Scalable Synthesis of 3-Hydroxy-2,3-Dihydrobenzofuran Derivatives for Advanced Pharmaceutical Intermediates
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and economically viable pathways to access complex heterocyclic scaffolds that serve as critical building blocks for drug discovery. Patent CN102267964B introduces a transformative methodology for the synthesis of 3-hydroxy-2,3-dihydrobenzofuran derivatives, a class of compounds renowned for their potent biological activities ranging from antifungal to central nervous system (CNS) therapeutic applications. This innovation addresses the longstanding challenges associated with traditional synthetic routes by employing a simple, base-catalyzed condensation between substituted salicylaldehydes and 1,3-dicarbonyl compounds. Unlike legacy methods that rely on precious metal catalysts and hazardous solvents, this novel approach utilizes inexpensive inorganic bases and environmentally benign reaction media, positioning it as a highly attractive option for the commercial scale-up of complex pharmaceutical intermediates. The versatility of this chemistry allows for the introduction of diverse functional groups, enabling medicinal chemists to rapidly explore structure-activity relationships while maintaining a favorable cost profile for large-scale production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the dihydrobenzofuran core has often relied on transition metal-catalyzed cyclizations, which present significant hurdles for industrial adoption. As illustrated in prior art references within the patent landscape, methods utilizing palladium catalysts such as Pd(OAc)2 in conjunction with oxidants or specialized ligands are commonplace but problematic.
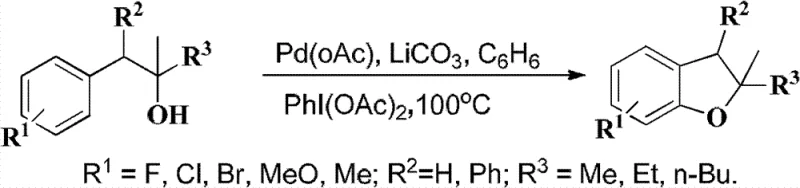
These conventional protocols typically require expensive noble metals, which not only inflate the raw material costs but also introduce stringent purification requirements to meet residual metal specifications for high-purity pharmaceutical intermediates. Furthermore, many of these established routes necessitate the use of toxic or difficult-to-remove solvents like hexafluorobenzene or dimethylformamide (DMF), creating substantial environmental liabilities and increasing waste treatment expenses. The multi-step nature of some traditional syntheses also leads to cumulative yield losses, making the final product economically unfeasible for cost-sensitive applications such as agrochemicals or generic drug manufacturing.
The Novel Approach
In stark contrast, the methodology disclosed in CN102267964B offers a streamlined, one-pot solution that bypasses the need for transition metals entirely. By leveraging the inherent nucleophilicity of 1,3-dicarbonyl compounds activated by simple bases like potassium carbonate or sodium acetate, the reaction proceeds efficiently under mild conditions. This shift from noble metal catalysis to organocatalytic or base-mediated processes represents a paradigm shift in cost reduction in API manufacturing. The ability to conduct the reaction in common solvents like acetone and ethanol, or even under solvent-free mechanochemical conditions, drastically simplifies the downstream processing. This operational simplicity translates directly into reduced cycle times and lower energy consumption, providing a compelling value proposition for reliable pharmaceutical intermediate suppliers aiming to optimize their production portfolios.
Mechanistic Insights into Base-Catalyzed Cyclization
The success of this synthetic strategy lies in the elegant mechanistic pathway that facilitates the rapid assembly of the benzofuran ring system. The reaction initiates with the deprotonation of the active methylene group in the 1,3-dicarbonyl compound by the basic catalyst, generating a stabilized enolate nucleophile. This species then undergoes a Michael-type addition to the carbonyl carbon of the salicylaldehyde, forming a key carbon-carbon bond. Subsequently, the phenolic hydroxyl group, now positioned in proximity to the electrophilic center, performs an intramolecular nucleophilic substitution, displacing the leaving group (typically a halide or nitro group from the 1,3-dicarbonyl precursor) to close the five-membered furan ring.
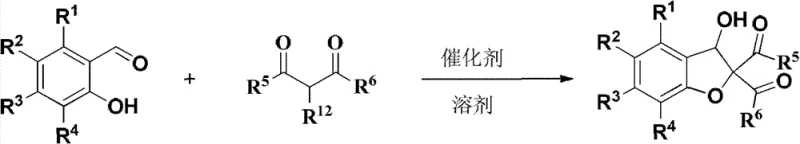
This cascade sequence is highly atom-economical and avoids the formation of complex byproducts often seen in radical-based cyclizations. From a quality control perspective, the mechanism ensures a clean impurity profile, as the primary side reactions are limited to simple hydrolysis or polymerization which are easily managed through pH control. The tolerance of the reaction to various substituents on the aromatic ring (R1-R4) and the carbonyl moiety (R5-R6) demonstrates the robustness of the catalytic cycle. For R&D directors, understanding this mechanism is crucial for troubleshooting and optimizing reaction parameters, ensuring that the commercial scale-up of complex heterocycles proceeds with consistent reproducibility and high purity standards required for regulatory filings.
