Advanced Photocatalytic Synthesis of Aryl Difluoromethyl Alkyl Sulfonyl Fluorides for Drug Discovery
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to access novel chemical spaces, particularly those enabled by click chemistry. Patent CN116253606A discloses a groundbreaking preparation method for aryl difluoromethyl alkyl sulfonyl fluoride compounds, which serve as critical warheads in Sulfur-Fluoride Exchange (SuFEx) reactions. This technology addresses the longstanding challenge of efficiently introducing sulfonyl fluoride motifs onto non-activated olefins under mild conditions. By leveraging a visible light-induced palladium catalytic system, this process enables the difunctionalization of olefins with aryl difluoromethyl radicals and sulfonyl fluoride groups in a single operational sequence. For R&D directors and procurement specialists, this represents a significant leap forward in accessing high-value building blocks for drug discovery and material science without the need for hazardous gaseous reagents or extreme thermal conditions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of alkyl sulfonyl fluorides has been fraught with significant operational hazards and synthetic limitations. Conventional routes often rely on the direct use of sulfur dioxide gas, which is toxic, corrosive, and requires specialized high-pressure equipment for safe handling, thereby increasing capital expenditure and safety risks in manufacturing facilities. Furthermore, existing methods for introducing fluorosulfonyl groups frequently suffer from poor functional group tolerance, necessitating extensive protection and deprotection strategies that drastically reduce overall atom economy. Many prior art processes require harsh oxidative conditions or cryogenic temperatures to stabilize reactive intermediates, leading to high energy consumption and limiting the scalability of these reactions for commercial production. The inability to efficiently functionalize non-activated olefins with high regioselectivity has also restricted the structural diversity accessible to medicinal chemists exploring SuFEx-based covalent inhibitors.
The Novel Approach
The methodology outlined in CN116253606A offers a transformative solution by utilizing a solid sulfur dioxide surrogate, DABSO, in conjunction with a visible light photocatalytic system. This approach eliminates the need for handling toxic SO2 gas, significantly enhancing workplace safety and simplifying the engineering controls required for scale-up. The reaction proceeds at room temperature under blue LED irradiation, which drastically reduces energy costs compared to thermal processes requiring heating or cooling. As illustrated in the general reaction scheme below, the process tolerates a wide array of substituents on the olefin substrate, including esters, amides, and heterocycles, allowing for the direct construction of complex molecular architectures.
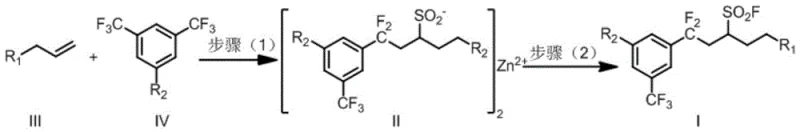
This novel pathway achieves the difunctionalization of non-activated olefins with high selectivity, introducing the sulfonyl fluoride group in situ without compromising other sensitive moieties. The use of readily available m-bistrifluoromethyl aromatic hydrocarbons as radical precursors further enhances the cost-effectiveness of the route, making it a highly attractive option for the reliable supply of pharmaceutical intermediates. By merging radical chemistry with transition metal catalysis, this method opens new avenues for the rapid assembly of fluorinated libraries essential for modern drug development programs.
Mechanistic Insights into Pd-Catalyzed Defluorination and Radical Addition
The core of this innovation lies in the light-induced palladium-catalyzed defluorination of the aryl trifluoromethyl group to generate a reactive aryl difluoromethyl radical species. Under blue LED irradiation, the palladium catalyst, coordinated with a bulky phosphine ligand such as Xantphos, facilitates the homolytic cleavage of the C-F bond. This generated radical subsequently undergoes addition across the double bond of the non-activated olefin substrate. The resulting carbon-centered radical then captures sulfur dioxide released from the DABSO additive in the presence of zinc powder. The zinc plays a dual critical role: it acts as a reducing agent to maintain the catalytic cycle and, more importantly, stabilizes the resulting sulfinate anion intermediate, preventing its premature decomposition or side reactions. This stabilization is crucial for ensuring high conversion rates and minimizing the formation of desulfonylated byproducts.
Following the formation of the zinc sulfinate intermediate, the second step involves the removal of the light source and the addition of an electrophilic fluorinating agent, typically NFSI (N-fluorobenzenesulfonimide). This step oxidizes the sulfinate to the final sulfonyl fluoride product with high fidelity. The mechanistic pathway ensures that the fluorine atom is introduced selectively at the sulfur center without affecting other fluorine atoms present in the difluoromethyl group or other parts of the molecule. This level of chemoselectivity is paramount for maintaining the integrity of complex drug candidates. The ability to control the radical generation and trapping through light modulation provides a unique handle for process optimization, allowing manufacturers to fine-tune reaction kinetics for maximum efficiency and purity in commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize Aryl Difluoromethyl Alkyl Sulfonyl Fluoride Efficiently
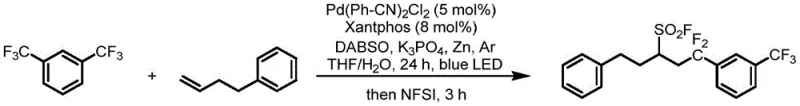
The practical implementation of this synthesis involves a straightforward two-step one-pot procedure that minimizes workup operations and solvent usage. The reaction is initiated by combining the olefin, the trifluoromethyl arene, DABSO, zinc powder, a phosphate base, and the palladium catalyst system in a mixture of THF and water. The addition of water is a critical parameter identified during optimization, as it helps dissolve inorganic salts to create a more homogeneous reaction environment, thereby enhancing molecular collision frequency and reaction rates. Following a 24-hour irradiation period, the fluorination reagent is added directly to the same vessel. Below is a representative example demonstrating the efficacy of this protocol on a phenyl-substituted olefin substrate, yielding the target sulfonyl fluoride in high purity.
