Advanced Manufacturing of Pitavastatin Calcium Intermediates via Stereoselective Routes
Advanced Manufacturing of Pitavastatin Calcium Intermediates via Stereoselective Routes
The pharmaceutical industry continuously seeks robust, scalable, and environmentally benign pathways for the production of high-value statin intermediates. Patent CN102898367B discloses a sophisticated preparation method for a pitavastatin calcium crude drug intermediate, addressing critical bottlenecks in stereocontrol and process safety. This technology leverages a modified Friedländer condensation followed by a highly stereoselective alkyne reduction to construct the core quinoline scaffold and the essential heptenoic acid side chain. By shifting away from toxic cyanide-based elongation strategies, this route offers a compelling value proposition for manufacturers aiming to optimize their supply chain for next-generation lipid-lowering agents.
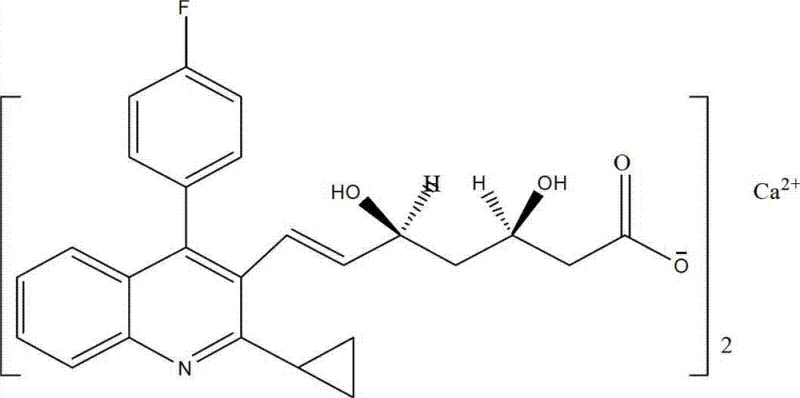
The structural complexity of pitavastatin calcium, characterized by its fluorophenyl-quinoline moiety and specific (3R, 5S) dihydroxy configuration, demands precise synthetic control. The disclosed method achieves this through a convergent strategy that couples a pre-functionalized quinoline ring with a chiral side chain precursor. This approach not only streamlines the synthesis but also ensures that the critical geometric and optical purity specifications are met early in the process, reducing the burden on downstream purification units.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of pitavastatin intermediates has been plagued by significant safety and efficiency challenges. Several prior art routes, such as those documented in Bull. Chem. Soc. Jpn., rely on the use of potassium cyanide for opening epoxide rings or elongating carbon chains. The handling of cyanide salts on an industrial scale introduces severe occupational health risks and necessitates expensive waste treatment protocols to neutralize toxic effluents. Furthermore, alternative routes involving noble metal catalysts or thiophenyl group introductions often suffer from low overall yields due to the formation of multiple chiral isomers. The separation of these diastereomers typically requires tedious chromatography or repeated crystallizations, which drastically increases production costs and extends lead times, rendering such processes economically unviable for large-scale commercialization.
The Novel Approach
The methodology outlined in CN102898367B represents a paradigm shift towards greener and more efficient manufacturing. By utilizing a bromoethyl cyclopropyl ketone condensation followed by coupling in liquid ammonia, the process bypasses the need for toxic cyanide reagents entirely. A key innovation lies in the reduction step, where sodium bis(2-methoxyethoxy)aluminum hydride (Red-Al) is employed to convert the alkyne functionality directly into the desired trans-olefin. This reagent choice is pivotal, as it provides exceptional stereocontrol without the need for precious metal catalysts. The result is a streamlined workflow that maintains high optical purity throughout the sequence, significantly simplifying the isolation of the final intermediate.
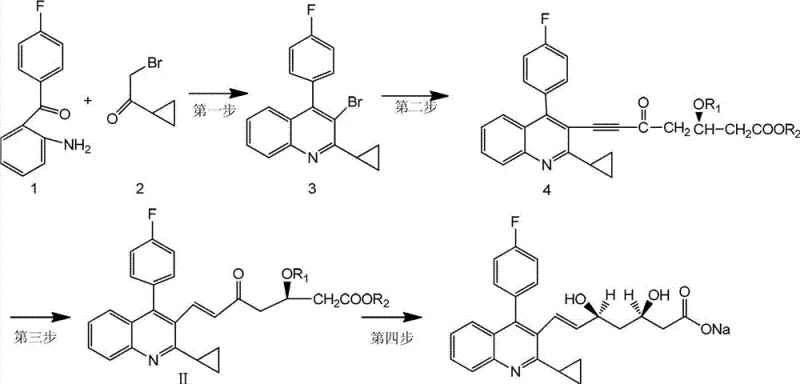
Mechanistic Insights into Friedländer Condensation and Stereoselective Reduction
The synthesis initiates with a classic Friedländer condensation between 2-amino-4'-fluorobenzophenone and bromoethyl cyclopropyl ketone. Catalyzed by a mixture of sulfuric acid and acetic acid in solvents like tetrahydrofuran or acetonitrile, this cyclization efficiently constructs the 2-cyclopropyl-4-(4-fluorophenyl)-3-bromoquinoline core. The reaction conditions are optimized to operate between 60°C and 160°C, ensuring complete conversion while minimizing degradation of the sensitive cyclopropyl ring. Following this, the quinoline bromide undergoes nucleophilic substitution with a chiral alkyne side chain in liquid ammonia at cryogenic temperatures (-70°C to -50°C). This low-temperature environment is crucial for suppressing side reactions and preserving the integrity of the chiral centers present in the side chain precursor.
