Scalable Double Grignard Synthesis of 3-Ethoxy-4-Carboxyphenylacetic Acid for Global Pharma Supply Chains
Scalable Double Grignard Synthesis of 3-Ethoxy-4-Carboxyphenylacetic Acid for Global Pharma Supply Chains
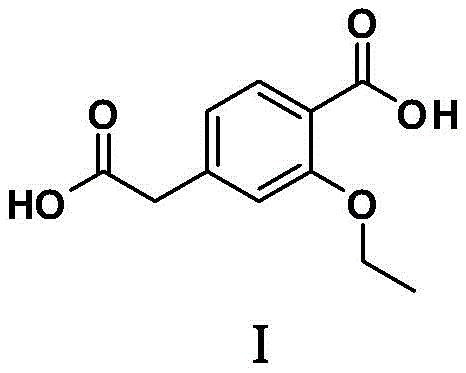
The pharmaceutical industry constantly seeks robust, safe, and cost-effective pathways for critical intermediates, particularly for high-volume drugs like Repaglinide. Patent CN108715576B introduces a transformative methodology for synthesizing 3-ethoxy-4-carboxyphenylacetic acid, a key building block also recognized as Repaglinide Impurity A by the British Pharmacopoeia. This innovative approach leverages a double Grignard reagent strategy, utilizing readily available dihalides as starting materials to construct the dicarboxylic acid framework in a streamlined fashion. By shifting away from hazardous cyanide-based chemistries, this process not only enhances operational safety but also simplifies the downstream purification landscape. For R&D directors and procurement specialists, understanding this shift is crucial for securing a reliable pharmaceutical intermediate supplier capable of delivering high-purity materials without the baggage of toxic waste streams. The structural integrity of the target molecule, essential for its function as both an intermediate and a reference standard, is preserved through mild acidic workup conditions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-ethoxy-4-carboxyphenylacetic acid has relied heavily on hydrolysis reactions that present significant logistical and safety hurdles. One prominent prior art method, disclosed in patent CN 104628518A, involves the simultaneous alkaline hydrolysis of both ester and nitrile groups on a 3-ethoxy-4-ethoxycarbonylphenylacetonitrile precursor. This route is fundamentally flawed for modern green chemistry standards because it necessitates the use of sodium cyanide, a substance with extreme toxicity and stringent regulatory handling requirements. Furthermore, alternative routes described in patents like CN 101830796B require the preparation of complex benzoate esters through multi-step sequences, often involving six or more reaction steps before the final hydrolysis can even occur. These elongated synthetic pathways inherently accumulate impurities, drive up raw material costs, and create bottlenecks in supply chain reliability due to the dependency on multiple specialized precursors. The environmental burden of treating cyanide-containing wastewater and the energy intensity of multi-step syntheses make these conventional methods increasingly obsolete for large-scale commercial manufacturing.
