Advanced Selective Acylation Process for High-Purity Pyripyropene Agrochemical Intermediates
Advanced Selective Acylation Process for High-Purity Pyripyropene Agrochemical Intermediates
The development of efficient synthetic routes for bioactive natural product derivatives remains a critical frontier in modern agrochemical research. Patent CN102348706A introduces a groundbreaking methodology for the production of pyripyropene derivatives, specifically targeting compounds with acyloxy groups at the 1 and 11 positions and a hydroxyl group at the 7 position. These structural motifs are essential for conferring potent insecticidal and therapeutic properties, making them highly valuable assets for pest control strategies. The disclosed process represents a significant departure from traditional multi-step syntheses, offering a streamlined approach that enhances both yield and operational simplicity. By leveraging selective acylation techniques on tri-deacetyl precursors, this technology addresses long-standing challenges in regioselectivity and purification that have historically hindered the scalable manufacturing of these complex molecules.
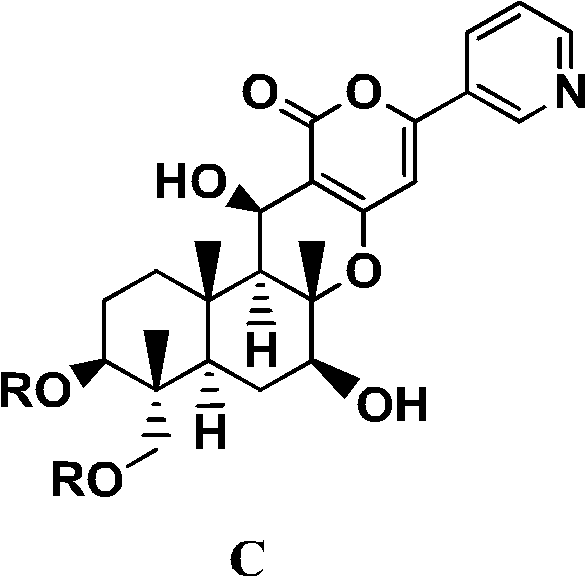
For procurement managers and supply chain directors seeking a reliable agrochemical intermediate supplier, the implications of this patent are profound. The ability to produce high-purity pyripyropene derivatives through a reduced number of steps translates directly into cost reduction in agrochemical manufacturing. Traditional methods often rely on non-selective hydrolysis of fully acylated starting materials, which necessitates cumbersome separation processes to isolate the desired isomer. In contrast, the novel approach described herein allows for the direct construction of the target molecular architecture from readily available tri-deacetyl compounds. This shift not only minimizes waste generation but also stabilizes the supply chain by reducing dependency on complex purification protocols that are prone to variability at scale.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical methods for synthesizing 1,11-diacyl-7-hydroxy pyripyropene derivatives, as referenced in prior art such as WO 2006/129714, typically involve the non-selective hydrolysis of 1,7,11-triacyl compounds. This conventional pathway is inherently inefficient because it generates a mixture of products where the acyl groups are randomly removed, requiring extensive chromatographic separation to isolate the specific 7-hydroxy isomer. Furthermore, alternative strategies involving the use of protecting groups to mask the 7-position hydroxyl during acylation add significant complexity, increasing both the time and material costs associated with production. These legacy processes often suffer from low overall yields due to cumulative losses across multiple protection, reaction, and deprotection stages, making them economically unviable for large-scale commercial operations.
The Novel Approach
The innovative strategy outlined in the patent data circumvents these inefficiencies by utilizing a tri-deacetyl compound (Formula B1) as the direct starting material for selective acylation. Instead of removing groups from a fully substituted molecule, this method builds the desired functionality directly onto the scaffold. As illustrated in the reaction scheme below, the process enables the introduction of acyl groups specifically at the 1 and 11 positions while leaving the 7-position hydroxyl intact. This regioselective transformation is achieved through precise control of reaction conditions, including the choice of base and solvent, eliminating the need for temporary protecting groups. The result is a drastically simplified workflow that improves atom economy and reduces the environmental footprint of the synthesis.