The subsequent reduction of the alkyne to the trans-alkene is the mechanistic highlight of this process. Sodium bis(2-methoxyethoxy)aluminum hydride acts as a source of hydride ions that add across the triple bond in a specific manner to favor the trans geometry. Unlike catalytic hydrogenation which may over-reduce or produce cis-isomers depending on the catalyst poison used, this stoichiometric reduction offers predictable and robust stereochemical outcomes. The final stage involves hydroboration using diethylmethylborane followed by oxidation with sodium borohydride. This sequence installs the requisite hydroxyl groups with high regioselectivity. The entire pathway is designed to minimize impurity generation, with HPLC analysis of the final product indicating purity levels exceeding 99.7%, thereby reducing the need for extensive recrystallization steps.
How to Synthesize Pitavastatin Calcium Intermediate Efficiently
The execution of this synthesis requires strict adherence to temperature controls and reagent stoichiometry to maximize yield and safety. The process is divided into four distinct operational stages, beginning with the acid-catalyzed cyclization and concluding with the hydrolysis of the ester to the carboxylate salt. Each step has been optimized for scalability, utilizing common industrial solvents such as toluene, THF, and ethanol. The detailed standardized operating procedures, including specific addition rates and quenching protocols necessary for safe handling of reactive aluminum hydrides and boranes, are critical for successful technology transfer.
- Condense 2-amino-4'-fluorobenzophenone with bromoethyl cyclopropyl ketone using acid catalysis to form the quinoline core.
- Couple the quinoline bromide with a chiral alkyne side chain precursor in liquid ammonia at low temperature.
- Reduce the alkyne moiety to a trans-olefin using sodium bis(2-methoxyethoxy)aluminum hydride (Red-Al).
- Perform hydroboration-oxidation and subsequent hydrolysis to yield the final dihydroxy heptenoic acid intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented process translates into tangible operational improvements and risk mitigation. By eliminating the reliance on highly regulated and toxic raw materials like potassium cyanide, facilities can significantly reduce their environmental compliance costs and insurance liabilities. The simplified work-up procedures, which avoid complex chiral separations, allow for faster batch turnover and higher throughput in existing reactor trains. Furthermore, the high yields reported in the patent embodiments suggest a substantial reduction in raw material consumption per kilogram of finished intermediate, directly impacting the cost of goods sold (COGS).
- Cost Reduction in Manufacturing: The elimination of expensive noble metal catalysts and toxic cyanide reagents leads to a direct decrease in raw material expenditures. Additionally, the high stereospecificity of the Red-Al reduction minimizes the loss of valuable chiral intermediates to unwanted isomers, effectively increasing the overall mass balance of the process. This efficiency gain allows manufacturers to achieve substantial cost savings without compromising on the quality of the active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: The starting materials, such as 2-amino-4'-fluorobenzophenone and cyclopropyl ketones, are commercially available commodity chemicals with stable supply lines. By avoiding proprietary or hard-to-source chiral auxiliaries that require long lead times, manufacturers can maintain a more agile and responsive inventory strategy. The robustness of the reaction conditions also reduces the risk of batch failures, ensuring consistent delivery schedules to downstream API producers.
- Scalability and Environmental Compliance: The process utilizes solvents and reagents that are well-suited for large-scale industrial application, with established protocols for recovery and recycling. The absence of heavy metal waste streams simplifies wastewater treatment requirements, aligning with increasingly stringent global environmental regulations. This 'green chemistry' profile not only facilitates regulatory approval but also enhances the corporate sustainability metrics of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the comparative data and experimental examples provided in the patent literature, offering a clear perspective on the operational benefits and chemical rationale behind the method.
Q: How does this process improve safety compared to traditional statin synthesis?
A: Unlike prior art routes that utilize highly toxic potassium cyanide for side-chain elongation, this patented method employs safer reagents like liquid ammonia and Red-Al, significantly reducing hazardous waste handling requirements.
Q: What is the stereochemical outcome of the reduction step?
A: The use of sodium bis(2-methoxyethoxy)aluminum hydride ensures high stereospecificity, selectively producing the thermodynamically stable trans-olefin geometry required for biological activity, minimizing isomer separation costs.
Q: Is this method suitable for large-scale commercial production?
A: Yes, the process features mild reaction conditions, high yields (up to 94% in initial steps), and avoids complex chromatographic separations, making it highly robust for multi-kilogram to ton-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pitavastatin Calcium Intermediate Supplier
At NINGBO INNO PHARMCHEM, we understand the critical importance of purity and consistency in the production of cardiovascular therapeutics. As a specialized CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific safety requirements of hydride reductions and cryogenic reactions, ensuring that every batch of pitavastatin intermediate meets stringent purity specifications. Our rigorous QC labs employ advanced analytical techniques to verify stereochemical integrity and impurity profiles, guaranteeing a product that is ready for immediate API synthesis.
We invite global partners to collaborate with us to leverage this advanced manufacturing technology for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage potential clients to contact us directly to obtain specific COA data and comprehensive route feasibility assessments, ensuring that your project moves from development to commercial success with maximum efficiency.