- Combine olefin, m-bistrifluoromethyl aromatic hydrocarbon, DABSO, zinc powder, base, Pd catalyst, and phosphine ligand in THF/Water under argon.
- Irradiate the mixture with blue LED light at room temperature for 24 hours to generate the sulfinate intermediate.
- Remove the light source, add NFSI as the fluorine source, and stir for 3 hours to obtain the final sulfonyl fluoride product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this photocatalytic technology offers substantial strategic benefits beyond mere chemical novelty. The shift from gaseous sulfur dioxide to the solid DABSO reagent fundamentally alters the safety profile of the manufacturing process, reducing the regulatory burden and insurance costs associated with handling toxic gases. This change also simplifies logistics, as solid reagents are easier to transport and store than compressed gases, ensuring greater supply chain reliability and continuity. Furthermore, the reaction operates at ambient temperature using visible light, which translates to significantly lower energy consumption compared to traditional thermal processes that require extensive heating or cryogenic cooling. These factors collectively contribute to a more sustainable and cost-efficient manufacturing footprint.
- Cost Reduction in Manufacturing: The elimination of hazardous gas handling infrastructure and the use of earth-abundant zinc powder as a stabilizer lead to drastic simplifications in plant requirements. By avoiding expensive cryogenic equipment and high-pressure reactors, capital expenditure is significantly lowered. Additionally, the high functional group tolerance reduces the number of synthetic steps required to reach the target intermediate, minimizing raw material waste and solvent consumption. This streamlined approach allows for substantial cost savings in pharmaceutical intermediates manufacturing, making high-quality SuFEx warheads more accessible for large-scale drug production.
- Enhanced Supply Chain Reliability: The starting materials, including various olefins and trifluoromethyl benzenes, are commercially available commodity chemicals with robust global supply chains. The reliance on stable solid reagents like DABSO and NFSI mitigates the risk of supply disruptions often associated with specialty gases. The mild reaction conditions also mean that the process can be easily transferred between different manufacturing sites without requiring specialized retrofitting, ensuring consistent supply availability. This flexibility is crucial for maintaining uninterrupted production schedules for critical active pharmaceutical ingredients.
- Scalability and Environmental Compliance: The use of visible light photocatalysis is inherently scalable, particularly when adapted to continuous flow chemistry systems which offer superior light penetration and heat transfer. The aqueous component in the solvent system aids in the dissolution of inorganic byproducts, simplifying the downstream purification and wastewater treatment processes. This aligns with increasingly stringent environmental regulations regarding organic solvent discharge and heavy metal waste. The process generates minimal hazardous waste, supporting corporate sustainability goals and reducing the environmental compliance costs associated with chemical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented synthesis method. These insights are derived directly from the experimental data and optimization studies detailed in the patent documentation, providing clarity on reaction parameters and substrate compatibility. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this technology into their existing production pipelines.
Q: What is the advantage of using DABSO over sulfur dioxide gas?
A: DABSO is a solid sulfur dioxide surrogate that is safer and easier to handle than toxic SO2 gas, facilitating safer commercial scale-up.
Q: Does this method tolerate complex functional groups?
A: Yes, the protocol demonstrates excellent tolerance for esters, amides, heterocycles, and halogens, making it ideal for late-stage functionalization.
Q: What is the role of Zinc powder in this reaction?
A: Zinc powder acts as a reducing metal that stabilizes the sulfinate anion intermediate, preventing decomposition and ensuring high yields during fluorination.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aryl Difluoromethyl Alkyl Sulfonyl Fluoride Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of SuFEx chemistry in accelerating drug discovery timelines. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop to plant is seamless and efficient. Our state-of-the-art facilities are equipped with advanced photocatalytic reactors and rigorous QC labs capable of meeting stringent purity specifications required for clinical and commercial supplies. We are committed to delivering high-purity aryl difluoromethyl alkyl sulfonyl fluorides that empower your research and development efforts with reliable, high-quality chemical building blocks.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of adopting this greener, more efficient methodology. We encourage potential partners to contact us for specific COA data and route feasibility assessments, ensuring that your supply chain is optimized for both performance and cost-effectiveness in the competitive landscape of fine chemical intermediates.
