Advanced Synthesis of Pyrazole Spiro Derivatives for Commercial Scale Fungicide Production
The agricultural chemical industry is constantly seeking novel molecular scaffolds that offer superior efficacy against resistant plant pathogens while maintaining economic viability in production. Patent CN103864801A introduces a significant breakthrough in this domain by disclosing a class of novel pyrazole spiro derivatives with potent fungicidal activity. This technology leverages a sophisticated one-step synthesis strategy that transforms electron-deficient olefins and azodicarboxylates into complex spirocyclic structures using triphenylphosphine as a key reagent. For R&D directors and procurement specialists seeking a reliable agrochemical intermediate supplier, understanding the underlying chemistry of this patent is crucial for evaluating its potential in next-generation fungicide formulations. The structural versatility of these compounds, defined by variable substituents on the indole or benzofuran backbone, allows for fine-tuning of biological properties to meet specific field requirements. As we delve into the technical specifics, it becomes evident that this methodology represents a paradigm shift from laborious multi-step syntheses to a more streamlined, efficient process.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of pyrazole derivatives has relied heavily on condensation reactions that often necessitate multiple transformation steps to achieve the desired cyclic structure. These traditional pathways frequently involve harsh reaction conditions, including high temperatures and the use of aggressive catalysts that can complicate downstream purification and waste management. For cost reduction in fungicide manufacturing, such inefficiencies are detrimental as they increase energy consumption and extend production cycles. Furthermore, conventional methods often struggle with regioselectivity, leading to complex impurity profiles that require extensive chromatographic separation, thereby reducing overall yield and increasing the cost of goods sold. The reliance on precious metal catalysts in some older protocols also introduces supply chain vulnerabilities and environmental compliance challenges regarding heavy metal residues in the final active ingredient. These factors collectively hinder the commercial scale-up of complex agrochemical intermediates, making it difficult for manufacturers to respond quickly to market demands for new fungicidal solutions.
The Novel Approach
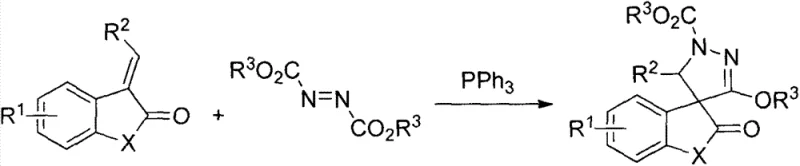
In stark contrast, the methodology outlined in CN103864801A utilizes a Huisgen zwitterion intermediate generated in situ from azodicarboxylates and triphenylphosphine to drive the cycloaddition reaction. This innovative approach allows for the construction of the pyrazole spiro core in a single operational step under remarkably mild conditions, specifically at room temperature. By eliminating the need for thermal activation and complex catalytic systems, this process inherently reduces the energy footprint and operational risks associated with high-pressure or high-temperature reactors. The reaction demonstrates excellent tolerance for various functional groups, enabling the synthesis of a diverse library of derivatives with high structural fidelity. For supply chain leaders focused on reducing lead time for high-purity agrochemical intermediates, this simplification translates directly into faster batch turnover and more predictable production schedules. The ability to achieve high yields, reported in the patent data to range significantly depending on the specific substituents, underscores the robustness of this chemical transformation for industrial applications.
Mechanistic Insights into Huisgen Zwitterion-Mediated Cyclization
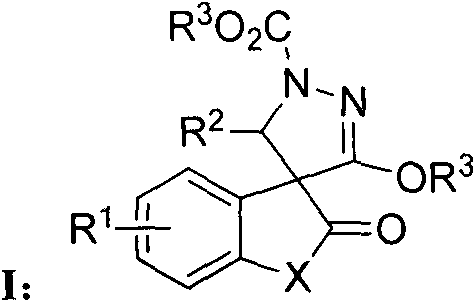
The core of this technological advancement lies in the generation and reactivity of the Huisgen zwitterion, a highly reactive species formed by the nucleophilic attack of triphenylphosphine on the azodicarboxylate. This intermediate acts as a 1,3-dipole that undergoes a [3+2] cycloaddition with the electron-deficient olefin derived from isatin or benzofurandione. The mechanism proceeds through a concerted pathway that efficiently constructs the five-membered pyrazole ring while simultaneously forming the spiro junction at the C-3 position of the oxindole or benzofuranone core. This mechanistic pathway is particularly advantageous because it avoids the formation of unstable intermediates that often plague traditional pyrazole syntheses. Understanding this mechanism is vital for R&D teams aiming to optimize reaction parameters for high-purity pyrazole spiro derivatives, as it highlights the critical stoichiometric balance required between the phosphine, the azo compound, and the olefin substrate to minimize side reactions.
Impurity control in this synthesis is inherently managed by the specificity of the zwitterion-olefin interaction, which favors the formation of the desired spirocyclic product over linear oligomers or alternative cyclization isomers. The patent data indicates that the reaction mixture can be purified effectively using standard silica gel column chromatography with a petroleum ether and ethyl acetate system, suggesting that the byproduct profile is relatively clean and manageable. For quality assurance professionals, this implies that achieving stringent purity specifications is feasible without resorting to exotic or prohibitively expensive purification technologies. The structural integrity of the spiro center is maintained throughout the process, ensuring that the biological activity associated with this specific 3D conformation is preserved in the final product. This level of mechanistic control is essential for developing consistent commercial batches that meet the rigorous regulatory standards required for agricultural chemical registration.
