Advanced 4-Step Synthesis of Phthiobuzone Intermediate for Scalable Antiviral Production
Introduction to Next-Generation Antiviral Intermediate Manufacturing
The pharmaceutical landscape continuously demands more efficient and safer pathways for producing critical active pharmaceutical ingredients (APIs) and their precursors. Patent CN109988095B introduces a transformative methodology for synthesizing 3-phthalimide-2-oxo-n-butyraldehyde bis-thiosemicarbazone, widely known as Phthiobuzone or Ftibamzone. This compound serves as a potent antiviral agent, particularly effective against Chlamydia trachomatis and herpes viruses, functioning by inhibiting viral DNA synthesis. The significance of this patent lies not merely in the molecule itself, but in the radical simplification of its production architecture. By shifting from legacy multi-step processes involving hazardous halogens to a streamlined four-step sequence starting from abundant amino acids, this technology addresses critical bottlenecks in cost, safety, and environmental compliance. For procurement leaders and R&D directors, this represents a pivotal opportunity to secure a more resilient supply chain for antiviral therapeutics.
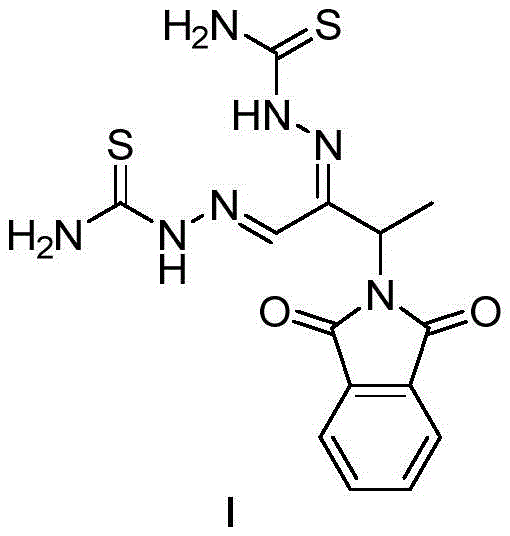
As a reliable pharmaceutical intermediate supplier, understanding the structural integrity and synthetic accessibility of such molecules is paramount. The new route leverages the chirality and availability of alanine, coupling it with N-ethoxycarbonyl phthalimide to build the core scaffold with high precision. This strategic pivot away from petrochemical-derived ketones towards bio-based amino acid derivatives aligns with modern green chemistry principles while simultaneously enhancing the economic viability of the final product. The following analysis dissects the technical merits of this innovation, contrasting it with historical methods to highlight its superiority in an industrial context.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of Phthiobuzone has been plagued by inefficient and hazardous synthetic strategies that pose significant operational risks. One prominent legacy method, documented in earlier literature, relies on a five-step synthesis initiating from 2-butanone. This pathway necessitates two distinct bromination steps to functionalize the carbon chain, introducing elemental bromine into the process stream.
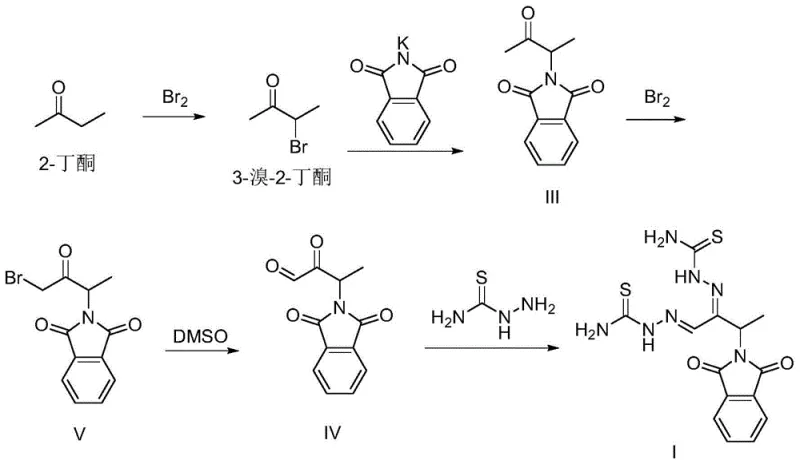
The use of bromine is notoriously problematic in large-scale manufacturing due to its extreme corrosivity, which accelerates the degradation of reactor vessels and piping, leading to frequent downtime and high capital expenditure for specialized corrosion-resistant equipment. Furthermore, brominated intermediates are often toxic and environmentally persistent, complicating waste disposal and increasing regulatory burdens. Another alternative six-step route utilizes acetaldehyde and nitroethane, involving an iron powder reduction step. While chemically feasible, this method suffers from severe practical drawbacks; the fine iron powder creates a sludge that is exceptionally difficult to filter, trapping product and reducing overall yield. These legacy processes exemplify the technological stagnation that increases the cost of goods sold (COGS) and jeopardizes supply continuity for essential antiviral medications.
