Advanced Synthesis of Umeclidinium Bromide Intermediate for High-Purity COPD Therapeutics
The global demand for Long-Acting Muscarinic Antagonists (LAMAs) in the treatment of Chronic Obstructive Pulmonary Disease (COPD) has necessitated the development of robust, scalable synthetic routes for key active pharmaceutical ingredients (APIs). A pivotal molecule in this therapeutic class is Umeclidinium Bromide, a potent anticholinergic agent. The commercial viability of this drug relies heavily on the efficiency of synthesizing its precursors, specifically the quaternary ammonium salt intermediate. Patent CN112645945A discloses a groundbreaking preparation method for the Wumei ammonium bromide intermediate (Intermediate III), addressing critical bottlenecks in yield and purity that have plagued previous manufacturing attempts. This technical insight report analyzes the novel two-step strategy that transforms a problematic one-pot reaction into a controlled, high-yield process suitable for global supply chains.
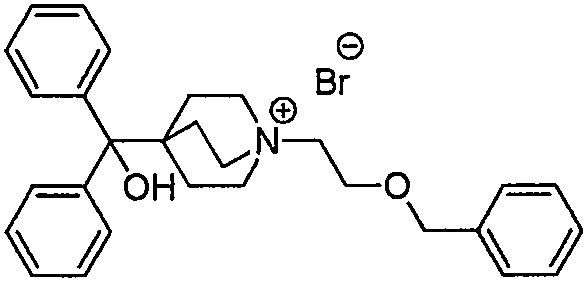
Traditional synthetic pathways for Umeclidinium Bromide intermediates have historically suffered from significant inefficiencies, primarily due to the reliance on telescoped one-pot reactions. As illustrated in the prior art disclosed in patents such as CN201380043392, the conventional approach attempts to perform cyclization and nucleophilic addition simultaneously or in rapid succession without isolating the bicyclic intermediate. This lack of process control leads to the accumulation of stubborn impurities, specifically compounds designated as Formula IV and Formula V, which are structurally similar to the target molecule and notoriously difficult to remove via standard crystallization or chromatography. Furthermore, the intermediate formed after cyclization (Formula II) typically exists as an unstable oil in these legacy methods, making it impossible to purify effectively before proceeding to the next step, thereby carrying forward contaminants that degrade the final API quality.
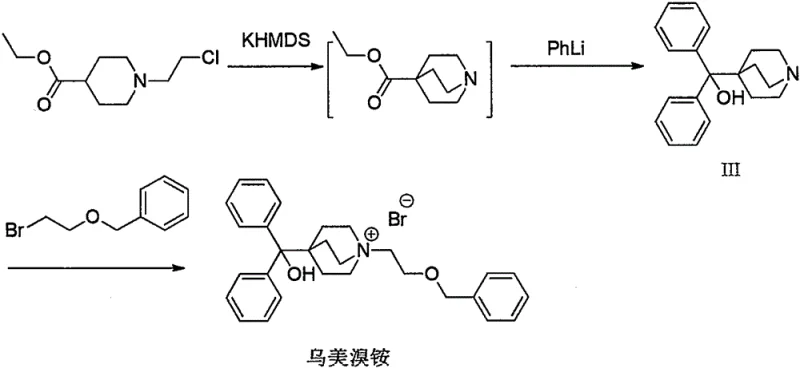
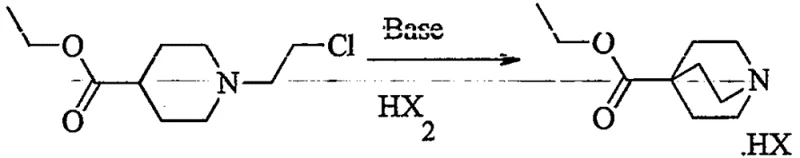
In stark contrast to the limitations of conventional methods, the novel approach detailed in CN112645945A introduces a strategic interruption in the synthesis sequence to isolate and purify the bicyclic core. The innovation lies in Step (a), where the starting material, ethyl 1-(2-chloroethyl)-4-piperidinecarboxylate (Formula I), undergoes cyclization under alkaline conditions using bases such as potassium hexamethyldisilazide (KHMDS) or lithium diisopropylamide (LDA). Crucially, instead of proceeding directly to addition, the reaction mixture is treated with hydrochloric acid to form a hydrochloride salt (Formula II-1). This salt formation converts the previously oily intermediate into a stable, crystalline solid that can be filtered, washed, and dried. This physical state change is the cornerstone of the new process, enabling the removal of unreacted starting materials and side products before the expensive phenyllithium addition step is even initiated.
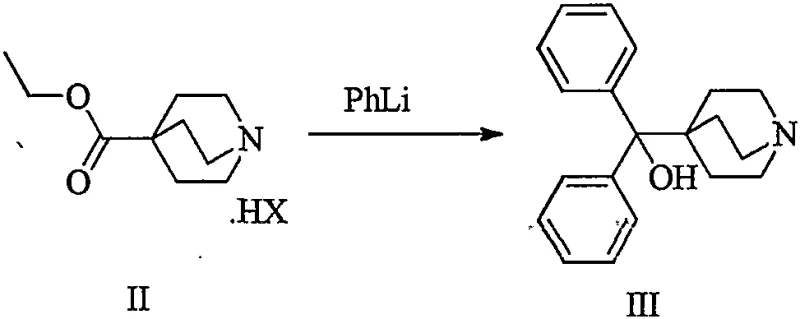
The mechanistic insights into this improved synthesis reveal a sophisticated understanding of impurity control and reaction kinetics. By isolating Intermediate II-1 as a crystalline hydrochloride salt, the process effectively resets the purity profile of the reaction stream. The patent provides detailed X-ray powder diffraction (XRPD) data for Formula II-1, confirming its crystalline nature with characteristic peaks at 2θ angles such as 14.41°, 19.43°, and 25.83°. This structural definition ensures batch-to-batch consistency, a critical parameter for regulatory compliance. In the subsequent Step (b), the purified salt is neutralized to its free base form and subjected to nucleophilic addition with phenyllithium (PhLi). Because the starting material for this step is now of high purity, the formation of the diphenyl hydroxymethyl group proceeds with minimal side reactions. The result is Intermediate III (1-azabicyclo[2.2.2]oct-4-yl(diphenyl)methanol) with an exceptional HPLC purity of 99.5% and single impurity levels reduced to below 0.1%, a significant improvement over the >1.0% impurity levels seen in comparative examples.
