Revolutionizing Triarylphosphine Production: A Metal-Free Route for High-Purity Pharmaceutical Intermediates
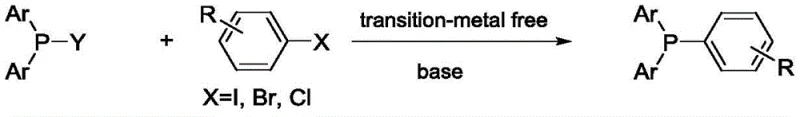
The landscape of fine chemical synthesis is undergoing a significant transformation with the emergence of greener, more efficient methodologies, as exemplified by the groundbreaking technology disclosed in patent CN109970789B. This intellectual property introduces a novel preparation method for triarylphosphine compounds that fundamentally challenges the status quo of transition-metal dependency in organophosphorus chemistry. Traditionally, the construction of the phosphorus-carbon bond in these valuable ligands has relied heavily on precious metal catalysts or hazardous organometallic reagents, creating bottlenecks in both cost and purity for downstream applications. By leveraging a unique base-mediated coupling strategy, this invention enables the direct assembly of triarylphosphines from diphenylphosphine precursors and aryl halides under remarkably mild conditions. For R&D directors and procurement specialists in the pharmaceutical sector, this represents a paradigm shift towards more sustainable and economically viable manufacturing processes for critical intermediates used in catalysis and organic synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial and laboratory-scale synthesis of triarylphosphine compounds has been dominated by the Grignard reagent method, a process fraught with significant operational hazards and technical limitations. This conventional approach necessitates the generation of highly reactive organomagnesium species from halogenated benzenes and metallic magnesium, typically requiring strictly anhydrous conditions and cryogenic temperatures often dropping to minus 40 degrees Celsius to control exothermicity. The inherent instability and pyrophoric nature of Grignard reagents pose severe safety risks during large-scale handling, while their extreme nucleophilicity severely restricts substrate scope, often leading to side reactions with sensitive functional groups on the aromatic ring. Furthermore, alternative metal-catalyzed cross-coupling routes frequently suffer from low overall yields and require complex ligand systems, making the expansion of substrate diversity difficult and the purification of the final product from trace metal contaminants a costly and time-consuming endeavor.
The Novel Approach
In stark contrast to these legacy techniques, the methodology outlined in CN109970789B offers a streamlined, transition-metal-free pathway that constructs the triarylphosphine skeleton in a single step with exceptional efficiency. By utilizing simple inorganic or organic bases such as sodium hydroxide, potassium acetate, or triethylamine, the reaction proceeds smoothly at temperatures ranging from ambient 25 degrees Celsius up to 120 degrees Celsius, eliminating the need for energy-intensive cooling or specialized high-pressure equipment. This innovative protocol not only simplifies the operational workflow by removing the requirement for inert atmosphere gloveboxes typically needed for air-sensitive reagents but also dramatically broadens the range of compatible substrates, including various substituted aryl iodides, bromides, and even chlorides. The result is a robust synthetic platform that delivers high-purity products with minimal byproduct formation, addressing the critical pain points of safety, cost, and scalability that have long plagued the production of these essential chemical building blocks.
Mechanistic Insights into Base-Mediated Phosphorus-Carbon Bond Formation
The core innovation of this technology lies in its ability to facilitate nucleophilic aromatic substitution or related coupling mechanisms without the assistance of transition metal catalysts, which are traditionally viewed as indispensable for activating aryl halides. In this metal-free system, the base plays a dual role: it activates the diphenylphosphine species to enhance its nucleophilicity and potentially assists in the leaving group departure from the aryl halide electrophile. This direct activation pathway bypasses the oxidative addition and reductive elimination cycles characteristic of palladium or nickel catalysis, thereby removing the risk of metal leaching into the final product. For pharmaceutical applications where residual metal limits are strictly regulated, this mechanistic advantage is paramount, as it inherently produces a cleaner crude reaction mixture that requires less aggressive purification strategies. The absence of metal-ligand complexes also means that the reaction kinetics are governed purely by electronic and steric factors of the substrates, allowing for predictable optimization across a wide array of structural analogues.
Furthermore, the impurity profile generated by this base-mediated process is significantly simpler compared to metal-catalyzed counterparts, which often produce homocoupling byproducts or dehalogenated species due to competing catalytic cycles. The controlled basic environment ensures that the primary reaction pathway favors the desired P-C bond formation, minimizing the formation of phosphine oxides or other oxidized impurities that can degrade the performance of the ligand in subsequent catalytic applications. This high level of chemoselectivity is particularly beneficial when synthesizing complex triarylphosphines containing sensitive functional groups, as the mild reaction conditions prevent decomposition or unwanted side reactions. Consequently, the final isolation of the target molecule is straightforward, often achievable through standard extraction and crystallization techniques, yielding a product with the stringent purity specifications required for use in high-value organic synthesis and drug discovery programs.
