formula III) using phosphorus pentachloride in polar solvent.
- Amidation with 2-amino-1,3-propanediol followed by acetate hydrolysis to yield high-purity Iopamidol with minimal impurities.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route translates into tangible strategic benefits beyond mere chemical elegance. The primary advantage lies in the significant optimization of raw material costs. By deferring the use of expensive chiral amino alcohols to the final stages of the synthesis, manufacturers can reduce the capital tied up in work-in-progress inventory and minimize the financial impact of yield losses in early steps. Additionally, the elimination of protection and deprotection steps reduces the total number of unit operations, which directly correlates to lower utility consumption and reduced labor costs per kilogram of finished product. This streamlined approach enhances the overall economic viability of producing high-purity pharmaceutical intermediates, making it a highly attractive option for cost-sensitive markets.
- Cost Reduction in Manufacturing: The process eliminates the need for expensive protecting groups and reduces the number of synthetic steps, leading to substantial cost savings in raw materials and processing time. By avoiding the early use of costly serinol, the financial risk associated with early-stage yield losses is drastically minimized, improving the overall cost of goods sold.
- Enhanced Supply Chain Reliability: The use of commercially available and stable reagents, such as the specific acyl chloride and phosphorus pentachloride, ensures a robust supply chain less susceptible to shortages. The simplified workup procedure, involving simple precipitation and filtration, reduces dependency on complex chromatographic purification, thereby shortening production cycles and improving delivery reliability.
- Scalability and Environmental Compliance: The methodology is inherently scalable, utilizing standard industrial solvents like DMA and straightforward isolation techniques that translate easily from pilot plant to commercial scale. Furthermore, the reduction in chemical waste and the avoidance of hazardous protection chemistry align with modern green chemistry principles, facilitating easier regulatory compliance and waste management.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and process descriptions found in the source documentation, providing clarity on purity standards, scalability, and impurity profiles. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into existing manufacturing frameworks.
Q: How does this novel route improve impurity profiles compared to conventional methods?
A: By introducing the chiral center via acylation early with a stable reagent and delaying the addition of expensive serinol, the process minimizes racemization and reduces acetyl/hydroxyacetyl analogues to trace levels (e.g., <0.005%).
Q: Is this process scalable for commercial API production?
A: Yes, the method utilizes robust solvents like DMA and simple precipitation workups, facilitating scale-up from pilot batches to multi-ton annual production without complex chromatography.
Q: What are the key quality specifications for the intermediate?
A: The novel intermediate (Formula II) achieves HPLC purity up to 99.9%, ensuring that downstream processing yields Iopamidol meeting strict pharmacopoeial standards for related substances.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Iopamidol Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of consistent quality and supply continuity in the pharmaceutical sector. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and reliability. We are committed to delivering stringent purity specifications and maintaining rigorous QC labs to verify that every batch of intermediate meets the highest international standards. Our infrastructure is designed to handle complex halogenated chemistries safely and efficiently, positioning us as a strategic partner for your long-term growth.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can be tailored to your specific production requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits for your organization. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is optimized for both performance and profitability.