How to Synthesize Umeclidinium Bromide Intermediate III Efficiently
The synthesis of this critical pharmaceutical intermediate requires precise control over reaction conditions to maximize the benefits of the new isolation strategy. The process begins with the cyclization of the piperidine derivative in a solvent like toluene at low temperatures (-20°C), followed by careful salification to precipitate the crystalline hydrochloride salt. Once isolated, this solid intermediate serves as a reliable feedstock for the subsequent Grignard-type addition. The detailed standardized synthesis steps, including specific molar equivalents, temperature ramps, and workup procedures required to achieve the reported 99.5% purity, are outlined in the guide below.
- Cyclize ethyl 1-(2-chloroethyl)-4-piperidinecarboxylate using a strong base like KHMDS or LDA, followed by salification with hydrochloric acid to isolate crystalline ethyl 1-azabicyclo[2.2.2]oct-4-carboxylate hydrochloride (II-1).
- Neutralize the isolated salt II-1 to its free base form in an organic solvent such as toluene.
- Perform an addition reaction with phenyllithium (PhLi) at controlled low temperatures (-20°C to 25°C) to generate the final intermediate III with high purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from a one-pot oily process to a stepwise crystalline process offers profound logistical and economic benefits. The ability to isolate Intermediate II-1 as a stable solid means that the production of the bicyclic core can be decoupled from the final addition step. This modularity allows manufacturers to stockpile the purified intermediate, mitigating risks associated with supply chain disruptions or batch failures in the final step. Furthermore, the elimination of difficult purification stages for the final product reduces the consumption of solvents and chromatography media, leading to substantial cost savings in raw material usage and waste disposal. The robustness of the crystallization process also ensures that the manufacturing timeline is more predictable, reducing the lead time for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The new route eliminates the need for complex and costly purification techniques often required to remove oily impurities from the final API precursor. By achieving high purity (99.5%) through simple crystallization and filtration of the intermediate salt, the process significantly lowers the operational expenditure related to solvent recovery and waste treatment. The higher overall yield of approximately 75% for the two steps, compared to the significantly lower yields of the prior art, directly translates to a reduced cost of goods sold (COGS) per kilogram of active ingredient.
- Enhanced Supply Chain Reliability: The conversion of the intermediate into a stable crystalline solid (II-1) fundamentally changes the inventory management strategy for this chemical. Unlike oils which may degrade or vary in quality over time, the crystalline salt can be stored and transported with greater stability, ensuring a consistent supply of high-quality starting material for the final quaternization step. This stability reduces the risk of batch rejection and ensures that the reliable pharmaceutical intermediates supplier can meet delivery schedules with greater confidence.
- Scalability and Environmental Compliance: The process is explicitly designed for industrialization, utilizing common reagents like KHMDS and PhLi in standard solvents such as toluene and ethanol. The simplicity of the unit operations—primarily stirring, cooling, filtration, and drying—facilitates easy scale-up from pilot plants to multi-ton commercial production without the need for specialized equipment. Additionally, the reduction in impurity load minimizes the environmental footprint of the manufacturing process by decreasing the volume of hazardous waste generated during purification.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Umeclidinium Bromide intermediates. These answers are derived directly from the experimental data and comparative analysis provided in the patent literature, offering clarity on how this new method resolves historical manufacturing challenges.
Q: How does the new method improve the purity of Intermediate III compared to prior art?
A: The new method isolates Intermediate II as a crystalline hydrochloride salt (II-1) before the addition step. This purification step removes impurities early, preventing the formation of byproducts IV and V, resulting in Intermediate III with >99.5% purity and single impurities <0.1%.
Q: What are the key advantages of isolating Intermediate II-1 as a solid?
A: In prior art, Intermediate II was an oil that was difficult to purify. The new method produces II-1 as a stable white crystalline powder (confirmed by XRPD), which allows for easy filtration, washing, and storage, significantly enhancing process robustness and supply chain reliability.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the patent explicitly states the process is suitable for industrialization. It utilizes commercially available reagents, avoids complex one-pot mixtures that are hard to control, and achieves a combined yield of approximately 75% with simple operational steps like crystallization and filtration.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Umeclidinium Bromide Intermediate Supplier
As the pharmaceutical industry continues to demand higher standards of purity and consistency for respiratory therapies, partnering with a manufacturer that understands the nuances of complex intermediate synthesis is essential. NINGBO INNO PHARMCHEM leverages extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to deliver intermediates that meet stringent purity specifications. Our rigorous QC labs ensure that every batch of Umeclidinium Bromide intermediate adheres to the highest quality standards, utilizing advanced analytical techniques to verify the absence of critical impurities like Formula IV and V.
We invite global partners to collaborate with us to optimize their supply chains for COPD medications. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production volumes. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced synthesis capabilities can support your long-term commercial goals.