How to Synthesize Pyrazole Spiro Derivatives Efficiently
Implementing this synthesis route requires careful attention to reagent quality and atmospheric conditions to maximize the efficiency of the Huisgen zwitterion formation. The standard protocol involves dissolving the electron-deficient olefin, azodicarboxylate, and triphenylphosphine in a suitable organic solvent such as tetrahydrofuran or dichloromethane under a nitrogen atmosphere. The detailed standardized synthesis steps are provided in the guide below, which outlines the precise molar ratios and workup procedures necessary to replicate the high yields reported in the patent literature. Adhering to these parameters ensures that the reaction proceeds to completion within the specified timeframe, typically around 24 hours at ambient temperature. This operational simplicity makes the process highly attractive for pilot plant studies and subsequent technology transfer to large-scale manufacturing facilities.
- Dissolve electron-deficient olefin, azodicarboxylate, and triphenylphosphine in an organic solvent such as THF or DCM under nitrogen protection.
- Stir the reaction mixture at room temperature for approximately 24 hours to allow the Huisgen zwitterion intermediate to react completely.
- Remove the solvent via rotary evaporation and purify the crude product using silica gel column chromatography with a petroleum ether and ethyl acetate gradient.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthesis technology offers substantial strategic benefits for procurement managers and supply chain directors looking to optimize their sourcing strategies for agrochemical intermediates. The elimination of multi-step sequences and harsh reaction conditions directly correlates with a reduction in processing time and utility consumption, leading to significant operational cost savings. By simplifying the manufacturing workflow, companies can mitigate the risks associated with complex supply chains that rely on multiple specialized reagents and catalysts. This streamlined approach enhances supply chain reliability by reducing the number of potential failure points in the production process, ensuring a more consistent flow of materials to formulation plants. Furthermore, the use of readily available starting materials such as triphenylphosphine and common azodicarboxylates minimizes exposure to volatile raw material markets, stabilizing long-term cost structures.
- Cost Reduction in Manufacturing: The one-step nature of this reaction eliminates the need for intermediate isolation and purification stages that are typical in conventional pyrazole synthesis, thereby drastically reducing labor and solvent costs. The absence of expensive transition metal catalysts removes the financial burden associated with catalyst recovery and heavy metal testing, further lowering the overall cost of production. Additionally, the mild reaction conditions reduce energy expenditures related to heating and cooling, contributing to a more sustainable and economically efficient manufacturing profile. These cumulative efficiencies allow for a more competitive pricing structure in the global agrochemical market without compromising on product quality.
- Enhanced Supply Chain Reliability: Sourcing strategies benefit from the use of commodity chemicals that are widely available from multiple global suppliers, reducing the risk of single-source bottlenecks. The robustness of the reaction at room temperature means that production is less susceptible to disruptions caused by utility failures or equipment limitations in regions with less developed infrastructure. This resilience ensures that delivery schedules can be maintained even under challenging operational conditions, providing greater certainty for downstream formulation partners. The ability to scale the reaction proportionally from laboratory to industrial volumes without significant re-optimization further strengthens the reliability of the supply chain.
- Scalability and Environmental Compliance: The process generates minimal hazardous waste compared to traditional methods, simplifying effluent treatment and reducing the environmental footprint of the manufacturing site. The use of standard organic solvents that can be recovered and recycled aligns with modern green chemistry principles and regulatory expectations for sustainable production. Scalability is facilitated by the exothermic nature of the zwitterion formation being manageable at room temperature, allowing for safe operation in large reactors without complex thermal control systems. This compliance readiness accelerates the regulatory approval process for new fungicide products containing these intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patent technology in industrial settings. These answers are derived directly from the experimental data and technical disclosures found within the patent documentation to ensure accuracy and relevance. Understanding these details is critical for stakeholders evaluating the feasibility of integrating this chemistry into their existing product portfolios. The information provided here serves as a foundational reference for further technical discussions and feasibility assessments with our engineering team.
Q: What is the primary advantage of this synthesis method over conventional pyrazole production?
A: Unlike traditional multi-step condensation methods, this patent describes a one-step transformation using Huisgen zwitterions, which drastically simplifies the process and reduces operational complexity.
Q: What is the biological efficacy of these pyrazole spiro derivatives?
A: The compounds exhibit excellent fungicidal activity against a broad spectrum of plant pathogens, including Alternaria Solani and Gibberella Zeae, often surpassing standard control agents like azoxystrobin.
Q: Are the reaction conditions suitable for large-scale manufacturing?
A: Yes, the reaction proceeds at room temperature under nitrogen protection without requiring extreme heating or pressure, making it highly amenable to commercial scale-up and safe industrial operation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrazole Spiro Derivatives Supplier
The technical potential of the pyrazole spiro derivatives described in CN103864801A is immense, offering a pathway to high-performance fungicides that address current resistance challenges in agriculture. NINGBO INNO PHARMCHEM stands ready to support the development and commercialization of these molecules, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped with rigorous QC labs and stringent purity specifications to ensure that every batch meets the exacting standards required for global agrochemical registration. We understand the critical importance of consistency and quality in the supply of active ingredients and intermediates, and our team is dedicated to delivering solutions that enhance your competitive edge in the market.
We invite you to engage with our technical procurement team to discuss how this technology can be adapted to your specific needs. Request a Customized Cost-Saving Analysis to understand the economic impact of switching to this streamlined synthesis route for your product line. Our experts are available to provide specific COA data and route feasibility assessments to help you make informed decisions about your supply chain strategy. By partnering with us, you gain access to a wealth of chemical expertise and manufacturing capacity designed to accelerate your time to market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