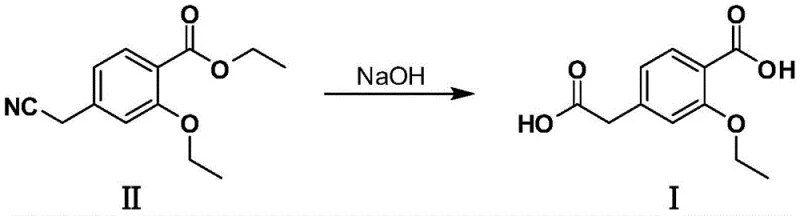
The Novel Approach
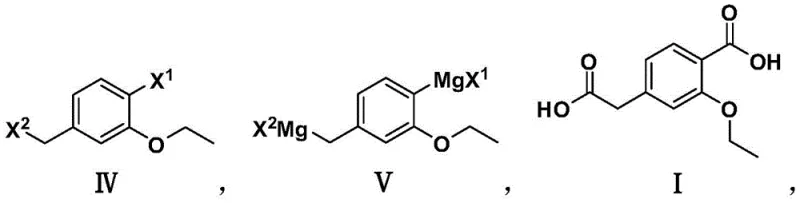
In stark contrast to the convoluted legacy methods, the novel approach detailed in CN108715576B utilizes a direct double carboxylation strategy that radically simplifies the molecular construction. By employing a dihalogenated compound (IV) as the sole organic starting material, the process bypasses the need for pre-functionalized nitriles or esters entirely. The core innovation lies in the generation of a double Grignard reagent (V), which serves as a potent nucleophile capable of attacking carbon dioxide at two distinct positions on the aromatic side chain and ring simultaneously. This telescoping of functionality introduction means that two carboxylic acid groups are installed in a single operational sequence following the Grignard formation. The use of carbon dioxide, an abundant and non-toxic C1 source, replaces expensive and hazardous cyanating agents, aligning the process with sustainable manufacturing principles. This strategic pivot not only reduces the number of unit operations but also significantly lowers the barrier to entry for scaling production, offering a clear pathway for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Double Grignard Carboxylation
The mechanistic elegance of this synthesis rests on the controlled formation and reactivity of the bis-organomagnesium species. The process initiates with the oxidative insertion of magnesium metal into the carbon-halogen bonds of the dihalide substrate, typically 4-bromomethyl-2-ethoxybromobenzene or its chloro-analog. To ensure complete conversion and prevent Wurtz-type coupling side reactions, the protocol specifies a substantial excess of magnesium, with a molar ratio ranging from 1:3 to 1:10 relative to the dihalide. The addition of catalytic iodine is critical here, as it activates the magnesium surface, facilitating the electron transfer required to generate the Grignard reagent in solvents like anhydrous tetrahydrofuran or diethyl ether. Once the double Grignard reagent (V) is formed, it presents two highly nucleophilic centers: one on the benzylic position and one on the aromatic ring ortho to the ethoxy group. When exposed to an excess of carbon dioxide, these nucleophiles attack the electrophilic carbon of the CO2 molecule, forming magnesium carboxylate salts at both sites. This dual carboxylation is the defining step that constructs the phenylacetic acid backbone and the benzoic acid moiety in a single pot.
Following the carboxylation event, the reaction mixture contains the magnesium salt of the target dicarboxylic acid. The final transformation involves a careful acidic quench, typically using 1M hydrochloric acid to adjust the pH to between 1 and 2. This protonation step liberates the free carboxylic acids from their magnesium salts, precipitating the product or allowing it to be extracted into an organic phase. The control of pH is vital; if the acidity is too low, the product may remain partially ionized and lost in the aqueous waste, while excessive acidity offers no benefit and increases corrosion risks. The resulting crude product is then purified via recrystallization from ethanol, which effectively removes residual magnesium salts and unreacted starting materials. This straightforward workup procedure ensures that the final API intermediate meets stringent purity specifications without requiring complex chromatographic separations, thereby enhancing the overall process efficiency and throughput.
How to Synthesize 3-Ethoxy-4-Carboxyphenylacetic Acid Efficiently
Implementing this synthesis requires precise control over reaction parameters to maximize yield and safety. The process begins under an inert atmosphere to protect the sensitive Grignard reagent from moisture and oxygen degradation. Magnesium powder is activated in dry solvent, followed by the slow addition of the dihalide solution to manage the exotherm. Once the Grignard formation is complete, indicated by the consumption of magnesium and TLC monitoring, the temperature is lowered to 0°C for the carboxylation step. Carbon dioxide is introduced either as dry ice or a gas stream, ensuring a molar excess of 2.5 to 12 times relative to the Grignard reagent to drive the equilibrium toward the dicarboxylate. After the reaction is quenched and the product isolated, standard analytical techniques confirm the structure and purity. For detailed operational parameters and specific stoichiometric ratios, please refer to the standardized synthesis guide below.
- Preparation of Double Grignard Reagent: React dihalide (IV) with magnesium metal (1:3 to 1:10 molar ratio) in anhydrous THF or diethyl ether with iodine initiation at 40-60°C.
