Optimizing Bisacodyl Production: A Technical Analysis of the Improved Acid-Catalyzed Route
Introduction to Advanced Bisacodyl Manufacturing
The pharmaceutical industry constantly seeks robust, scalable, and environmentally conscious pathways for producing critical active pharmaceutical ingredients (APIs) and their intermediates. A significant breakthrough in this domain is detailed in patent CN101973932A, which outlines an improved preparation method for Bisacodyl, a widely used stimulant laxative. This patent addresses long-standing inefficiencies in traditional synthetic routes, specifically targeting the low yields and complex purification steps associated with the formation of the key intermediate, 4',4''-dihydroxydiphenyl-(2-pyridine)-methane. By shifting from cumbersome column chromatography or inefficient recrystallization techniques to a sophisticated pH-controlled selective precipitation strategy, this technology offers a compelling value proposition for manufacturers aiming to optimize their supply chains. The method leverages common industrial reagents like sulfuric acid and ethyl acetate to achieve a product purity of 99% and an overall yield that significantly outperforms historical benchmarks. For R&D directors and procurement specialists, understanding the nuances of this route is essential for evaluating potential partnerships with reliable API intermediate suppliers who can deliver high-quality materials at competitive price points.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
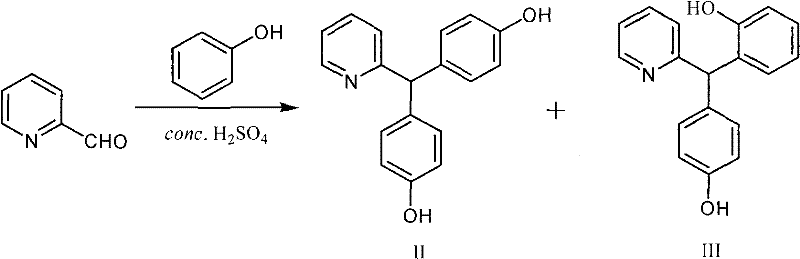
Historically, the synthesis of Bisacodyl has been plagued by technical hurdles that inflate production costs and complicate scale-up operations. Traditional routes often involve the condensation of 2-pyridylaldehyde with phenol, which inevitably produces a mixture of the desired para-isomer and the unwanted ortho-isomer (2',4'-dihydroxydiphenyl-(2-pyridine)-methane). Separating these isomers has traditionally been a bottleneck; methods such as those reported by Mereyala required Fries rearrangement and subsequent column chromatography, a technique that is notoriously difficult to implement on a multi-ton commercial scale due to solvent intensity and low throughput. Other conventional approaches attempted to bypass separation by directly acetylating the crude mixture, but this strategy resulted in excessive consumption of acetic anhydride, as the reagent was wasted on acetylating the ortho-isomer impurities. Furthermore, older recrystallization techniques using ethanol and water often yielded poor recovery rates for the key intermediate, sometimes as low as 26%, leading to substantial material loss and increased waste disposal burdens. These inefficiencies not only drive up the cost of goods sold (COGS) but also introduce variability in the final API quality, posing risks for regulatory compliance.
The Novel Approach
The methodology described in CN101973932A introduces a paradigm shift by integrating a highly effective purification step prior to the final acetylation. Instead of struggling to separate isomers via chromatography or wasting reagents on mixed streams, this novel approach utilizes the distinct solubility profiles of the isomers in ethyl acetate at a neutral pH. After the initial sulfuric acid-catalyzed condensation, the reaction mixture is carefully neutralized to pH 7.0 using sodium hydroxide. At this specific pH and in the presence of ethyl acetate, the desired 4',4''-dihydroxydiphenyl-(2-pyridine)-methane precipitates out as a solid, while the ortho-isomer and excess phenol remain dissolved in the organic phase. This elegant physical separation allows for the isolation of the intermediate with 98% content and a yield of 68%, a dramatic improvement over conventional methods. Following this purification, the acetylation step proceeds with high efficiency, utilizing DMAP as a catalyst to ensure complete conversion without mono-acetylated byproducts. This streamlined process not only simplifies operations but also drastically reduces the environmental footprint by enabling the recovery and reuse of phenol from the filtrate.