The Novel Approach
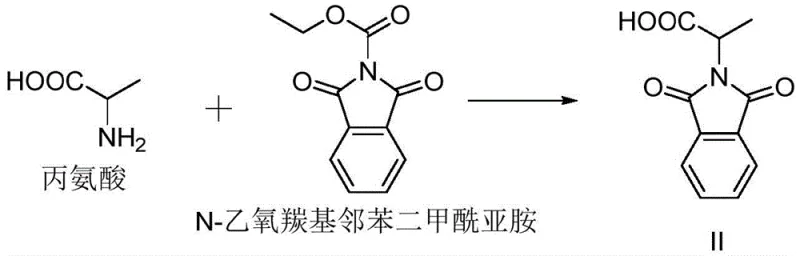
In stark contrast, the methodology disclosed in CN109988095B offers a sophisticated solution that bypasses these historical pitfalls through a concise four-step design. The innovation begins with the coupling of alanine and N-ethoxycarbonyl phthalimide, a reaction that proceeds under mild alkaline conditions to form the protected amino acid intermediate. This initial step sets a foundation of high atom economy and safety, avoiding the introduction of heavy metals or halogens at the outset.
Subsequent transformations utilize methyllithium for precise carbon chain extension and selenium dioxide for selective oxidation, culminating in a condensation reaction with thiosemicarbazide. This route eliminates the need for corrosive bromine and difficult-to-remove iron catalysts entirely. By reducing the step count from five or six down to four, the process inherently minimizes material loss at each stage, theoretically boosting the overall yield without requiring complex purification protocols. For a cost reduction in API manufacturing context, this simplification translates directly to lower solvent consumption, reduced energy usage for heating and cooling cycles, and a smaller physical footprint for production facilities, making it an ideal candidate for modern, sustainable chemical manufacturing.
Mechanistic Insights into Aminoacylation and Selective Oxidation
The core of this synthetic breakthrough lies in the precise control of reactivity during the aminoacylation and oxidation phases. The first step involves the nucleophilic attack of the amino group of alanine on the activated carbonyl of N-ethoxycarbonyl phthalimide. Conducted in an aqueous or alcoholic medium with inorganic bases like sodium carbonate, this reaction is highly exothermic but manageable, typically proceeding efficiently at temperatures between 10°C and 20°C. The choice of base is critical; carbonates provide sufficient deprotonation to drive the reaction forward without inducing racemization or hydrolysis of the sensitive phthalimide ring, ensuring the stereochemical integrity of the alanine moiety is preserved where necessary for downstream biological activity.
Following the formation of the ketone intermediate via methyllithium addition, the process employs selenium dioxide (SeO2) for the oxidation of the methyl group to an aldehyde. This is a mechanistically elegant solution to a common problem: achieving selectivity. Stronger oxidants might over-oxidize the substrate to a carboxylic acid or degrade the phthalimide ring. SeO2, however, operates via an ene-reaction mechanism that specifically targets allylic or benzylic-like positions, or in this case, the alpha-methyl group adjacent to the carbonyl, facilitating the conversion to the aldehyde with high fidelity. This selectivity is crucial for impurity control, as it prevents the formation of acidic byproducts that would complicate the final condensation step and require extensive chromatographic purification, thereby streamlining the path to high-purity pharmaceutical intermediates.