- Carboxylation Reaction: Treat the double Grignard reagent (V) with excess carbon dioxide (1:2.5 to 1:12 molar ratio) using dry ice or dry CO2 gas at 0-10°C.
- Acidic Hydrolysis and Isolation: Quench the reaction mixture with 1M HCl to adjust pH to 1-2, separate organic phases, and recrystallize from ethanol to obtain the final acid (I).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this double Grignard technology represents a strategic opportunity to de-risk the supply of Repaglinide intermediates. The elimination of sodium cyanide from the supply chain removes a major regulatory and safety liability, simplifying vendor audits and reducing the costs associated with hazardous material handling and disposal. Furthermore, the reliance on commodity chemicals like magnesium, dihalides, and carbon dioxide insulates the production process from the volatility of specialized reagent markets. This robustness translates directly into enhanced supply chain reliability, as the raw materials are globally sourced and less prone to shortages compared to complex nitrile precursors. The simplified workflow also意味着 fewer processing steps, which inherently reduces the lead time for high-purity pharmaceutical intermediates and allows for faster response to market demand fluctuations.
- Cost Reduction in Manufacturing: The economic benefits of this route are driven primarily by the substitution of expensive and toxic reagents with cheap, abundant alternatives. By removing the need for multi-step precursor synthesis and eliminating cyanide waste treatment costs, the overall cost of goods sold (COGS) is significantly lowered. The high atom economy of using CO2 as a carbon source further contributes to financial efficiency, making this a superior choice for cost-sensitive generic drug production.
- Enhanced Supply Chain Reliability: The use of stable, commercially available dihalides ensures a consistent feedstock supply, mitigating the risk of production stoppages due to raw material scarcity. Unlike routes dependent on custom-synthesized nitriles, this method allows manufacturers to pivot quickly between suppliers of basic chemical commodities, ensuring continuous operation and on-time delivery for downstream API manufacturers.
- Scalability and Environmental Compliance: The process is inherently scalable, with reaction conditions (60°C reflux, 0°C carboxylation) that are easily managed in standard stainless steel reactors. The absence of heavy metals or persistent organic pollutants in the waste stream simplifies environmental compliance, facilitating easier permitting for capacity expansion and supporting long-term sustainability goals without compromising output volume.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and claims within CN108715576B, providing a factual basis for evaluating the technology's fit within your existing manufacturing portfolio. Understanding these nuances is essential for making informed decisions about process adoption and supplier qualification.
Q: What are the primary safety advantages of the double Grignard route over traditional nitrile hydrolysis?
A: The double Grignard route completely eliminates the use of sodium cyanide, a highly toxic substance required in traditional nitrile hydrolysis methods (such as CN 104628518A). This significantly reduces environmental hazards and operational risks, simplifying waste treatment and enhancing workplace safety compliance.
Q: Can this synthesis method be scaled for commercial production of Repaglinide intermediates?
A: Yes, the method utilizes commercially available dihalides and inexpensive carbon dioxide, making it highly suitable for industrial scale-up. The reaction conditions, such as refluxing at 60°C and controlled CO2 addition, are manageable in standard chemical reactors, ensuring robust supply chain continuity.
Q: What is the expected purity and yield profile for this synthetic pathway?
A: Patent examples demonstrate yields ranging from 61% to 65% with high purity achievable through simple ethanol recrystallization. The process avoids complex multi-step precursors, reducing the accumulation of difficult-to-remove impurities often associated with longer synthetic sequences.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Ethoxy-4-Carboxyphenylacetic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global pharmaceutical supply chain. Our technical team has extensively analyzed the double Grignard carboxylation pathway and possesses the expertise to implement this route with precision and safety. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements whether you are in the clinical trial phase or full-scale commercialization. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 3-ethoxy-4-carboxyphenylacetic acid meets the demanding standards required for Repaglinide synthesis.
We invite you to collaborate with us to leverage this advanced synthetic technology for your projects. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume needs. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of chemical excellence and commercial reliability.