How to Synthesize 3-Hydroxy-2,3-Dihydrobenzofuran Derivatives Efficiently
The practical execution of this synthesis is designed for flexibility, accommodating both laboratory-scale optimization and pilot-plant production. The general protocol involves combining the salicylaldehyde and the 1,3-dicarbonyl substrate in a reaction vessel, followed by the addition of the base catalyst and solvent. The mixture is then agitated at controlled temperatures, typically ranging from ambient to moderately elevated levels, until conversion is complete. Workup procedures are straightforward, usually involving aqueous quenching, organic extraction, and final purification via crystallization or chromatography depending on the desired purity grade. Detailed standardized operating procedures for specific derivatives can be found below.
- Mix substituted or unsubstituted salicylaldehyde with a 1,3-dicarbonyl compound in a suitable solvent such as acetone or ethanol, or under solvent-free grinding conditions.
- Add a basic catalyst such as potassium carbonate, sodium acetate, or cesium carbonate to the reaction mixture to initiate the Michael addition and subsequent cyclization.
- Stir the reaction at temperatures ranging from -10°C to 100°C for 10 minutes to 96 hours, followed by extraction, drying, and purification via recrystallization or column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this base-catalyzed technology offers tangible strategic benefits beyond mere chemical novelty. The elimination of palladium and other precious metals removes a significant source of price volatility and supply risk from the raw material basket. Since the catalysts employed are commodity chemicals like potassium carbonate, the supply chain becomes inherently more stable and less susceptible to geopolitical disruptions affecting rare earth or noble metal markets. Furthermore, the use of green solvents aligns with increasingly strict environmental regulations, reducing the compliance burden and potential fines associated with hazardous waste disposal.
- Cost Reduction in Manufacturing: The removal of expensive transition metal catalysts and the ability to use cheaper, recyclable solvents significantly lowers the direct material costs per kilogram of product. Additionally, the simplified workup reduces the consumption of silica gel and eluents typically required for removing metal residues, leading to substantial operational savings. The high yields reported in the patent data suggest that less starting material is wasted, further enhancing the overall process economics and improving the margin profile for bulk manufacturing.
- Enhanced Supply Chain Reliability: The starting materials, specifically substituted salicylaldehydes and beta-keto esters, are widely available from multiple global vendors, preventing single-source bottlenecks. The robustness of the reaction conditions means that production can be maintained even if specific high-grade reagents are temporarily unavailable, as the process tolerates a range of commercial-grade inputs. This flexibility ensures reducing lead time for high-purity pharmaceutical intermediates and guarantees continuity of supply for downstream drug manufacturers who rely on just-in-time delivery models.
- Scalability and Environmental Compliance: The solvent-free variant of this process offers a unique advantage for facilities looking to minimize their environmental footprint. By eliminating solvent usage entirely in certain embodiments, the plant reduces its volatile organic compound (VOC) emissions and fire safety risks. The scalability is proven by the linear relationship between reactant ratios and yield, allowing for seamless transfer from gram-scale R&D to tonnage-scale commercial production without the need for complex engineering modifications or specialized high-pressure equipment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing clarity on the feasibility and scope of the method for potential licensees or manufacturing partners.
Q: What are the primary advantages of this base-catalyzed method over traditional palladium-catalyzed routes?
A: The base-catalyzed method eliminates the need for expensive and toxic transition metal catalysts like palladium, significantly reducing raw material costs and simplifying the removal of heavy metal impurities, which is critical for pharmaceutical compliance.
Q: Can this synthesis be performed without organic solvents?
A: Yes, the patent explicitly describes a solvent-free variation where reactants are ground together, offering an environmentally friendly alternative that reduces waste disposal costs and improves the overall green chemistry profile of the manufacturing process.
Q: What is the biological activity profile of the resulting derivatives?
A: The synthesized 3-hydroxy-2,3-dihydrobenzofuran derivatives exhibit broad-spectrum antibacterial activity, showing particular efficacy against Candida albicans and Staphylococcus aureus, making them valuable candidates for developing new antimicrobial therapies.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Hydroxy-2,3-Dihydrobenzofuran Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient synthetic routes in the development of next-generation therapeutics. Our team of expert process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to market availability is seamless. We are committed to delivering stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 3-hydroxy-2,3-dihydrobenzofuran derivatives meets the exacting standards required for clinical and commercial applications. Our state-of-the-art facilities are equipped to handle the specific solvent and safety requirements of this base-catalyzed chemistry, providing a secure and compliant manufacturing environment.
We invite you to collaborate with us to leverage this innovative technology for your specific drug development programs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can accelerate your project timelines and optimize your budget.
