Revolutionizing 2-Diarylmethylbenzofuran Production: A Scalable FeCl2-Catalyzed Oxidative Coupling Strategy
Revolutionizing 2-Diarylmethylbenzofuran Production: A Scalable FeCl2-Catalyzed Oxidative Coupling Strategy
The landscape of organic synthesis for bioactive heterocycles is constantly evolving, driven by the need for more efficient, cost-effective, and environmentally benign methodologies. A significant advancement in this domain is detailed in Chinese Patent CN112851608B, which discloses a robust catalytic oxidation synthesis method for 2-diarylmethylbenzofuran compounds. This technology represents a paradigm shift for manufacturers of pharmaceutical intermediates, offering a direct route to couple benzofuran derivatives with diarylmethanes using an inexpensive iron-based catalyst system. By leveraging FeCl2 as the catalyst and 2,3-dichloro-5,6-dicyano-1,4-benzoquinone (DDQ) as the oxidant, this process circumvents the limitations of previous art that relied on precious metals or harsh acidic conditions. For R&D directors and procurement managers alike, this patent provides a blueprint for producing high-purity pharmaceutical intermediates with improved economic viability and supply chain resilience.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-diarylmethylbenzofurans has been fraught with challenges that hinder large-scale commercial adoption. Prior art, such as methods reported in Tetrahedron (2007) and Organic Letters (2016), often necessitated the use of expensive and toxic catalysts like trifluoromethanesulfonic acid, palladium chloride, or scandium trifluoromethanesulfonate. Furthermore, many of these conventional routes required the presence of specific directing groups on the substrate to achieve regioselectivity, adding extra synthetic steps and increasing the overall cost of goods sold (COGS). Electrochemical methods, while innovative, often suffer from scalability issues and complex equipment requirements that are ill-suited for standard batch processing in fine chemical plants. These factors collectively create bottlenecks in the supply chain, leading to longer lead times and higher prices for the final active pharmaceutical ingredients (APIs).
The Novel Approach
The methodology outlined in patent CN112851608B addresses these pain points by introducing a direct oxidative coupling strategy that operates under relatively mild thermal conditions. By utilizing a combination of ferrous chloride (FeCl2) and DDQ in 1,2-dichloroethane, the reaction proceeds efficiently at temperatures between 90°C and 110°C. This approach eliminates the necessity for directing groups, allowing for the direct functionalization of the benzofuran C2 position with a wide variety of diarylmethanes. The simplicity of the reagent system—using earth-abundant iron instead of precious metals—drastically reduces the catalyst cost burden. Moreover, the workup procedure is straightforward, involving solvent evaporation and standard column chromatography, which facilitates easier purification and scale-up compared to methods requiring complex aqueous quenching or metal scavenging steps.
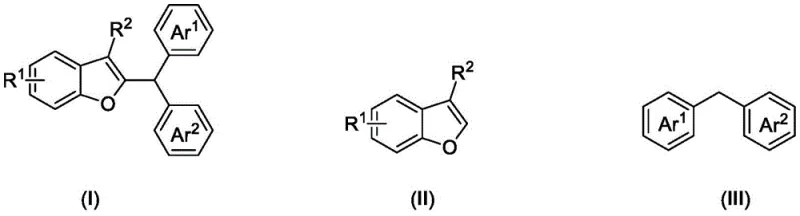
Mechanistic Insights into FeCl2-Catalyzed Oxidative Coupling
The core of this technological breakthrough lies in the synergistic interaction between the iron catalyst and the quinone oxidant. Mechanistically, the FeCl2 likely acts as a Lewis acid to activate the benzofuran ring or coordinate with the oxidant, facilitating the generation of a reactive carbocation or radical species from the diarylmethane substrate. The DDQ serves as a potent hydride acceptor, driving the dehydrogenative coupling forward by removing hydrogen equivalents from the reaction intermediate. This oxidative pathway ensures that the reaction proceeds to completion without the accumulation of reduced byproducts that could complicate downstream purification. The tolerance of this system to various electronic environments is remarkable; electron-rich and electron-deficient substituents on both the benzofuran and diarylmethane rings are well-tolerated, suggesting a robust catalytic cycle that is not easily poisoned by common functional groups found in drug-like molecules.
From an impurity control perspective, the absence of heavy metals like palladium is a critical advantage for pharmaceutical applications. Residual metal limits in APIs are strictly regulated, and removing trace palladium often requires specialized resin treatments that add cost and complexity. By switching to an iron-based system, the impurity profile is significantly simplified, as iron salts are generally easier to remove via aqueous washes or precipitation. Additionally, the reaction conditions (10-14 hours at 100°C) are optimized to balance conversion rates with selectivity, minimizing the formation of over-oxidized side products or polymerization of the reactive intermediates. This level of control is essential for maintaining the high purity specifications required by global regulatory bodies.