Mechanistic Insights into Acid-Catalyzed Condensation and Selective Precipitation
The core of this synthetic innovation lies in the precise control of reaction conditions during the electrophilic aromatic substitution and the subsequent workup. The condensation reaction between 2-pyridylaldehyde and phenol is catalyzed by concentrated sulfuric acid at low temperatures (0-15°C). Under these acidic conditions, the aldehyde is activated to form an electrophilic species that attacks the electron-rich phenol ring, predominantly at the para-position due to steric and electronic factors, though the ortho-isomer is inevitably formed as a byproduct. The critical mechanistic advancement, however, occurs during the workup phase. By adjusting the pH to exactly 7.0, the phenolic hydroxyl groups are deprotonated just enough to alter their solubility characteristics without forming stable salts that would remain in the aqueous layer. The addition of ethyl acetate creates a biphasic system where the thermodynamic stability of the para-isomer crystal lattice drives its precipitation. This phenomenon exploits the subtle differences in molecular symmetry and hydrogen bonding capabilities between the 4',4''-isomer and the 2',4''-isomer. The para-isomer, being more symmetrical, packs more efficiently in the solid state, making it less soluble in the ethyl acetate/phenol mixture compared to the asymmetrical ortho-isomer. This physical chemistry principle is the cornerstone of the process's success, allowing for high-purity isolation without the need for energy-intensive distillation or chromatographic columns.
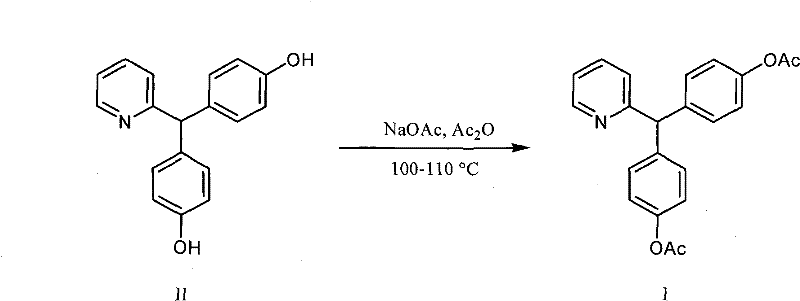
Following the isolation of the pure intermediate, the acetylation mechanism proceeds via nucleophilic acyl substitution. The use of 4-Dimethylaminopyridine (DMAP) is crucial here; DMAP acts as a potent nucleophilic catalyst, forming a highly reactive acetylpyridinium ion intermediate with acetic anhydride. This activated species is far more electrophilic than acetic anhydride alone, allowing it to rapidly acetylate the phenolic hydroxyl groups of the intermediate even at room temperature. This mild condition prevents the degradation of the sensitive pyridine ring or the methane bridge, which could occur under harsher acidic or thermal conditions. The result is a clean conversion to the diacetate ester (Bisacodyl) with minimal formation of mono-acetylated impurities, ensuring that the final recrystallization from ethanol yields a product with 99% purity. This mechanistic understanding assures R&D teams that the process is robust and controllable, minimizing the risk of batch-to-batch variability.
How to Synthesize Bisacodyl Efficiently
Implementing this synthesis route requires strict adherence to the temperature and pH parameters defined in the patent to maximize the selectivity of the precipitation step. The process begins with the controlled addition of 2-pyridylaldehyde to a phenol-sulfuric acid mixture, maintaining the temperature below 15°C to suppress side reactions. The subsequent neutralization and ethyl acetate treatment must be performed with precise stoichiometry to ensure the ortho-isomer remains in solution while the target compound crystallizes. Once the high-purity intermediate is secured, the acetylation is straightforward, requiring only standard mixing and ambient temperature conditions. For a detailed breakdown of the specific reagent ratios, stirring times, and filtration protocols necessary to replicate this high-yield process, please refer to the standardized synthesis guide below.
- Conduct condensation of 2-pyridylaldehyde and phenol using sulfuric acid catalyst at 0-15°C to form the crude methane intermediate.