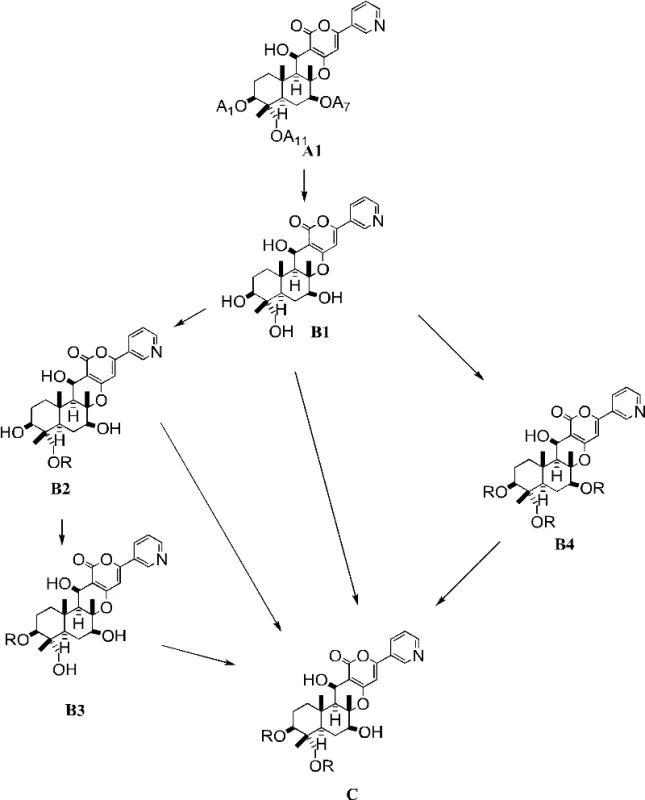
Mechanistic Insights into Regioselective Acylation
The success of this synthetic route hinges on the subtle electronic and steric differences between the hydroxyl groups at the 1, 7, and 11 positions of the pyripyropene scaffold. The patent data reveals that the hydroxyl groups at positions 1 and 11 exhibit higher nucleophilicity or accessibility under specific basic conditions compared to the 7-position hydroxyl. By employing organic bases such as 2,6-lutidine or 1,8-diazabicyclo[5.4.0]undec-7-ene (DBU), the reaction environment can be tuned to favor acylation at the desired sites. For instance, the use of 2,6-lutidine in polar aprotic solvents like N-methyl-2-pyrrolidone (NMP) facilitates the formation of the 1,11-diacyl product with high selectivity. This mechanistic understanding allows chemists to fine-tune the stoichiometry of the acylating agent, typically using between 2.0 to 5.0 equivalents, to drive the reaction to completion without over-acylating the 7-position.
Impurity control is another critical aspect addressed by this mechanism. In traditional hydrolysis routes, partially hydrolyzed byproducts are difficult to separate due to their similar polarity. However, in this direct acylation method, the primary impurities are either the unreacted starting material or the fully acylated tri-ester, both of which possess distinct physical properties that facilitate removal. The patent describes a robust crystallization protocol where the target Compound C forms stable solvate crystals, particularly with ethyl acetate. This phase behavior difference allows for the effective exclusion of impurities during the solidification process. By optimizing parameters such as temperature gradients and the addition of anti-solvents like hexane, manufacturers can achieve purity levels exceeding 90%, ensuring the material meets the stringent specifications required for downstream formulation.
How to Synthesize 1,11-Diacyl-7-hydroxy Pyripyropene Derivatives Efficiently
The synthesis of these valuable intermediates begins with the preparation of the tri-deacetyl precursor, Compound B1, which can be derived from natural Pyripyropene A or synthesized analogues. Once obtained, the core transformation involves suspending Compound B1 in a suitable solvent such as NMP and adding a controlled amount of acylating agent, such as cyclopropanecarbonyl chloride, in the presence of a base. The reaction is typically conducted at temperatures ranging from -20°C to 50°C, with reaction times varying from a few hours to several days depending on the specific reagents used. Following the reaction, the mixture is quenched, and the product is isolated through a specialized crystallization process that leverages the unique solubility profile of the target molecule.