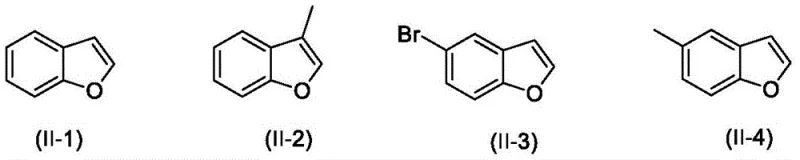
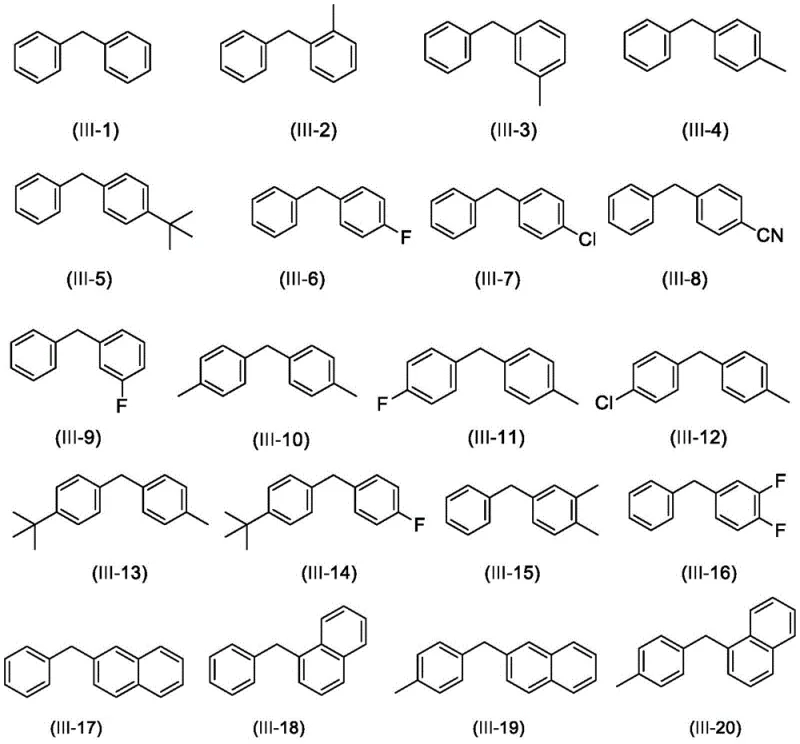
How to Synthesize 2-Diarylmethylbenzofuran Efficiently
Implementing this synthesis route in a production environment requires careful attention to stoichiometry and reaction parameters to maximize yield and safety. The patent specifies a mass ratio of benzofuran compounds to diarylmethane ranging from 100:150 to 100:700, indicating that an excess of the diarylmethane is often beneficial to drive the equilibrium towards the product. The catalyst loading is kept low, with a mass ratio of benzofuran to FeCl2 between 100:8 and 100:15, ensuring economic efficiency. The detailed standardized synthesis steps below outline the precise operational protocol derived from the patent examples, providing a clear roadmap for process chemists to replicate these results in the laboratory or pilot plant.
- Charge benzofuran compounds, diarylmethane, FeCl2 catalyst, and DDQ oxidant into a pressure-resistant bottle under nitrogen protection.
- Add 1,2-dichloroethane solvent and heat the mixture in an oil bath at 90-110°C for 10-14 hours to facilitate the coupling reaction.
- Upon completion, evaporate the solvent under reduced pressure and purify the crude product via column chromatography using ethyl acetate/petroleum ether.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this FeCl2-catalyzed process offers tangible strategic benefits beyond mere technical feasibility. The primary driver for adoption is the substantial reduction in raw material costs associated with replacing precious metal catalysts with commodity iron salts. This shift not only lowers the direct cost of manufacturing but also mitigates the supply risk associated with fluctuating prices of rare earth metals and palladium. Furthermore, the simplified downstream processing reduces the consumption of auxiliary materials such as metal scavengers and specialized filtration media, contributing to a leaner and more cost-effective production workflow.
- Cost Reduction in Manufacturing: The elimination of expensive catalysts like palladium chloride or scandium triflate results in immediate savings on the bill of materials. Since iron chloride is a bulk commodity chemical, its price stability ensures predictable budgeting for long-term production campaigns. Additionally, the avoidance of complex metal removal steps reduces the operational expenditure related to waste treatment and quality control testing for residual metals, further enhancing the overall profit margin for the final pharmaceutical intermediate.
- Enhanced Supply Chain Reliability: The starting materials, specifically benzofurans and diarylmethanes, are widely available from multiple global suppliers, reducing the risk of single-source dependency. The robustness of the reaction conditions means that the process is less sensitive to minor variations in reagent quality, which enhances batch-to-b consistency. This reliability is crucial for maintaining continuous supply to downstream API manufacturers, preventing costly production stoppages due to raw material shortages or quality deviations.
- Scalability and Environmental Compliance: The use of 1,2-dichloroethane as a solvent, while requiring proper handling, is a standard industrial solvent with established recovery and recycling protocols. The reaction does not generate hazardous gaseous byproducts, simplifying the engineering controls needed for scale-up. The straightforward workup procedure allows for easy adaptation to larger reactor volumes, facilitating the commercial scale-up of complex pharmaceutical intermediates from kilogram to multi-ton scales without significant re-engineering of the process infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims within patent CN112851608B, providing clarity on the scope and limitations of the method for potential licensees or manufacturing partners.
Q: What are the key advantages of the FeCl2/DDQ system over traditional palladium catalysts?
A: The FeCl2/DDQ system eliminates the need for expensive transition metals like palladium or scandium, significantly reducing raw material costs and simplifying heavy metal removal processes required for pharmaceutical compliance.
Q: Does this synthesis method require specific directing groups on the benzofuran ring?
A: No, a major breakthrough of patent CN112851608B is that it allows for direct coupling without the need for directing groups, expanding the scope of accessible derivatives and simplifying precursor synthesis.
Q: What is the typical yield range for this oxidative coupling reaction?
A: Depending on the substituents on the diarylmethane and benzofuran substrates, isolated yields typically range from moderate to high, with optimized examples achieving up to 84% yield under standard conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Diarylmethylbenzofuran Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the FeCl2-catalyzed oxidative coupling method for the production of high-value benzofuran derivatives. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 2-diarylmethylbenzofuran intermediates meets the exacting standards required for pharmaceutical applications.
We invite you to collaborate with us to leverage this advanced synthesis technology for your next drug development program. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you optimize your supply chain for maximum efficiency and cost-effectiveness.