- Adjust pH to 7.0 with sodium hydroxide and utilize ethyl acetate extraction to selectively precipitate the pure 4',4''-dihydroxydiphenyl-(2-pyridine)-methane, removing ortho-isomers.
- Perform acetylation on the purified intermediate using acetic anhydride and DMAP catalyst in dichloromethane, followed by ethanol recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route translates into tangible operational efficiencies and cost reductions. The primary economic driver is the significant improvement in material utilization. By recovering phenol from the filtrate at rates reaching 85-90%, the process transforms what was once a waste stream into a valuable resource, effectively lowering the net consumption of raw materials. This closed-loop approach not only reduces the direct cost of goods but also mitigates the volatility associated with raw material pricing fluctuations. Furthermore, the elimination of column chromatography and the reduction in acetic anhydride usage simplify the supply chain requirements, reducing the number of specialized reagents that need to be sourced and managed. The simplicity of the unit operations—primarily stirring, filtration, and crystallization—means that the process can be easily scaled from pilot plant to commercial production without requiring exotic equipment or extensive re-engineering.
- Cost Reduction in Manufacturing: The economic benefits of this route are driven by qualitative improvements in process efficiency rather than just yield numbers. By removing the ortho-isomer before the acetylation step, the process eliminates the wasteful consumption of acetic anhydride on unwanted byproducts, leading to substantial savings in reagent costs. Additionally, the ability to recover and reuse phenol significantly lowers the effective cost of the starting materials. The simplified workflow, which avoids complex separation techniques like column chromatography, reduces labor hours and solvent consumption, further driving down the overall manufacturing overhead. These factors combine to create a leaner production model that is highly resilient to market pressures.
- Enhanced Supply Chain Reliability: From a supply chain perspective, the reliance on commodity chemicals such as phenol, sulfuric acid, and ethyl acetate ensures a stable and secure supply of raw materials. Unlike routes that depend on specialized or scarce catalysts, this method utilizes reagents that are readily available from multiple global suppliers, reducing the risk of supply disruptions. The robustness of the synthesis, characterized by high yields and simple purification steps, also enhances production predictability. Manufacturers can confidently plan production schedules knowing that the process is less prone to failures or delays caused by difficult separations, thereby ensuring consistent on-time delivery for downstream API manufacturers.
- Scalability and Environmental Compliance: The environmental profile of this method aligns well with modern green chemistry principles and regulatory expectations. The reduction in solvent usage, particularly the avoidance of large volumes of eluents required for chromatography, minimizes the generation of hazardous waste. The recovery of phenol not only saves money but also alleviates the environmental burden of treating phenol-contaminated wastewater. The process operates at mild temperatures and uses standard reactors, making it inherently scalable from kilogram to multi-ton quantities without encountering the heat transfer or mixing limitations often seen in more complex chemistries. This scalability ensures that suppliers can meet surging demand without compromising on quality or environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Bisacodyl synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent literature, offering clarity on yield improvements, impurity control, and resource recovery. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this method for commercial production.
Q: How does the new method improve yield compared to conventional ethanol recrystallization?
A: The patented method utilizes ethyl acetate extraction at neutral pH to exploit solubility differences, increasing the yield of the key intermediate from roughly 26-43% in conventional methods to 68%.
Q: What is the advantage of removing the ortho-isomer before acetylation?
A: Removing the ortho-isomer (2',4'-dihydroxydiphenyl-(2-pyridine)-methane) prior to acetylation significantly reduces the consumption of acetic anhydride, as it prevents the wasteful acetylation of unwanted byproducts.
Q: Can phenol be recovered in this process to reduce costs?
A: Yes, the process allows for the recovery of phenol from the filtrate with a recovery rate of 85-90%, which economizes resources and lowers overall production costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bisacodyl Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical advantages of the CN101973932A route are fully realized in practice. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of Bisacodyl meets the highest international standards. Our commitment to quality assurance means that we can consistently deliver high-purity intermediates that facilitate smooth downstream API synthesis for our global partners.
We invite pharmaceutical and chemical companies to collaborate with us to leverage these advanced manufacturing technologies. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and supply chain needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you optimize your Bisacodyl supply chain with a solution that balances cost, quality, and reliability.
