Optimizing Vinpocetine Production: A High-Yield Semi-Synthetic Route for Global Pharmaceutical Supply Chains
Optimizing Vinpocetine Production: A High-Yield Semi-Synthetic Route for Global Pharmaceutical Supply Chains
The global demand for cerebrovascular therapeutic agents continues to rise, driven by an aging population and the increasing prevalence of cognitive disorders. At the forefront of this market is Vinpocetine, a potent derivative of the indole alkaloid Vincamine, renowned for its ability to improve cerebral blood flow and metabolism. However, the commercial viability of this critical active pharmaceutical ingredient (API) often hinges on the efficiency and sustainability of its manufacturing process. Patent CN102702191B introduces a refined semi-synthetic methodology that addresses historical bottlenecks in Vinpocetine production. This technical insight report analyzes the proprietary synthesis route disclosed in the patent, highlighting its potential to redefine supply chain reliability and cost structures for international pharmaceutical manufacturers seeking a reliable vinpocetine supplier.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of Vinpocetine has been plagued by significant technical and economic challenges that hinder efficient cost reduction in pharmaceutical intermediates manufacturing. Traditional total synthesis routes, while chemically elegant, often involve excessive step counts and the utilization of hazardous reagents such as 2-fluoro-1,3,5-trinitrobenzene, which poses severe safety risks and environmental disposal issues. Furthermore, earlier semi-synthetic approaches utilizing titanium tetraethoxide (Ti(OEt)4) as a catalyst for transesterification have demonstrated suboptimal total recovery rates, leading to substantial material loss and inflated production costs. These legacy methods frequently require complex purification protocols, including column chromatography, to remove trace metal impurities and side products, thereby extending lead times and complicating the commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
In stark contrast to these cumbersome legacy protocols, the methodology outlined in patent CN102702191B offers a streamlined, two-step transformation that maximizes atom economy and operational simplicity. By leveraging the natural chirality of Vincamine extracted from Apocynaceae periwinkle, this route bypasses the need for asymmetric synthesis entirely. The core innovation lies in the strategic activation of the C-14 hydroxyl group followed by a controlled base-mediated elimination. This approach not only drastically simplifies the reaction workflow but also utilizes inexpensive, commodity-grade reagents such as methanesulfonyl chloride and sodium ethoxide. The result is a robust process capable of delivering high-purity Vinpocetine with minimal environmental footprint, directly addressing the industry's urgent need for greener and more economical manufacturing solutions.
Mechanistic Insights into Base-Catalyzed Elimination and Rearrangement
The chemical elegance of this synthesis is rooted in a precise two-stage mechanistic sequence that ensures high fidelity in product formation. The first stage involves the conversion of the secondary alcohol at the C-14 position of the Vincamine scaffold into a mesylate ester. This activation step is critical, as it transforms a poor leaving group (hydroxyl) into an excellent leaving group (mesylate), setting the stage for the subsequent elimination. The reaction is conducted in toluene with triethylamine acting as a proton scavenger, ensuring that the acidic byproduct (HCl) is neutralized immediately to prevent degradation of the sensitive indole alkaloid structure. This mild condition preserves the stereochemical integrity of the molecule while preparing it for the decisive structural modification.
The second stage constitutes the heart of the transformation, where the mesylate intermediate undergoes a base-catalyzed elimination reaction to form the exocyclic double bond characteristic of the apovincamine skeleton. Upon treatment with sodium ethoxide in ethanol at elevated temperatures, the molecule undergoes an E2-type elimination coupled with a skeletal rearrangement. This step is highly sensitive to thermal conditions; maintaining the reaction at approximately 80°C allows for the complete conversion of the intermediate while minimizing the formation of polymeric byproducts. The mechanism effectively expels the mesylate group and a proton from the adjacent carbon, establishing the conjugated system essential for the pharmacological activity of Vinpocetine. 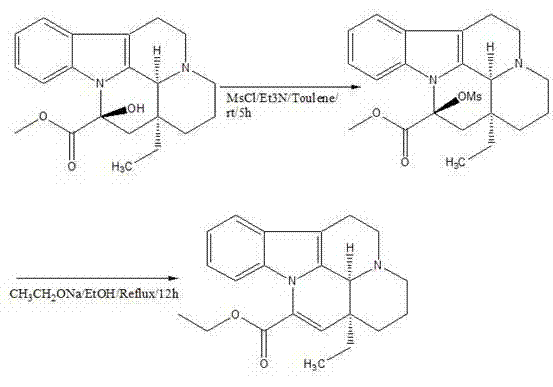
How to Synthesize Vinpocetine Efficiently
Implementing this synthesis requires strict adherence to the specified stoichiometric ratios and thermal profiles to ensure optimal yield and purity. The process begins with the dissolution of Vincamine in toluene, followed by the controlled addition of methanesulfonyl chloride under cooling to manage the exotherm. Once the intermediate is isolated, it is immediately subjected to the elimination conditions using freshly prepared sodium ethoxide. The detailed standardized operating procedures, including specific stirring rates, addition times, and quenching protocols necessary for GMP-compliant production, are outlined below to guide process engineers in replicating this high-efficiency route.