- Prepare the tri-deacetyl precursor (Compound B1) via hydrolysis of naturally occurring Pyripyropene A or its analogues.
- Perform selective acylation at the 1-position and 11-position using an acylating agent (e.g., cyclopropanecarbonyl chloride) in the presence of a base like 2,6-lutidine or DBU.
- Purify the resulting Compound C through crystallization from organic solvents such as ethyl acetate, optionally adding a poor solvent like hexane to induce precipitation.
Commercial Advantages for Procurement and Supply Chain Teams
For organizations focused on cost reduction in agrochemical manufacturing, this patented process offers a compelling value proposition by fundamentally altering the economics of production. The elimination of protection and deprotection steps removes entire categories of reagents and unit operations from the supply chain, leading to substantial cost savings. Moreover, the use of commodity chemicals like cyclopropanecarbonyl chloride and common organic bases ensures that raw material sourcing remains stable and predictable, mitigating the risks associated with specialty reagent shortages. The simplified workflow also reduces the demand for highly skilled labor and complex equipment, further driving down the operational expenditure required to bring these intermediates to market.
- Cost Reduction in Manufacturing: The direct acylation strategy significantly lowers production costs by reducing the total number of synthetic steps. By avoiding the use of expensive protecting groups and the associated reagents required for their installation and removal, the process achieves a leaner material balance. Additionally, the high selectivity of the reaction minimizes the formation of hard-to-separate byproducts, which reduces the load on purification systems and decreases solvent consumption. This efficiency translates into a lower cost of goods sold (COGS), providing a competitive edge in the global agrochemical marketplace.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials and standard reagents enhances the resilience of the supply chain. Unlike processes that depend on exotic catalysts or unstable intermediates, this method utilizes robust chemistry that can be sourced from multiple vendors globally. The ability to produce the tri-deacetyl precursor from natural sources or established synthetic routes further diversifies the supply base. This redundancy ensures continuity of supply even in the face of market fluctuations, allowing procurement teams to negotiate better terms and secure long-term contracts with confidence.
- Scalability and Environmental Compliance: From an environmental perspective, the process aligns well with green chemistry principles by minimizing waste generation and solvent usage. The crystallization-based purification method is inherently more scalable than chromatographic techniques, which are often difficult to translate from laboratory to industrial scale. Furthermore, the reduced number of steps leads to a lower overall energy consumption and a smaller carbon footprint. This sustainability profile is increasingly important for meeting regulatory requirements and corporate social responsibility goals, making the technology attractive for environmentally conscious manufacturers.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthetic route. Understanding these details is crucial for R&D teams evaluating the feasibility of adopting this technology for their own production lines. The answers are derived directly from the experimental data and embodiments provided in the patent documentation, ensuring accuracy and relevance for practical application.
Q: What is the key advantage of the selective acylation method described in CN102348706A?
A: The primary advantage is the ability to directly introduce acyl groups at the 1 and 11 positions of the tri-deacetyl compound (B1) without needing complex protection and deprotection steps for the 7-position hydroxyl group, significantly simplifying the synthetic route.
Q: Which bases are preferred for ensuring regioselectivity in this synthesis?
A: The patent highlights organic bases such as 2,6-lutidine, 2,4,6-trimethylpyridine, and 1,8-diazabicyclo[5.4.0]undec-7-ene (DBU) as highly effective for controlling the acylation site and minimizing side reactions.
Q: How is the final product purified to meet high-purity standards?
A: The process utilizes a robust crystallization technique where the crude product is dissolved in ethyl acetate and treated with a poor solvent like hexane or water, allowing for the isolation of high-purity solvate crystals suitable for commercial application.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyripyropene Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the selective acylation technology described in CN102348706A for the agrochemical sector. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent quality regardless of volume. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of pyripyropene derivatives meets the highest industry standards. We are committed to bridging the gap between innovative patent chemistry and commercial reality, delivering solutions that drive value for our partners.
We invite you to engage with our technical procurement team to discuss how we can tailor this advanced synthesis to your specific needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this streamlined process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that will enhance your supply chain efficiency and product competitiveness in the global market.