How to Synthesize Triarylphosphine Efficiently
The practical implementation of this synthesis route is designed for ease of adoption in both laboratory and pilot plant settings, requiring only standard chemical processing equipment and readily available reagents. The process begins by charging a reaction vessel with the diphenylphosphine precursor and the chosen aryl halide, followed by the addition of a polar or non-polar solvent such as acetonitrile, toluene, or dimethyl sulfoxide depending on the solubility profile of the substrates. Once the base is introduced, the mixture is heated to the specified temperature range, allowing the coupling to proceed to completion over a period of 1 to 36 hours, after which standard aqueous workup and chromatographic purification yield the pure triarylphosphine. Detailed standardized synthesis steps for specific derivatives are provided in the guide below to assist technical teams in replicating these results.
- Mix a diphenylphosphine compound and an aryl halide in a suitable solvent such as acetonitrile or toluene.
- Add an organic or inorganic base like sodium hydroxide or potassium acetate to the reaction mixture.
- Heat the mixture between 25°C and 120°C for 1 to 36 hours to complete the coupling reaction without metal catalysts.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this transition-metal-free technology offers profound benefits for procurement managers and supply chain leaders seeking to optimize the cost structure and reliability of their chemical supply chains. By completely eliminating the need for expensive precious metal catalysts such as palladium or platinum, the direct material costs associated with the synthesis are drastically reduced, removing a significant variable from the pricing model of these intermediates. Moreover, the simplified post-treatment process, which no longer requires specialized metal scavenging resins or complex filtration steps to meet regulatory limits on heavy metals, translates into substantial savings in processing time and consumable expenses. This streamlined workflow enhances the overall throughput of the manufacturing facility, allowing for faster turnaround times and more responsive fulfillment of customer orders in the competitive fine chemicals market.
- Cost Reduction in Manufacturing: The removal of transition metal catalysts represents a major economic advantage, as these materials are not only costly to purchase but also add significant expense to the waste disposal and purification stages of production. Without the need for metal scavengers or extensive chromatography to remove trace residues, the operational expenditure per kilogram of product is significantly lowered, improving the margin profile for manufacturers. Additionally, the use of inexpensive and abundant bases like sodium hydroxide or potassium acetate further drives down the raw material bill, making the process highly attractive for large-scale commercial production where cost efficiency is the primary driver of competitiveness.
- Enhanced Supply Chain Reliability: Relying on a synthesis route that utilizes commodity chemicals rather than specialized catalytic systems mitigates the risk of supply disruptions caused by the volatility of the precious metals market. The raw materials required for this process, including aryl halides and diphenylphosphines, are widely available from multiple global suppliers, ensuring a stable and continuous flow of inputs for manufacturing operations. This diversification of the supply base reduces dependency on single-source vendors for critical catalysts, thereby strengthening the resilience of the supply chain against geopolitical or logistical shocks that could otherwise halt production lines.
- Scalability and Environmental Compliance: The mild reaction conditions and absence of toxic heavy metals make this process inherently safer and more environmentally friendly, aligning perfectly with modern green chemistry principles and increasingly stringent environmental regulations. Scaling up the reaction does not require complex engineering controls for handling pyrophoric reagents or high-pressure hydrogenation equipment, facilitating a smoother transition from bench scale to multi-ton production. The reduction in hazardous waste generation, particularly metal-contaminated sludge, simplifies waste management protocols and lowers the environmental compliance burden, positioning manufacturers as responsible stewards of sustainability in the chemical industry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel triarylphosphine synthesis technology, providing clarity for stakeholders evaluating its potential impact on their operations. These insights are derived directly from the experimental data and technical specifications detailed in the patent documentation, ensuring accuracy and relevance for decision-makers. Understanding these nuances is crucial for assessing the feasibility of integrating this method into existing production workflows or new product development pipelines.
Q: What are the advantages of this metal-free synthesis over traditional Grignard methods?
A: Unlike traditional Grignard methods which require hazardous pyrophoric reagents and cryogenic conditions, this patented process operates under mild heating (25-120°C) with simple bases, significantly improving operational safety and substrate compatibility.
Q: Does this method eliminate the need for expensive transition metal catalysts?
A: Yes, the process explicitly avoids the use of palladium, nickel, or other transition metals, thereby removing the costly and complex downstream purification steps required to remove trace metal residues from pharmaceutical intermediates.
Q: What types of aryl halides are compatible with this reaction?
A: The method demonstrates excellent substrate compatibility, successfully reacting with iodobenzene, bromobenzene, and various substituted aryl chlorides to produce diverse triarylphosphine derivatives including biphenyl and naphthyl variants.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Triarylphosphine Supplier
As the demand for high-performance ligands and intermediates continues to grow across the pharmaceutical and agrochemical sectors, partnering with an experienced CDMO like NINGBO INNO PHARMCHEM ensures access to cutting-edge synthesis technologies and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, leveraging our deep expertise in process optimization to deliver consistent quality and supply security. We maintain stringent purity specifications through our rigorous QC labs, ensuring that every batch of triarylphosphine meets the exacting standards required for sensitive catalytic applications and drug synthesis, thereby minimizing risk for our global partners.
We invite you to engage with our technical procurement team to discuss how this metal-free synthesis route can be tailored to your specific project requirements and volume needs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this greener methodology for your supply chain. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive efficiency and innovation in your chemical sourcing strategy.