- Activate the hydroxyl group of Vincamine using Methanesulfonyl chloride (MsCl) and Triethylamine in Toluene to form the mesylate intermediate.
- Perform base-catalyzed elimination using Sodium Ethoxide in Ethanol at reflux temperatures to induce rearrangement and double bond formation.
- Isolate the final product through acid-base extraction and filtration, achieving high purity without recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route translates into tangible strategic benefits that extend beyond mere chemical yield. By eliminating the reliance on exotic catalysts and reducing the number of unit operations, the process inherently lowers the barrier to entry for large-scale production. The use of common solvents like toluene and ethanol simplifies solvent recovery and recycling systems, contributing to a significant reduction in overall operating expenses. Furthermore, the high purity achieved directly from crystallization reduces the dependency on resource-intensive purification steps, allowing for faster batch turnover and improved responsiveness to market demand fluctuations.
- Cost Reduction in Manufacturing: The economic model of this process is fundamentally superior due to the substitution of expensive transition metal catalysts with inexpensive organic bases. By removing the need for titanium-based reagents and avoiding the toxic nitro-compounds used in total synthesis, the raw material costs are drastically simplified. Additionally, the high yield reported in the patent embodiments implies a substantial decrease in the cost of goods sold (COGS) per kilogram, as less starting material is wasted to side reactions. This efficiency allows manufacturers to offer competitive pricing without compromising on quality standards.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of readily available starting materials and reagents. Vincamine, being a natural extract, has a well-established supply network, and the reagents MsCl and NaOEt are commodity chemicals produced globally in vast quantities. This reduces the risk of supply disruptions caused by the scarcity of specialized catalysts. Moreover, the robustness of the reaction conditions means that the process is less susceptible to minor variations in raw material quality, ensuring consistent output and reliable delivery schedules for downstream API manufacturers.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route offers a cleaner profile that aligns with increasingly stringent global regulations. The avoidance of heavy metals and highly toxic nitrated aromatics simplifies waste stream management and reduces the cost of effluent treatment. The process is inherently scalable, as demonstrated by the successful execution in multi-liter reactors described in the patent, making it suitable for commercial scale-up of complex pharmaceutical intermediates. This environmental compliance not only mitigates regulatory risk but also enhances the corporate sustainability profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Vinpocetine synthesis route. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity on process capabilities and product quality attributes. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this method into their existing production portfolios.
Q: What are the key advantages of this semi-synthetic route over total synthesis?
A: This route utilizes naturally derived Vincamine as a starting material, significantly shortening the synthetic sequence compared to total synthesis. It avoids the use of rare and toxic catalysts like 2-fluoro-1,3,5-trinitrobenzene, resulting in a more environmentally friendly process with higher overall yields.
Q: How is product purity controlled in this manufacturing process?
A: The process achieves high purity (up to 99%) through precise control of reaction conditions, specifically the temperature during the elimination step and the pH regulation during workup. The method eliminates the need for complex chromatographic purification, relying instead on efficient crystallization and filtration.
Q: Is this process scalable for industrial production?
A: Yes, the process is designed for scalability. It uses common solvents like Toluene and Ethanol and avoids hazardous reagents that complicate large-scale handling. The simple workup procedure involving filtration and extraction facilitates easy transition from laboratory to commercial scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Vinpocetine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires deep technical expertise and robust infrastructure. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this synthesis are fully realized in practice. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications, consistently meeting or exceeding the 99% purity benchmark established in the patent examples. We are committed to delivering high-purity pharmaceutical intermediates that empower our partners to bring life-saving medications to market faster.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this optimized synthesis technology. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to request specific COA data and comprehensive route feasibility assessments, ensuring that your supply chain is built on a foundation of scientific excellence and commercial reliability.