How to Synthesize 3-Phthalimide-2-oxo-n-butyraldehyde Bis-thiosemicarbazone Efficiently
Implementing this synthesis requires careful attention to stoichiometry and temperature control, particularly during the organometallic addition and oxidation stages. The patent outlines specific molar ratios, such as using a slight excess of methyllithium (2.2 equivalents) to ensure complete conversion of the carboxylic acid precursor, and maintaining the oxidation temperature between 90°C and 100°C to drive the selenium-mediated transformation to completion. The detailed standardized synthesis steps see the guide below.
- Perform aminoacylation of alanine with N-ethoxycarbonyl phthalimide under alkaline conditions to form 2-phthalimidopropionic acid.
- React the resulting acid with methyllithium at low temperatures to generate 3-phthalimide-2-butanone via nucleophilic addition.
- Oxidize the methyl group using selenium dioxide in dioxane to yield the key aldehyde intermediate, 3-phthalimide-2-oxo-n-butyraldehyde.
- Condense the aldehyde intermediate with thiosemicarbazide in an alcoholic solvent to finalize the bis-thiosemicarbazone structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers tangible strategic benefits that extend beyond simple chemistry. The primary advantage is the drastic simplification of the supply chain risk profile. By eliminating elemental bromine, manufacturers remove a highly regulated and hazardous material from their inventory, reducing insurance premiums and safety training costs. Furthermore, the removal of iron powder filtration steps resolves a major bottleneck in batch cycle times, allowing for faster throughput and more predictable delivery schedules for downstream API producers.
- Cost Reduction in Manufacturing: The elimination of corrosive reagents significantly extends the lifespan of production equipment, deferring capital expenditures on reactor replacements. Additionally, the reduction in synthesis steps from six to four inherently lowers utility consumption and solvent waste disposal costs, driving down the overall cost of goods without compromising quality.
- Enhanced Supply Chain Reliability: Starting materials such as alanine and phthalimide derivatives are commodity chemicals with robust global supply networks, unlike specialized brominated intermediates which may have limited suppliers. This commoditization of raw materials ensures greater stability in pricing and availability, shielding the production schedule from raw material shortages.
- Scalability and Environmental Compliance: The process generates significantly less hazardous waste, particularly avoiding brominated organic waste streams which are expensive to treat. This aligns with increasingly stringent environmental regulations, facilitating easier permitting for capacity expansion and ensuring long-term operational continuity in regulated markets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this method outperforms legacy techniques in terms of safety, efficiency, and product quality.
Q: How does this new synthesis route improve upon traditional bromination methods?
A: Traditional methods rely on hazardous bromination steps which are highly corrosive to equipment and generate toxic byproducts. This new route utilizes aminoacylation and oxidation, eliminating the need for elemental bromine and significantly reducing equipment maintenance costs and safety risks.
Q: What are the scalability advantages regarding purification?
A: Previous nitro-reduction routes suffered from difficult filtration due to fine iron powder residues. The new method avoids heterogeneous metal reductions entirely, utilizing homogeneous reactions that simplify work-up procedures and ensure higher purity suitable for GMP manufacturing.
Q: Is the selenium dioxide oxidation step safe for large-scale production?
A: Yes, the oxidation using selenium dioxide is conducted in a controlled dioxane-water system at moderate temperatures (90-100°C). This specific oxidant offers high selectivity for the methyl group, minimizing over-oxidation impurities and ensuring a robust profile for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Phthalimide-2-oxo-n-butyraldehyde Bis-thiosemicarbazone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the adoption of advanced synthetic routes like the one described in CN109988095B is essential for maintaining competitiveness in the global antiviral market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this four-step process are fully realized in a GMP-compliant environment. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Phthiobuzone intermediate meets the exacting standards required for pharmaceutical formulation, providing our partners with a secure and high-quality supply source.
We invite you to collaborate with us to leverage this innovative technology for your antiviral drug development programs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how switching to this optimized synthesis can enhance your project's economics and timeline.
